Standard capsules deliver under 2% bioavailability. LVLUP nasal, VERISORB sublingual, and injection all sit in different ranges. Here's the data.
TL;DR: Standard peptide capsules: under 2% bioavailability. Nasal sprays bypass the gut but face mucociliary clearance limits. VERISORB sublingual reaches up to 24.1% relative to subcutaneous injection in formulation research. Subcutaneous injection is the reference standard. Route determines systemic exposure as directly as dose.
When you search for the best oral peptide bioavailability, you're really asking which format actually gets the compound into your bloodstream. The honest answer starts with a number most peptide brands won't highlight: standard capsules deliver under 2% bioavailability, meaning out of every 100 milligrams, 98 or more never reach your blood. This content is published for research purposes only.
Four formats are competing to solve that problem: standard capsules, nasal sprays like those from LVLUP Health, sublingual delivery systems like VERO's VERISORB platform, and subcutaneous injection — which is the reference standard everything else gets measured against. Their numbers aren't in the same ballpark. Here's the comparison laid out clearly.
The Problem Every Format Is Trying to Solve
Bioavailability — how much of what you take actually reaches your bloodstream — is the only number that matters here. The label tells you what's in the bottle. Your blood only sees a fraction of that.
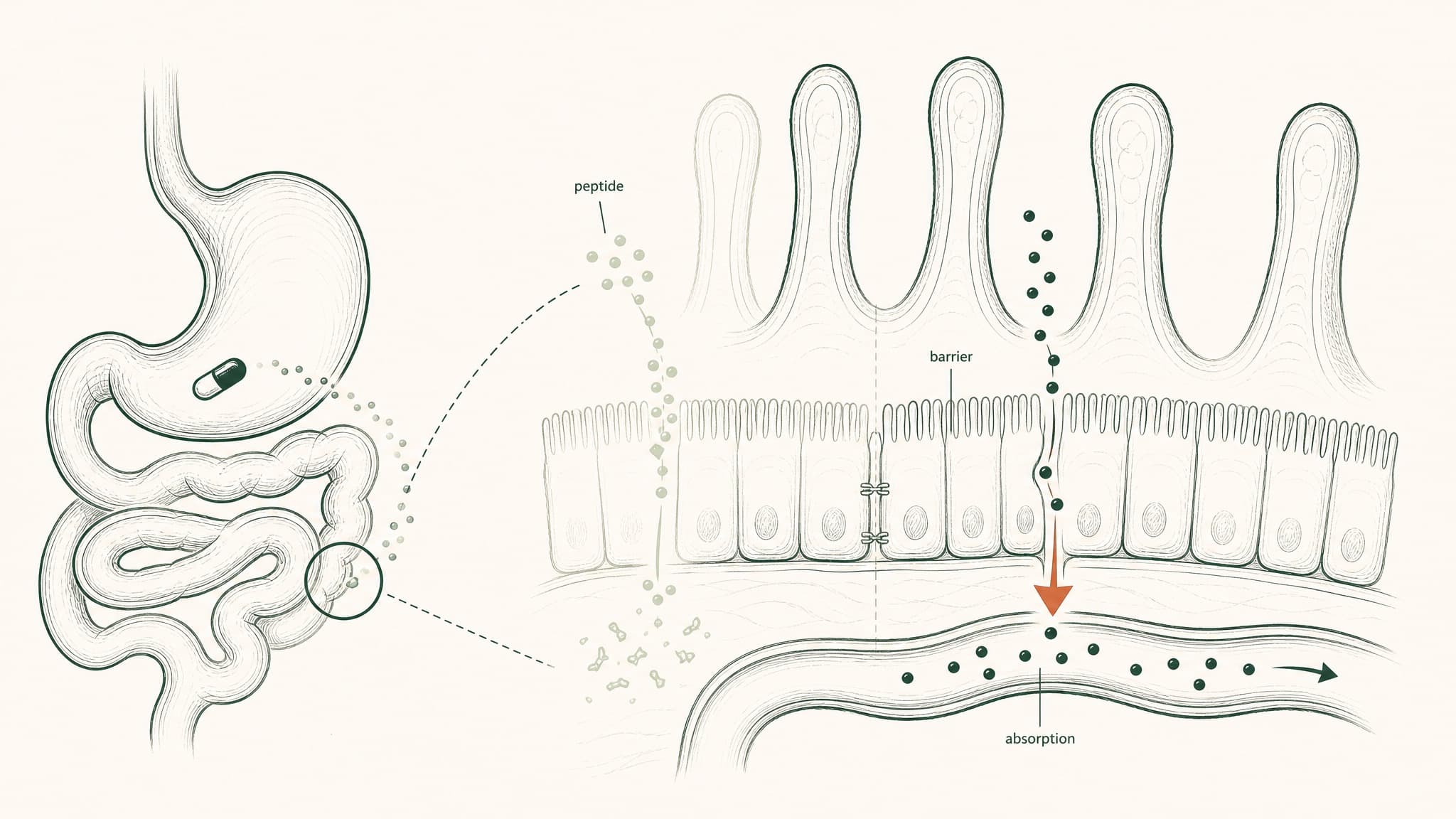
Your digestive system is a precision protein-dismantling machine. It wasn't built to block peptide supplements specifically — it was built to process food. But peptides are chains of amino acids, just like food protein, and the gut runs both through the same destruction sequence.
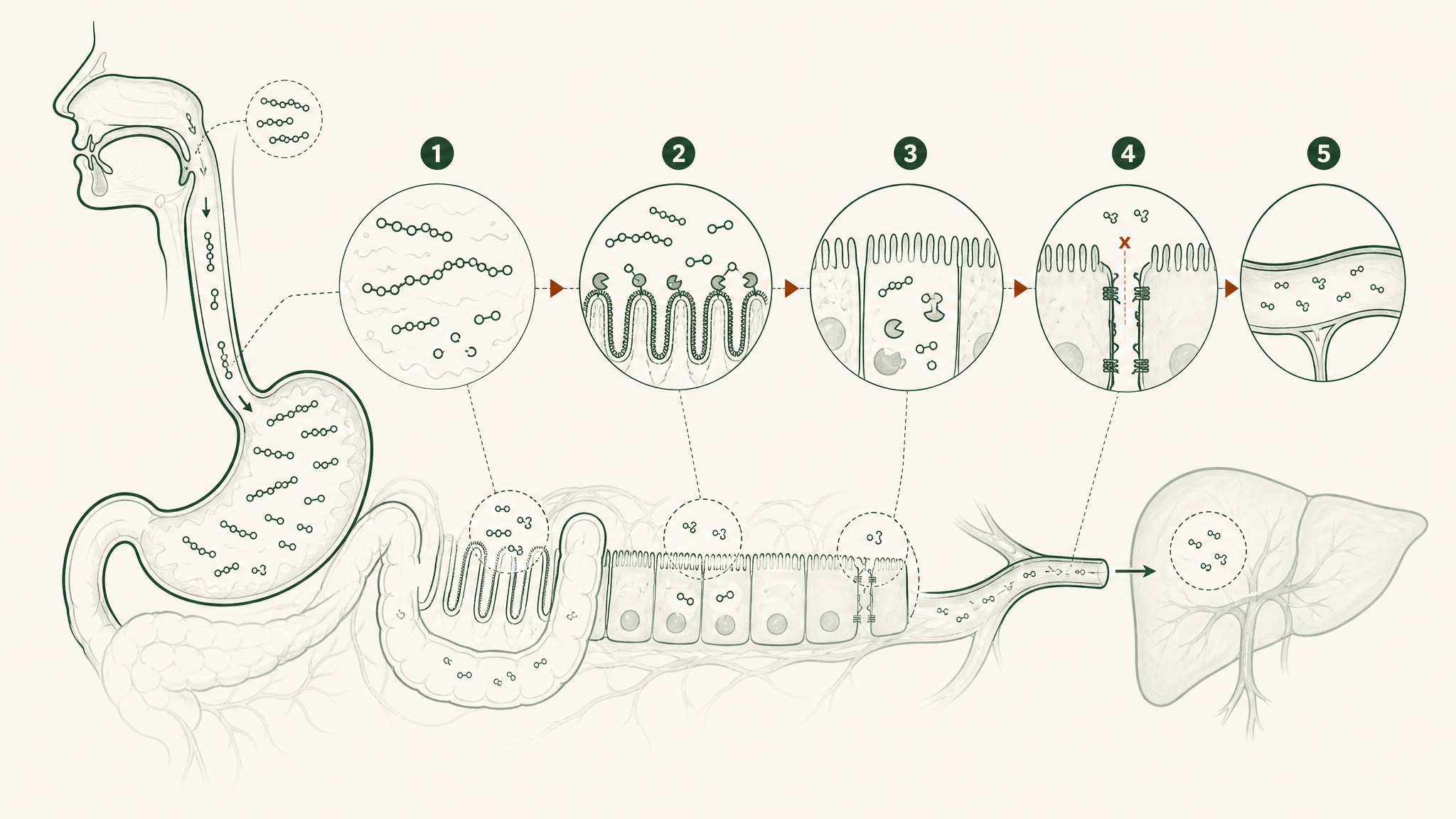
Five sequential barriers in your gut destroy most peptides before they reach your bloodstream.
Here's what each stage in that sequence actually does:
- Gastric acid (pH 1.5–3.5, roughly battery acid territory): begins to unfold peptide molecular architecture; produces pepsin, an enzyme whose entire purpose is cutting peptide bonds
- Brush-border peptidases: a second enzyme layer coating the intestinal wall surface, continuing the dismantling of anything that survived the stomach
- Epithelial barrier: a continuous cell layer that functions as both a physical wall and a biochemical checkpoint, with its own intracellular enzymes that process anything attempting to cross
- Tight junctions: protein structures sealing the gaps between intestinal cells; block most peptide-sized molecules from slipping through
- Hepatic first-pass metabolism: everything absorbed from the gut routes to the liver before the rest of your body sees it; hepatic proteases take a final cut
Each barrier works on whatever the previous stage left over. They compound on each other — they don't subtract a fixed percentage of the original dose. That's how you arrive at sub-2-percent bioavailability even from a well-manufactured capsule.
Standard Peptide Capsules: Under 2%, Full Stop
Unmodified oral peptides hit this wall consistently. The documented outcome for standard capsule formats is below 1 to 2 percent, driven by the sequential effect of enzymatic degradation, poor cellular membrane permeability, and hepatic first-pass metabolism operating as compounding obstacles (Renukuntla et al. 2013, Int J Pharm, PMC3680128; Verma et al. 2021, Drug Dev Res, PMID 33988872).
The gut does have one active uptake mechanism for intact peptides: a transporter protein called PepT1 that shuttles peptide fragments across the intestinal wall. The limitation is its substrate window — PepT1 only transports di-peptides and tri-peptides, chains of exactly two or three amino acids, because its biological function is absorbing the output of the gut's own dismantling process (Freeman 2015, World J Gastrointest Pharmacol Ther, PMC4419090). BPC-157 is 15 amino acids long. Most research peptides are far outside this window. The one transporter that could theoretically help is built for the wrong chain length.
The pharmaceutical ceiling is worth understanding. The best any oral format has ever achieved in commercial development is 0.8 percent — oral semaglutide (Rybelsus), engineered with a proprietary absorption enhancer called SNAC that creates a localised gastric pH rise, taken alone, 30 minutes before food, no more than 120ml of water (Overgaard et al. 2021, Clin Pharmacokinet, PMID 33969456). Semaglutide works at 0.8% because it's pharmacologically potent at very low concentrations. That headroom doesn't transfer to most research peptides. And no standard supplement capsule uses SNAC technology.
The capsule category's practical verdict: it's not that the compound inside is fake. It's that the format can't reliably get the compound into your bloodstream, regardless of what's on the label.
LVLUP Nasal Peptides: When You Skip the Gut but Face a Different Constraint
LVLUP Health is among the more visible brands using intranasal delivery — nasal sprays rather than capsules or injections. The appeal of this route is real and it's not marketing: nasal delivery genuinely bypasses the gastrointestinal tract. No gastric acid, no pepsin, no intestinal enzymes, no epithelial barrier, no tight junctions.
Users report that nasal formats feel noticeably different from oral capsules — which tracks, because the absorption pathway is genuinely different. The compound is reaching the bloodstream rather than being dismantled in the gut.
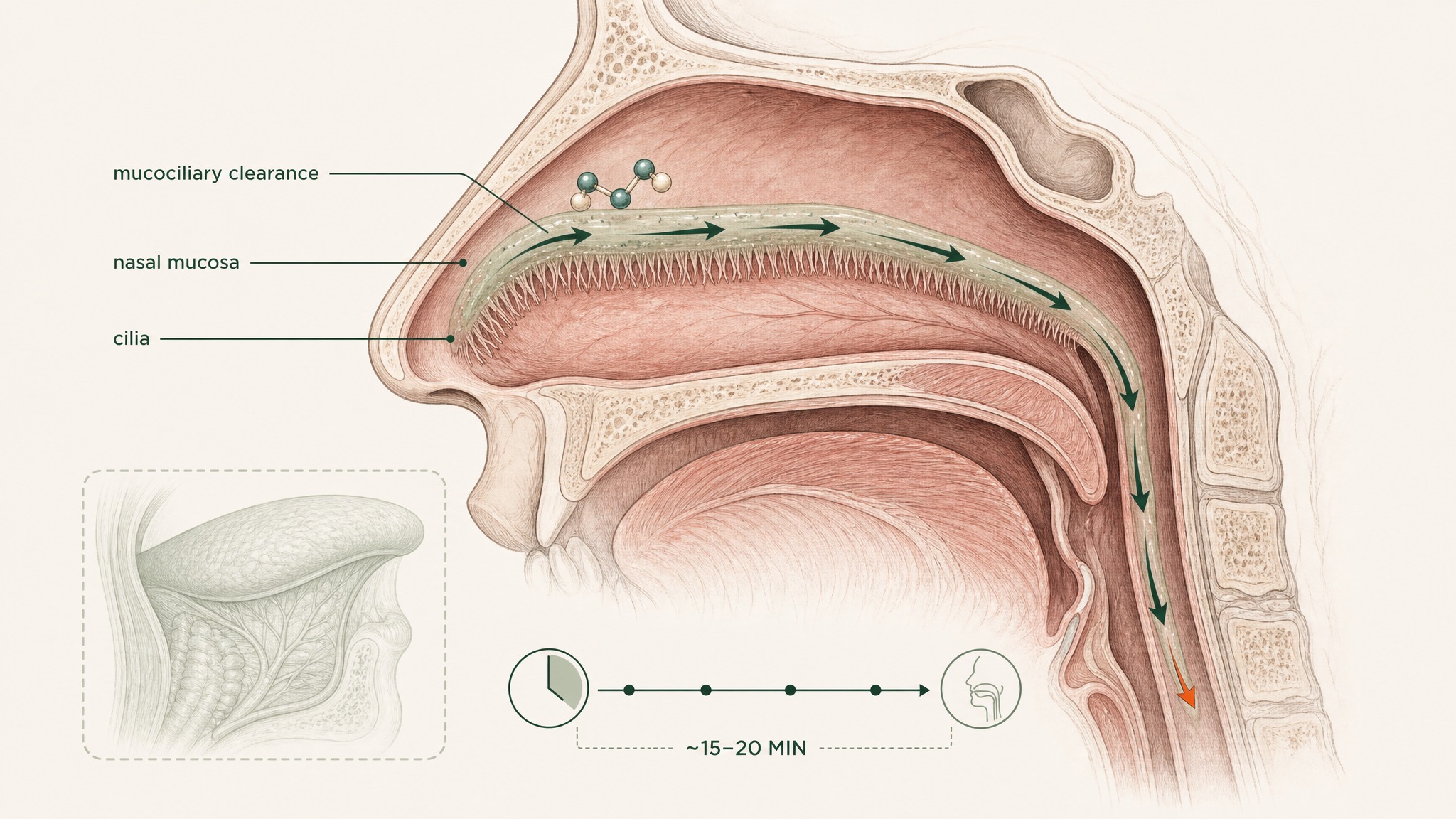
Nasal cilia sweep mucus and any peptide toward your throat within 15 to 20 minutes.
The nasal route's constraint isn't the mucosa itself. It's the biology of the nasal cavity.
- Mucociliary clearance: your nasal cavity clears material from its surface in approximately 15 to 20 minutes via the mucociliary escalator — the moving mucus layer your nasal cilia drive toward the throat. A compound has to absorb across the nasal mucosa faster than it gets cleared, or a portion gets swallowed and enters the gut anyway
- Surface area: the nasal mucosa offers less absorptive surface area than the tissue under your tongue, and the contact dynamics differ significantly from a formulation held sublingually
- Compound variability: nasal bioavailability across peptide classes varies considerably by molecular weight, lipophilicity, and formulation — there isn't a fixed route-level number the way there is for injection
Published route comparisons place nasal, sublingual, buccal, pulmonary, and oral delivery at distinct positions on both bioavailability and practicality axes (Zaman et al. 2016, Curr Pharm Des, PMID 27510479). Nasal sits meaningfully above oral on the bioavailability spectrum — the GI bypass is real — but below sublingual in the documented formulation comparisons for most compound classes, largely because mucociliary clearance limits contact time and the nasal mucosa is more keratinised than the tissue beneath your tongue.
LVLUP's formulations may produce meaningful absorption for the specific compounds they've engineered for. The point isn't to dismiss the nasal route. It's to place it accurately against the alternatives.
VERISORB Sublingual: What the Bypass Route Data Actually Shows
The anatomy under your tongue is different from anything in your nose or your gut.
The underside of your tongue is lined with non-keratinised epithelium — thinner, more permeable, and less enzymatically aggressive than any surface in your gastrointestinal tract — directly over a dense venous network. Compounds absorbed there drain into the sublingual and lingual veins, entering the internal jugular vein and superior vena cava, which means they reach systemic circulation before the liver processes them (Zhang, Zhang & Streisand 2002, Clin Pharmacokinet, PMID 12126458). The stomach, intestinal enzymes, epithelial barrier, tight junctions, and hepatic first-pass metabolism are all out of the pathway.
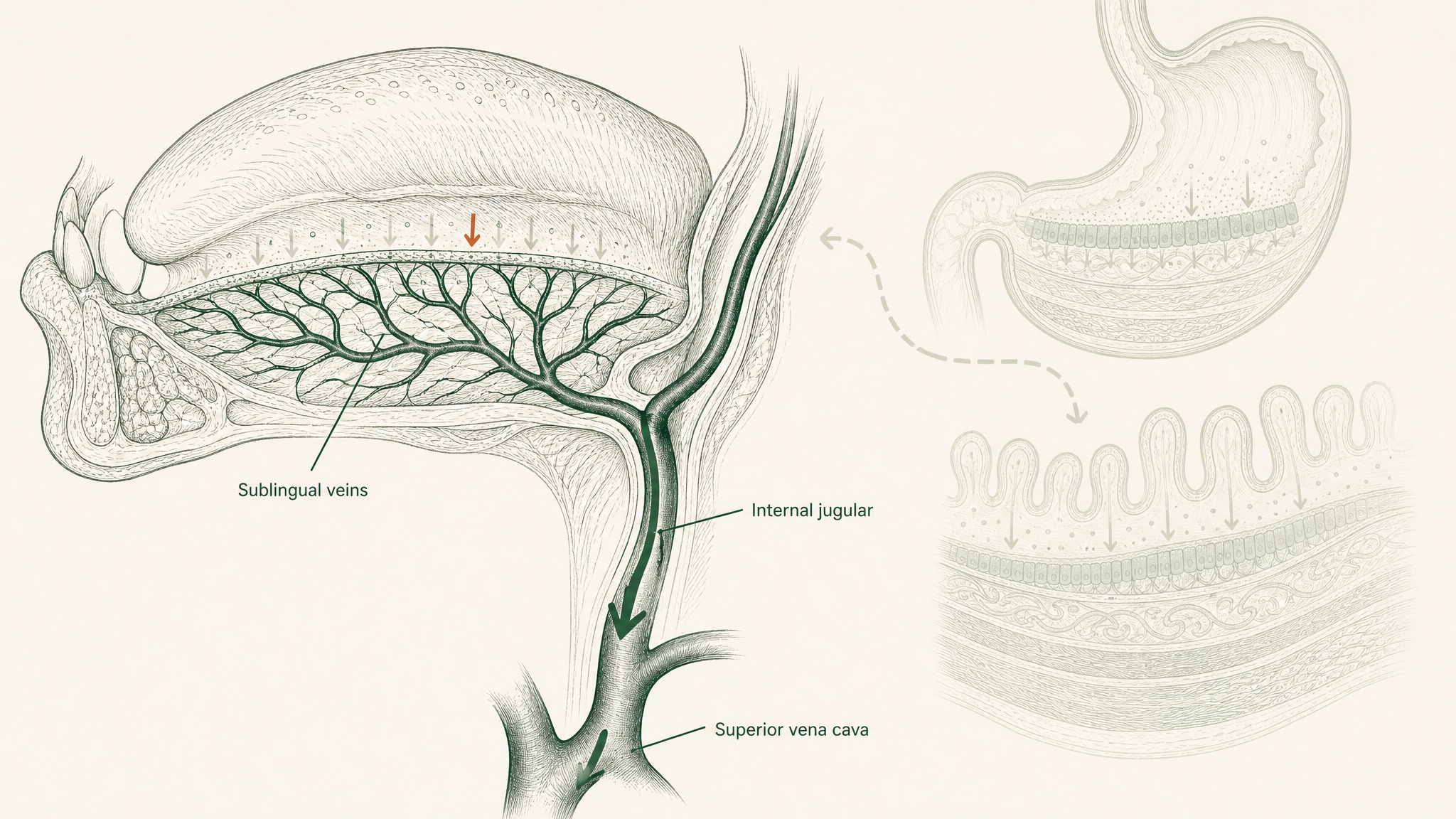
Sublingual tissue is thinner and more permeable than the stomach or intestines, with direct access to veins that bypass the liver.
That's five barriers removed, not one.
What the formulation data shows for this route: sublingual insulin nanoparticles achieved absolute pharmacological availability of 20.2% and absolute bioavailability of 24.1% relative to a subcutaneous injection reference in diabetic rat models (Patil & Devarajan 2016, Drug Deliv, PMID 24901208). Those are compound-specific, formulation-specific numbers from an animal model — they don't transfer directly across species or peptide types. What they establish is the order-of-magnitude difference the sublingual route produces when the formulation is engineered specifically for the route.
Not every peptide crosses the sublingual mucosa equally well without formulation help. Lower molecular weight, more lipophilic compounds can absorb across the mucosal surface more readily. Larger or more hydrophilic molecules need additional engineering: mucoadhesive polymers that hold the compound against the mucosal surface long enough to cross, permeation enhancers matched to the peptide's molecular characteristics, vehicle chemistry designed specifically for the sublingual environment.
An unformulated peptide placed under the tongue doesn't automatically absorb, any more than a capsule crosses the intestinal wall. The sublingual route removes five barriers from the oral route; a purpose-built formulation addresses the one that remains. That's the engineering VERO's VERISORB platform is built around.
Members experience the practical side of this most clearly in extended protocols. A delivery format that achieves meaningful systemic exposure without requiring injection changes what actually gets followed through across six or twelve weeks consistently.
Subcutaneous Injection: The Number Everything Else Is Measured Against
Subcutaneous injection places a compound directly into vascularised tissue just beneath your skin. The GI tract is never in the picture. No gastric acid, no proteases, no epithelial barrier, no hepatic first-pass cut. The compound deposits where blood vessels are already present.
This is why injection remains the reference standard for peptide delivery in research — the dose-to-exposure relationship is reliable and predictable (Zhang et al. 2020, Int J Pharm, PMID 32622810). When a research endpoint depends on specific systemic concentrations, subcutaneous injection is what's used because it consistently delivers.
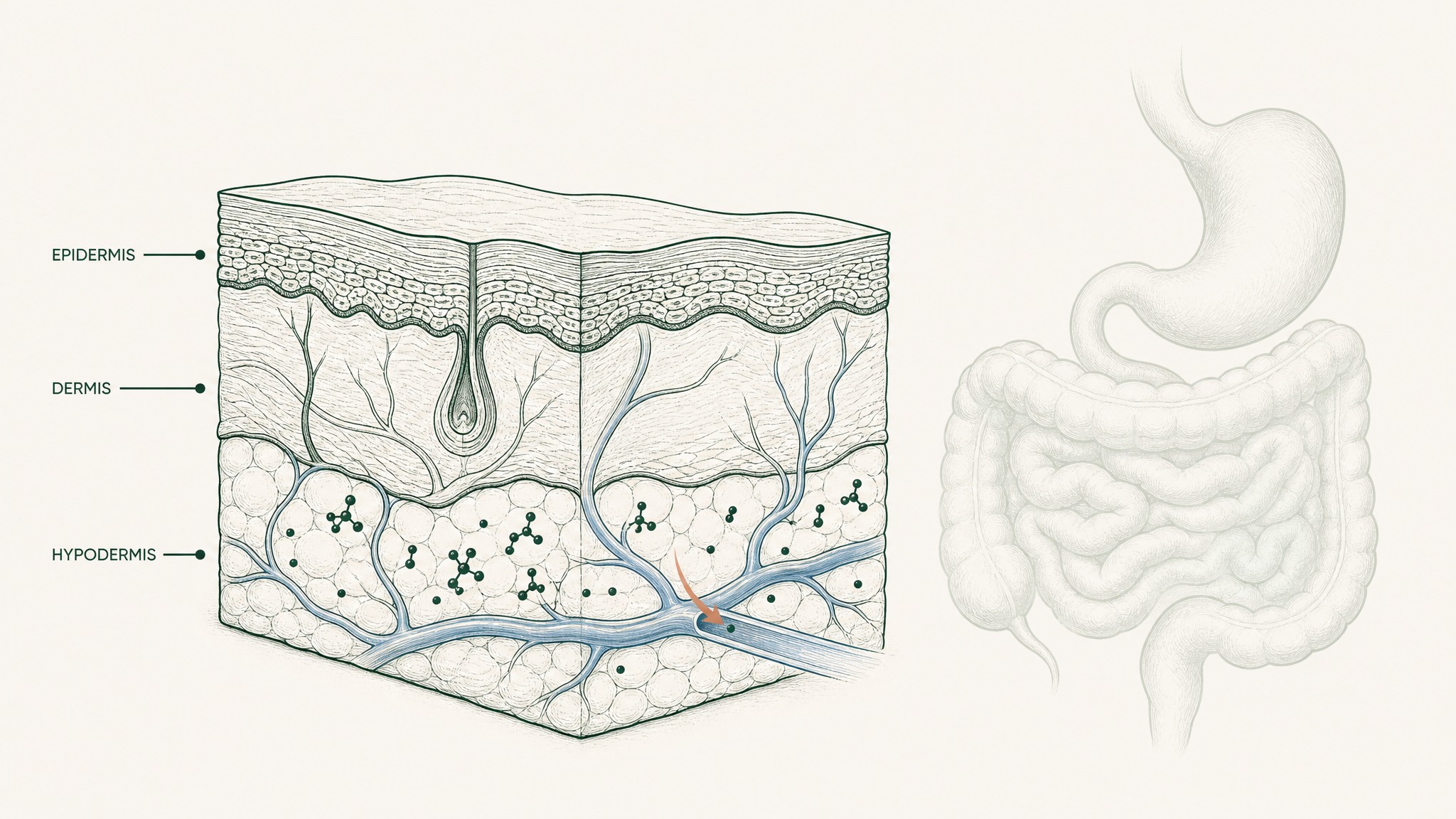
Subcutaneous injection deposits peptides directly into tissue where blood vessels are already present.
Injectable protocols carry their own practical constraints: sterile preparation, consistent technique, site rotation, tolerability across months of a protocol. None of these affect the pharmacokinetics, but they do affect what actually gets done. The delivery-science question the field keeps returning to is whether a non-injectable format can produce meaningful systemic exposure for the compounds and endpoints researchers care about. That's the gap VERISORB and nasal delivery are both attempting to fill, at different points on the spectrum.
The Four-Format Head-to-Head
| Format | Bypasses GI Tract | Bypasses Liver First-Pass | Documented Bioavailability |
|---|---|---|---|
| Standard capsules | No | No | <2% consistently (Renukuntla et al. 2013) |
| Nasal spray (LVLUP) | Yes | Yes (mostly) | Compound-dependent; limited by mucociliary clearance (Zaman et al. 2016) |
| Sublingual — VERISORB | Yes | Yes | Up to 24.1% vs subcutaneous in peptide formulation research (Patil & Devarajan 2016) |
| Subcutaneous injection | Yes (bypasses entirely) | Yes | Reference standard |
A few notes on what the table doesn't capture:
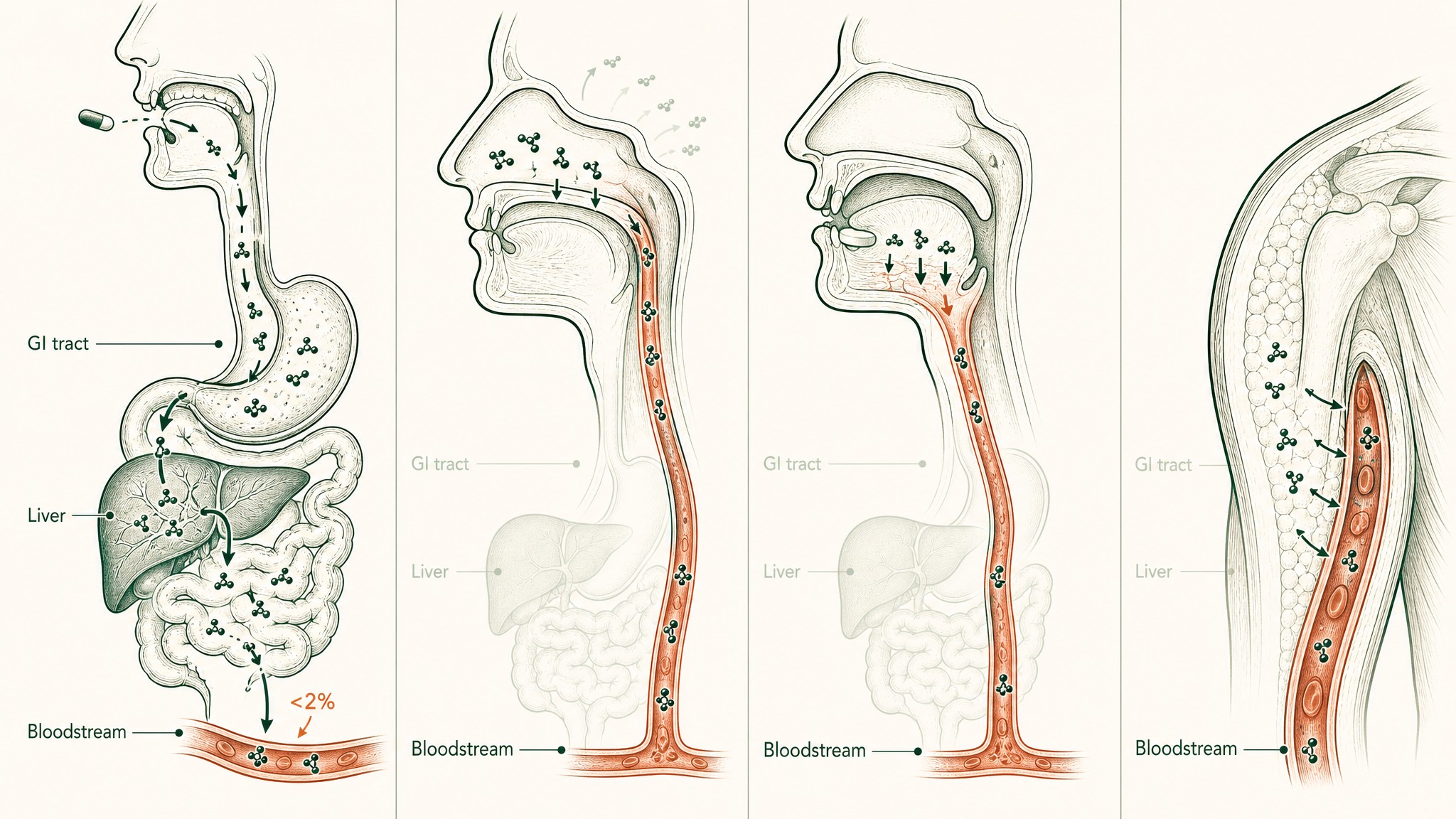
How four delivery routes get peptides into your bloodstream with vastly different efficiency.
- Nasal bioavailability varies more across compounds than sublingual. Mucociliary clearance is the variable that makes this route compound-dependent in a way the sublingual route is less prone to.
- The 24.1% sublingual figure comes from a specific formulation study. A sublingual product without purpose-built formulation engineering won't reach the upper range of what the route is capable of.
- Injection isn't something to "beat" — it's the benchmark. The value of the non-injectable formats is practical: meaningful exposure without the compliance burden of a sustained injection protocol.
What This Means When You're Designing a Protocol
Route selection determines systemic peptide exposure as directly as the label dose does. A compound at 10mg via standard oral capsule with 1.5% bioavailability produces fundamentally different blood levels than the same compound at the same dose via VERISORB sublingual. That gap doesn't close by using a higher-quality capsule. It's structural to the format.
The practical question for most people isn't which format wins on a single pharmacokinetic data point — it's which format achieves meaningful systemic exposure while being sustainable across the protocol duration. Consistent dosing is its own variable. A protocol that requires daily injection may produce better single-dose numbers but lower average exposure if adherence drops off by week four. A format that gets done reliably, every day, across the full protocol duration often outperforms the theoretically superior option in practice.
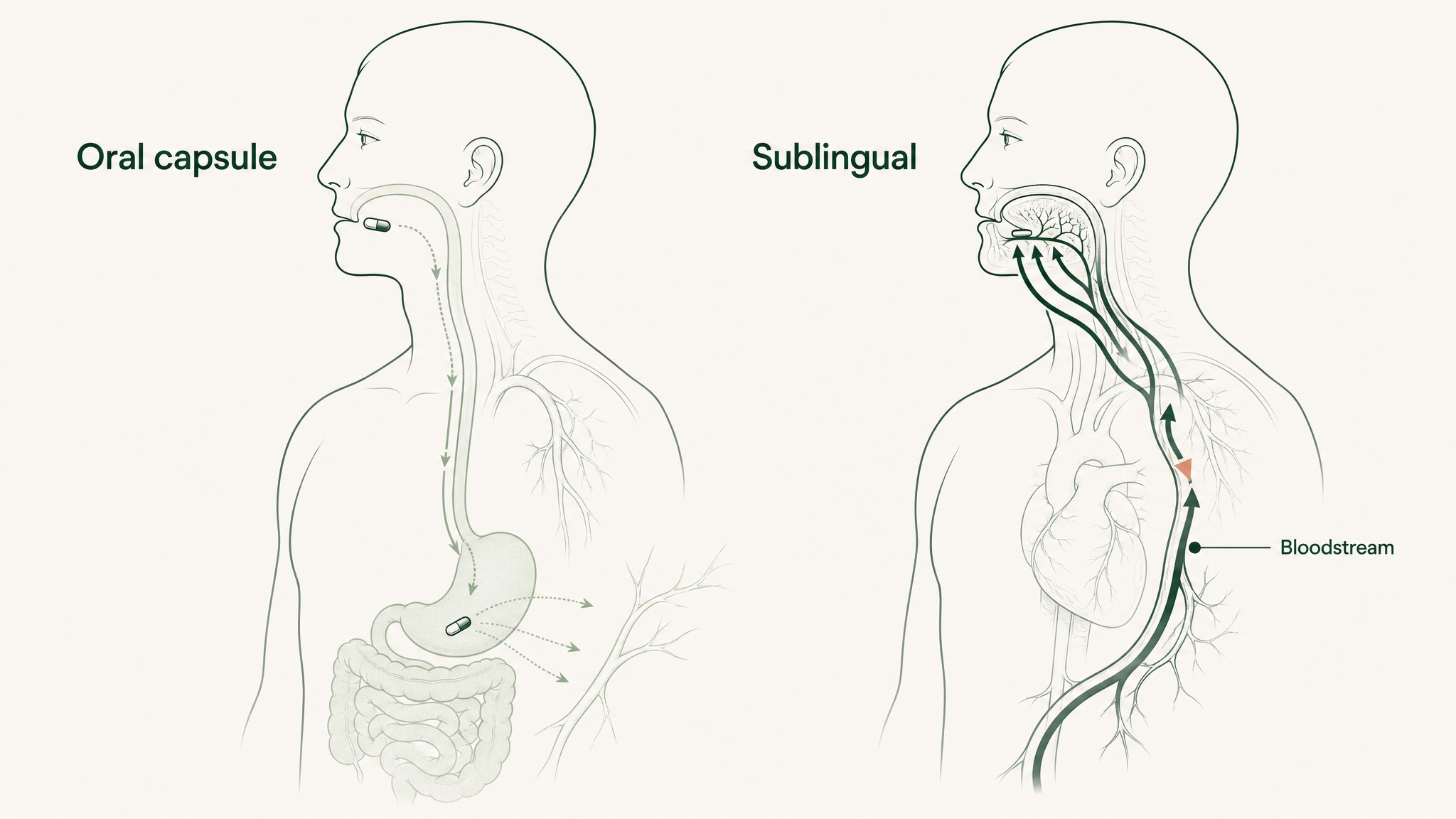
Same peptide dose produces different blood levels depending on whether it's swallowed or placed under the tongue.
For non-injectable formats, the sublingual route with purpose-built delivery engineering has the strongest documented bioavailability data of the currently available options — and it's the route VERISORB is specifically designed to exploit.
Key Takeaways
- Standard oral peptide capsules document bioavailability below 1 to 2 percent. The gut's multi-stage destruction sequence — gastric acid, pepsin, brush-border peptidases, epithelial barrier, tight junctions, hepatic first-pass — compounds across each stage rather than adding up (Renukuntla et al. 2013, PMC3680128; Verma et al. 2021, PMID 33988872).
- The PepT1 intestinal transporter only accepts di-peptides and tri-peptides. Most research peptides exceed this chain-length threshold and don't benefit meaningfully from this pathway (Freeman 2015, PMC4419090).
- The best oral peptide bioavailability pharmaceutical engineering has achieved commercially is 0.8% — oral semaglutide with SNAC under strict dosing conditions (Overgaard et al. 2021, PMID 33969456). Standard supplement capsules sit below this ceiling.
- Nasal spray formats (LVLUP and others) bypass the GI tract but face mucociliary clearance limitations that reduce contact time and produce compound-dependent variability in bioavailability outcomes (Zaman et al. 2016, PMID 27510479).
- Sublingual delivery (VERISORB) bypasses the GI tract and hepatic first-pass metabolism via the sublingual mucosal venous network (Zhang, Zhang & Streisand 2002, PMID 12126458). Purpose-formulated sublingual peptide preparations reach up to 24.1% bioavailability relative to subcutaneous in peptide formulation research (Patil & Devarajan 2016, PMID 24901208).
- Subcutaneous injection remains the research reference standard — reliable, predictable dose-to-exposure, and the benchmark all other formats are measured against (Zhang et al. 2020, PMID 32622810).
- Route selection determines systemic peptide exposure as directly as the label dose. Building a protocol without accounting for route-specific bioavailability is designing against the data.
Ready to see what purpose-formulated sublingual delivery looks like in practice? Start with the RESTORE Protocol →
This content is published for research purposes only and does not constitute medical advice. It is not intended to diagnose, treat, cure, or prevent any disease. Anyone considering a peptide research protocol should consult a qualified clinician before beginning.
RESTORE™
BPC-157Engineered around 500mcg BPC-157. Accelerates deep tissue and joint recovery
Clinical Context
Important Notice: VERO protocols are nutritional and systemic optimisation formats. They are not intended to diagnose, treat, cure, or prevent any medical condition. These statements have not been evaluated by the Food and Drug Administration.