Some peptides decline measurably with age — and the research on supplementing them spans skin repair, cellular longevity, and metabolic function.
Age-supportive peptides are signalling molecules your body produces naturally and measurably less of as the decades pass. Three of the most researched in this category are GHK-Cu (a copper tripeptide first isolated from human blood), Epithalon (a synthetic tetrapeptide linked to telomere biology), and MOTS-c (a 16-amino-acid peptide encoded in your mitochondrial DNA). All three are studied for research purposes only. None are approved therapeutic compounds. What they share is a documented connection to biological processes that change substantially as your body ages, and a growing body of preclinical research asking what happens when you restore them experimentally.
What "Age-Supportive" Actually Means in Research Terms
Let's be precise about the phrase first, because the alternative, "anti-aging," carries a lot of marketing noise and almost no scientific specificity.
What researchers actually study is the biology of biological decline: which molecules fall in concentration as you get older, what cellular functions those molecules support, and whether restoring them has any measurable effect on those functions in cellular and animal models.
Age-supportive peptides aren't a magic category. They're signalling molecules that appear repeatedly across the longevity research literature because they happen to do two things: decline naturally with age, and show activity in biological processes associated with how your cells repair, divide, and maintain function over time.
Think of it this way. Your body runs maintenance routines at the cellular level constantly — repairing DNA, turning over structural proteins like collagen, managing energy production, keeping telomeres (the protective caps at the ends of your chromosomes) intact. Peptides are some of the molecular tools it uses to coordinate those routines. When those peptides fall below a certain concentration, some of the coordination starts to slip. Not dramatically. Not all at once. Just gradually, measurably, and across multiple systems at the same time.
That's what the research in this space is actually about. Not reversing time. Just understanding which signalling molecules matter, how much they decline, and what happens when you restore them in controlled models. Research suggests the three compounds below are among the most rigorously documented in this framework.
GHK-Cu: The Signalling Molecule Your Body Stops Prioritising at 40
GHK-Cu is a tripeptide (three amino acids linked in a chain) called glycine-histidine-lysine, bound to a copper ion. Your body produces it naturally and circulates it in your blood. Loren Pickart and M.M. Thaler first isolated it from human serum in 1973 (PMID 4349963).
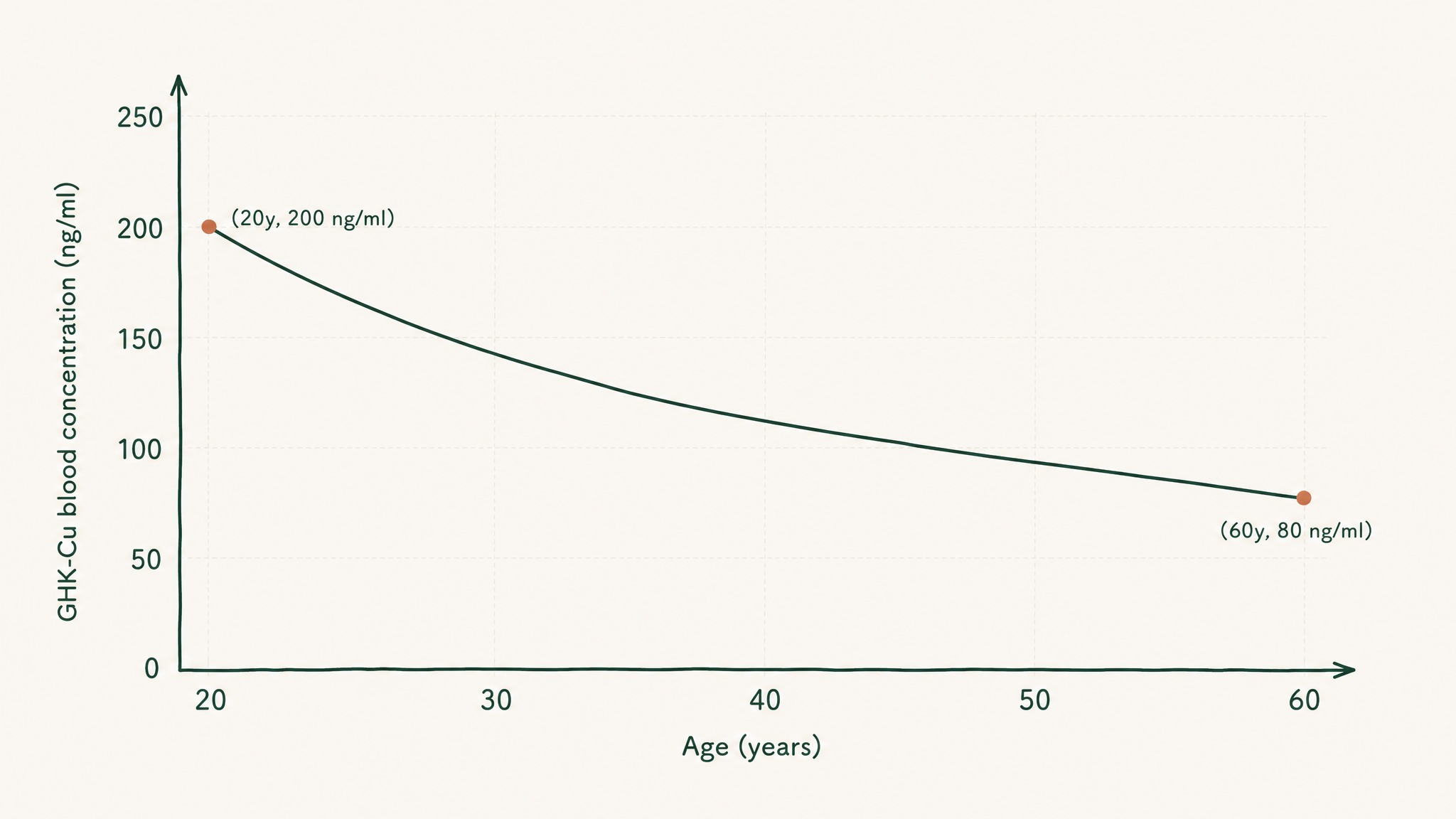
Here's the detail that makes GHK-Cu particularly relevant to longevity research: plasma GHK levels run at around 200 nanograms per millilitre in your twenties and fall to around 80 ng/mL by the time you're 60 — a roughly 60% drop across four decades (Pickart & Margolina, Int J Mol Sci 2018, PMC6073405). That's not incidental. That decline correlates with the period when your body's repair and regeneration outputs start visibly changing too.
What does GHK-Cu actually do at a molecular level? The scope is genuinely unusual for a single compound. Research suggests it modulates expression by at least 50% in 31.2% of human genes, covering tissue remodelling, antioxidant defence, anti-inflammatory signalling, and neurological maintenance in the same molecule (Pickart & Margolina, Int J Mol Sci 2018, PMC6073405). A 2014 genomic analysis confirmed GHK changed expression by at least 50% in 32.1% of 13,424 human genes across a broad panel (Pickart, Vasquez-Soltero & Margolina, BioMed Res Int 2014, PMC4180391). For context: roughly one in three genes across your entire genome, responding to a single small molecule your body already makes.
The skin research is the most clinically accessible part of the GHK-Cu literature. A placebo-controlled study found collagen production increased in 70% of women using GHK-Cu formulations over one month, outperforming vitamin C cream (50%) and retinoic acid, a vitamin A derivative widely used in skincare (40%), in the same study conditions (Pickart, Vasquez-Soltero & Margolina, BioMed Res Int 2015, PMC4508379). What makes that number interesting: vitamin C and retinoic acid are two of the most well-researched skin compounds on the market. GHK-Cu outperformed both in the same study setup.
The antioxidant findings add a separate layer of relevance. Research found a 75% reduction in lipid peroxidation (that's when free radicals attack and damage your cell membranes, like rust on metal at a cellular level) in gastric tissue samples, alongside documented suppression of TGF-beta and TNF-alpha, two pro-inflammatory signalling proteins that drive chronic systemic inflammation when they stay elevated long-term (Pickart, Vasquez-Soltero & Margolina, Oxid Med Cell Longev 2012, PMC3359723). The same research identified GHK as an HDAC inhibitor, meaning it works against enzymes that physically compress and silence gene expression as cells age. Think of HDACs as locks that get slapped onto parts of your DNA over time, turning off genes that used to be active. GHK-Cu's activity suggests it may be countering some of that silencing.
Members experience the skin texture and recovery changes most consistently in the 4–8 week window of a well-delivered GHK-Cu protocol.
Epithalon and the Science of How Many Times Your Cells Can Divide
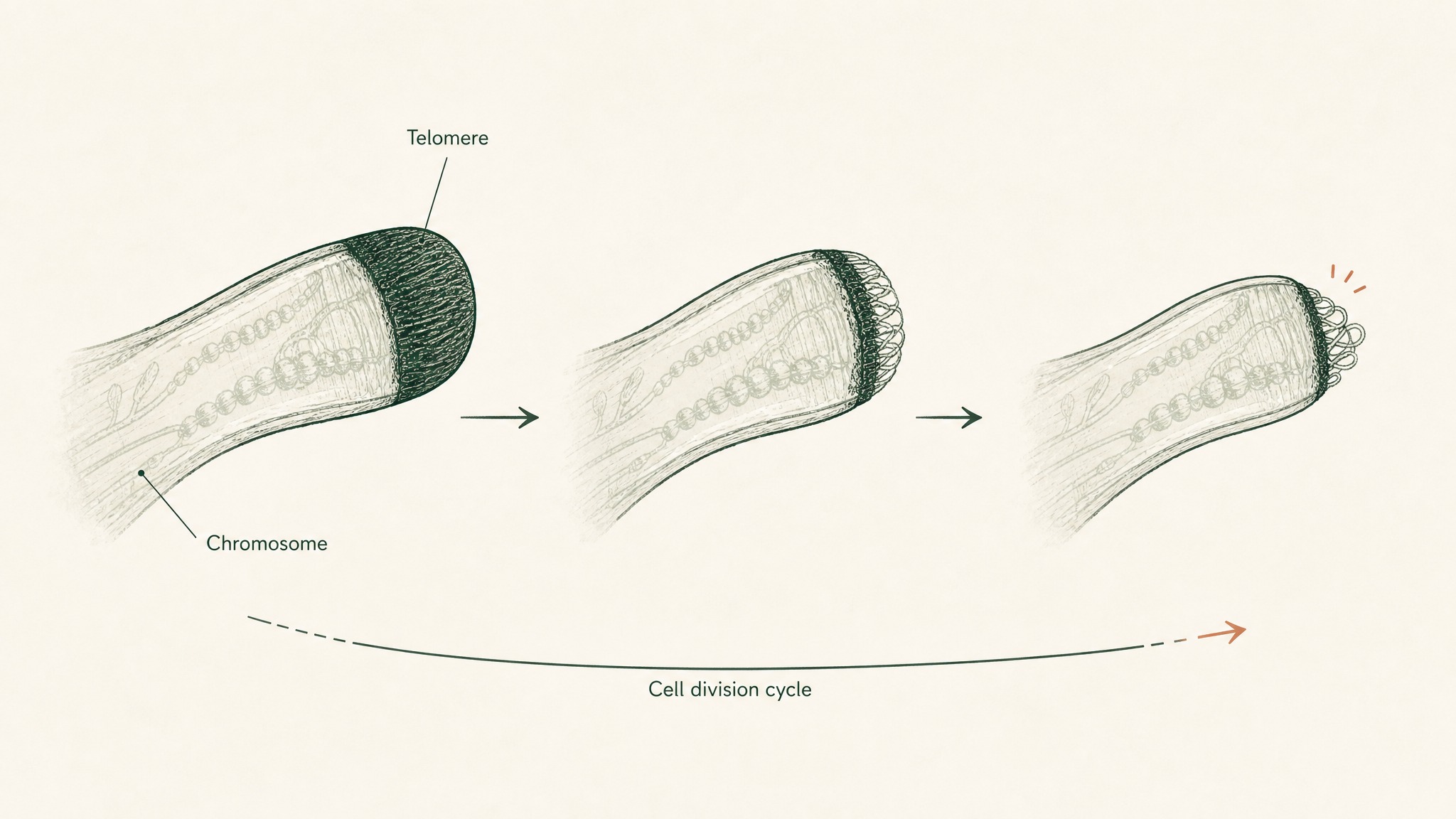
Picture the plastic tips at the ends of a shoelace. Without them, the lace frays and eventually falls apart. Telomeres do essentially the same job for your chromosomes: they're protective end-caps made of repeated DNA sequences, and they keep your genetic material from unravelling during cell division.
Here's the catch. Every time one of your cells divides to replace itself, the telomere gets a little shorter. When it gets short enough, the cell can no longer divide at all — it enters a dormant state called senescence. That limit is known as the Hayflick limit (named for the cell biologist who first observed it). It's one of the fundamental mechanisms through which biological ageing operates at the cellular level.
Epithalon is a synthetic tetrapeptide (four amino acids) originally developed at the St. Petersburg Institute of Bioregulation and Gerontology by Khavinson and colleagues. Its research centres specifically on telomerase (the enzyme your body uses to rebuild and maintain telomere length) and what happens in aging cells when that enzyme is reactivated.
A 2004 paper in the Bulletin of Experimental Biology and Medicine found that applying Epithalon to aging human fetal fibroblasts (connective tissue cells past their normal division limit) produced telomere elongation to lengths comparable to those seen in early-passage (that is, young) cells, enabling roughly 10 additional cell divisions beyond the cells' normal stopping point (Khavinson et al., Bull Exp Biol Med 2004, PMID 15455129). One cell model, in a controlled laboratory setting. But the mechanism it demonstrates, that telomere length can be altered in human cell models using a small synthetic peptide, is part of what makes Epithalon a recurring subject in cellular longevity research.
Epithalon's published literature is smaller than GHK-Cu's, and a significant portion of it originates from Russian-language sources and the same research group. That's worth knowing when you read the evidence base. What makes Epithalon distinctly relevant to age-supportive protocols isn't volume of data. It's non-redundancy. Its mechanism (telomerase activation in dividing cells) is entirely different from every other compound in this category. Pairing it with GHK-Cu doesn't give you the same biology twice. You're covering two entirely separate aspects of cellular ageing from two different biological directions.
MOTS-c: The Peptide Your Cells' Power Plants Generate
Most peptides your body produces originate from genes in your cell's nucleus, the central control room where most genetic information lives. MOTS-c is different in a way that matters.
It's encoded in your mitochondrial DNA, the separate genetic material inside your mitochondria. Think of mitochondria as the power plants of your cells: they convert the nutrients you eat into ATP, the chemical currency your body runs on. The fact that MOTS-c is encoded in the power plant's own instruction set, not in the central control room, tells you something about what it's doing. It's a message from the energy infrastructure to the rest of the body, adjusting how fuel is used based on the power plant's own status.
MOTS-c is 16 amino acids long. A landmark 2015 paper in Cell Metabolism found that MOTS-c administration in mouse models significantly reduced both age-dependent and diet-induced insulin resistance (the state where your cells stop responding properly to insulin, making it harder to regulate blood sugar and store energy correctly) and excess adiposity. The mechanism involved AMPK activation via the folate cycle and purine biosynthesis pathways (AMPK being a molecular master switch that tells your cells to shift into energy-conservation and metabolic repair mode) (Lee et al., Cell Metab 2015;21(3):443–454, PMID 25738459). The same paper documented that MOTS-c functions as what researchers described as a mitochondria-to-nucleus messenger: your cells' energy infrastructure directly signalling the genetic control room to adjust whole-body fuel metabolism.
What makes this relevant to the age-supportive category: the 2015 study specifically examined age-dependent insulin resistance, not just diet-induced. Metabolic flexibility (how well your body shifts between fuel sources, responds to insulin, and sustains energy production during exercise) changes measurably as you get older. MOTS-c is among the clearest examples of a mitochondria-derived compound being studied specifically in the context of those age-related changes.
Its mechanism is entirely distinct from peptides that influence metabolic outcomes through growth hormone secretagogue pathways. That's why it appears as a complement rather than a substitute in longevity-focused research stacks.
Users report that MOTS-c protocols produce the most noticeable changes in energy levels and metabolic response to exercise, though individual variation across protocols is significant.
What Happens When These Signals Decline Together
The reason age-supportive peptides matter as a category, not just as individual compounds, is the pattern they form across biology.
GHK-Cu falls roughly 60% in plasma concentration from your twenties to your sixties. The cellular mechanisms MOTS-c supports, AMPK activation and mitochondrial fuel signalling, show age-dependent changes in the same research landscape. Telomere length shortens predictably with each cell division, across decades.
These aren't isolated phenomena running in parallel. They're interconnected aspects of how your body's maintenance systems gradually slow their output. GHK-Cu coordinates gene expression across tissue remodelling, antioxidant defence, and structural maintenance. When those levels fall, the coordination slips across multiple domains at once. MOTS-c keeps the energy infrastructure responsive to insulin and exercise signals. Telomere shortening limits how many times your cells can divide to replace damaged tissue.
No single compound covers all of this. That's the honest framing for anything in this category. What the research is probing is whether targeted molecular support at specific points in these interconnected declining systems has measurable effects in cellular and animal models, and what the early human data suggests when it exists.
Research suggests the most productive approach treats these three mechanisms as complementary pieces, not competing options.
Delivery: Why This Category Needs More Than a Capsule
Here's the problem with oral peptide administration, and it applies to every compound in this article.
Your digestive system treats peptides like food. Enzymes in your stomach and gut break down protein chains (which is exactly what any peptide is) before they can be absorbed in meaningful quantities. Oral bioavailability for most short-chain peptides sits below 2%. Out of every 100 milligrams in a capsule, 98 or more get destroyed before reaching your bloodstream.
Research confirms this isn't a formulation quality issue — it's a route issue. A 2022 review in the Journal of Controlled Release examined the challenges of delivering peptides and other large biological molecules across mucosal surfaces and found that conventional oral delivery fails this entire compound class, and that newer formulation strategies, including penetration enhancers, mucoadhesive polymers (materials that stick to mucosal tissue and slow the compound's removal), and hybrid nanosystems, can substantially improve absorption compared to unformulated approaches (Rawas-Qalaji, Thu & Hussain, J Control Release 2022;352:726–746, PMID 36334858).
Injectable administration bypasses the degradation problem entirely — no gut, no liver first-pass metabolism, direct entry into circulation. That's why subcutaneous injection (an injection just under the skin) remains the research reference standard for most of these compounds. The tissue repair and gene modulation findings cited above were produced in protocols using injectable delivery or engineered carrier systems.
Sublingual delivery takes advantage of different anatomy. The tissue under your tongue is thin and richly supplied with blood vessels. A well-formulated sublingual system, with mucoadhesive polymers to extend contact time and permeation enhancers to move the compound across the membrane, can achieve meaningful systemic exposure without the enzymatic destruction of the gut route.
VERO's VERISORB sublingual technology was developed to address exactly the engineering requirements the 2022 Journal of Controlled Release review identified as the barrier for this compound class.
Building an Age-Supportive Research Protocol
The most consistently documented approach to age-supportive research protocols maps compounds to distinct, non-overlapping mechanisms and introduces them one at a time.
GHK-Cu and Epithalon are frequently paired in longevity-focused research structures, and the biological logic is clear: GHK-Cu acts on extracellular matrix composition (the structural scaffolding between and around your cells), antioxidant gene expression, and broad tissue remodelling signalling. Epithalon acts on telomerase activity in dividing cells. Neither mechanism overlaps the other. Together they address two separate aspects of cellular ageing from two completely different biological directions.
Adding MOTS-c to that foundation brings in the mitochondrial metabolic angle: energy signalling via AMPK, insulin sensitivity, and the metabolic response to physical stress. Three non-overlapping mechanisms covering three distinct biological domains, each with a documented connection to how your body changes as it ages.
The research community's consistent practice is sequential introduction: one compound first, establish your baseline response across several weeks, then introduce a second. Running all three simultaneously from day one makes it nearly impossible to attribute any observed change to a specific compound. Members experience significantly cleaner signal from sequential protocols, even when it takes longer to reach full stack depth.
A typical age-supportive research cycle runs 8–12 weeks on, followed by an off period before reassessment. That structure reflects both the observation windows used in the preclinical literature and the practical need for rest intervals in cyclical research protocols. These compounds are studied for research purposes only, and any protocol structure should be designed with that framing in mind.
Frequently Asked Questions
What's the difference between age-supportive peptides and anti-aging supplements?
Most anti-aging supplements target broad markers: antioxidants, vitamins, adaptogens. None of that targets a specific mechanism tied to measurable biological decline. Age-supportive peptides like GHK-Cu, Epithalon, and MOTS-c are studied because they decline measurably in the body with age and have documented activity in cellular processes that change with ageing. The research framing is narrower and more specific: researchers can point to a molecule, a mechanism, and a measurable change. These compounds are studied for research purposes only and are not supplements in the DSHEA sense.
Can GHK-Cu, Epithalon, and MOTS-c be used in the same research protocol?
Yes, and their non-overlapping mechanisms are the reason they're frequently paired. GHK-Cu works through gene expression modulation and extracellular matrix signalling. Epithalon works through telomerase activation. MOTS-c works through AMPK activation via the mitochondrial-nuclear signalling pathway. None of these mechanisms compete with the others. The standard research community practice is sequential introduction: one compound first, then add a second once you've established a baseline response, rather than starting all three simultaneously.
How do age-supportive peptides differ from NAD+ and sirtuins in longevity research?
NAD+ and sirtuins work through cellular energy metabolism and DNA repair regulation: the sirtuin pathway is activated by NAD+ and affects gene expression through deacetylation. Age-supportive peptides work through separate molecular routes: GHK-Cu inhibits HDACs (a different gene-silencing mechanism), Epithalon targets telomere length directly, and MOTS-c works upstream in the energy-regulation pathway via AMPK. Research suggests these systems are complementary rather than redundant. That's why they appear together in longevity-focused research stacks rather than being treated as alternatives.
Key Takeaways
- Age-supportive peptides are signalling molecules that decline measurably with age. GHK-Cu plasma levels drop roughly 60% from your twenties to your sixties (PMC6073405). That decline maps directly to the period when cellular repair and regeneration outputs start shifting.
- GHK-Cu modulates expression in over 31% of human genes simultaneously, covering tissue remodelling, antioxidant defence, and neurological maintenance in the same molecule (PMC6073405; PMC4180391). Skin collagen research found increases in 70% of subjects over one month, outperforming vitamin C cream and retinoic acid in the same conditions (PMC4508379).
- Epithalon research centres on telomere biology. A 2004 paper found it enabled approximately 10 additional cell divisions beyond the Hayflick limit in aging human fetal fibroblast models (PMID 15455129) — a non-redundant mechanism that no other compound in this category replicates.
- MOTS-c is encoded in mitochondrial DNA and functions as an energy-system messenger. The 2015 Cell Metabolism paper found it significantly reduced age-dependent insulin resistance in mouse models via AMPK activation (PMID 25738459).
- Delivery format is a prerequisite for this category. Oral bioavailability for short-chain peptides sits below 2%; injectable and properly formulated sublingual delivery are the routes with established research support (PMID 36334858). Delivery isn't a product detail — it's the variable that determines whether you're connected to the research outcomes or not.
- GHK-Cu, Epithalon, and MOTS-c have non-overlapping mechanisms — gene expression modulation, telomere biology, and mitochondrial metabolic signalling. That non-redundancy is what makes them complementary in a protocol rather than interchangeable.
Ready to explore a structured age-supportive research protocol? Start RESTORE →
This content is published for research and educational purposes only. It is not intended to diagnose, treat, cure, or prevent any disease, and it does not constitute medical advice. Readers should consult a qualified healthcare professional before making any decisions about their own health. Statements on this page have not been evaluated by the FDA. VERO's peptide products are supplied for research purposes only.
RESTORE™
BPC-157Engineered around 500mcg BPC-157. Accelerates deep tissue and joint recovery
Clinical Context
Important Notice: VERO protocols are nutritional and systemic optimisation formats. They are not intended to diagnose, treat, cure, or prevent any medical condition. These statements have not been evaluated by the Food and Drug Administration.

