What the published research actually shows about skin peptides — GHK-Cu collagen signalling, the topical delivery problem, hyaluronic acid and vitamin C as cofactors, and how RADIANCE addresses the full system.
Peptides for skin are one of those topics where the consumer market and the published research genuinely point in different directions. The research is real — fifty years of documented mechanisms, published outcomes, verified biology. The products in most skin-care aisles often deliver something quite different. This guide covers what the science actually shows, which compounds have the most evidence, and why delivery format changes the outcome entirely. This content is produced for research purposes only.
What Are Skin Peptides and How Do They Work?
Think of your skin like a building — a once-solid structure that quietly deteriorates as you age. Collagen is the steel frame. Elastin is the suspension system that lets the frame flex without cracking. Hyaluronic acid is the insulation filling the space between the two. And the cells that build and maintain all of it — fibroblasts — are the construction crew inside the walls.
Skin peptides are the work orders. Short chains of amino acids (typically two to fifty residues) that bind to receptors on fibroblasts and tell those cells what to build. They're not drugs in the classical pharmacological sense. They're signals. The cells decide what to do with them.
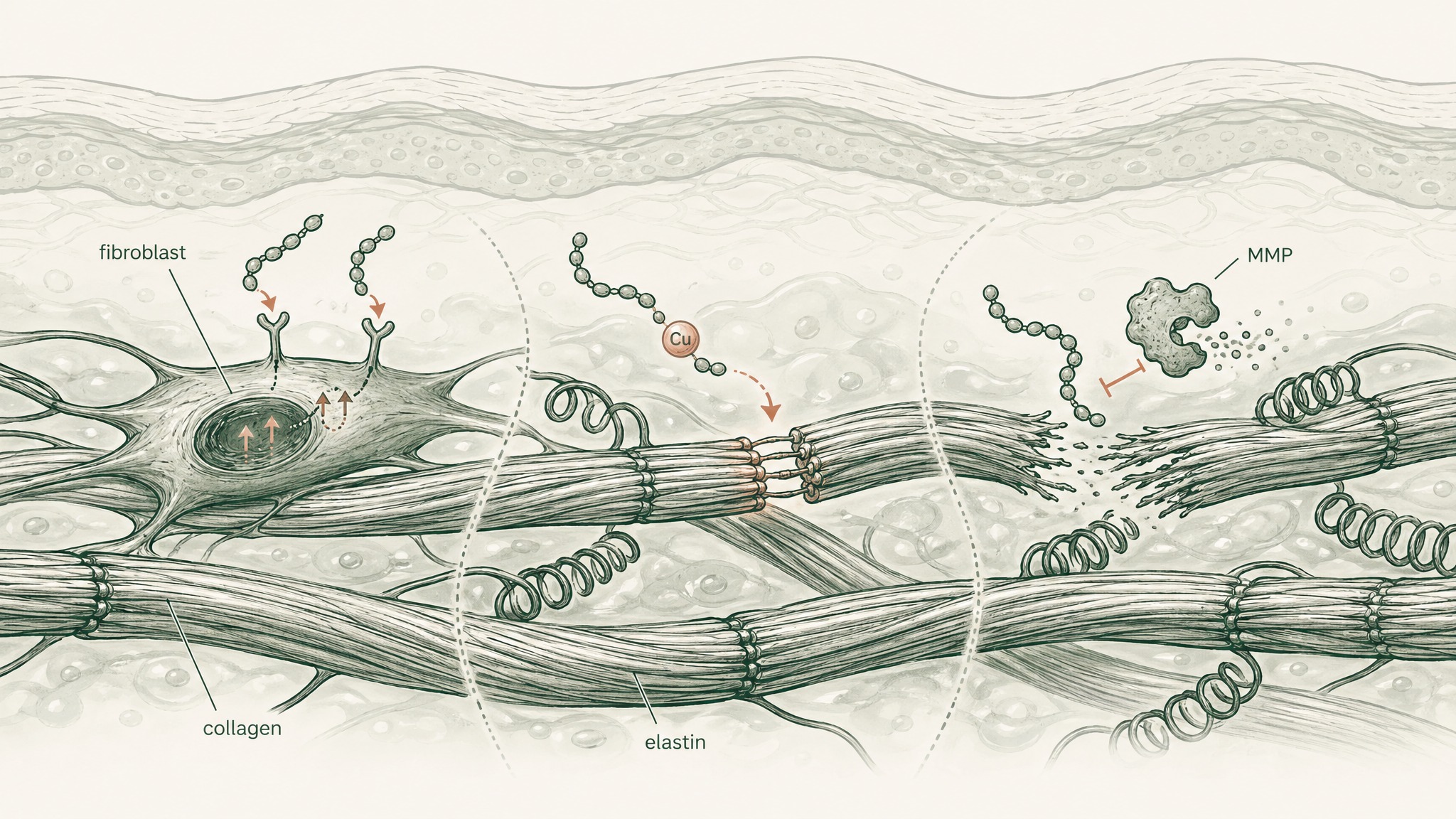
Researchers classify skin peptides in three functional categories:
Signal peptides bind to fibroblast receptors and trigger synthesis of structural proteins — collagen, elastin, and the proteoglycans (structural sugars that fill the matrix between fibres). GHK-Cu is the most extensively researched member of this class.
Carrier peptides deliver trace minerals that enzymatic processes in the dermis need to function. GHK-Cu works here simultaneously: the copper it carries is a cofactor for lysyl oxidase, the enzyme that performs the final cross-linking step as collagen fibres form. Without that copper, fibres don't properly lock together and the resulting structure is weaker.
Enzyme-inhibitor peptides slow the breakdown side of collagen turnover. Matrix metalloproteinases — MMPs, the enzymes that dissolve the collagen scaffold between synthesis cycles — are the target. Slowing degradation shifts the net balance in the same direction as accelerating synthesis, without touching the production machinery at all.
Most protocols focus on signal peptides because they address production directly. But understanding all three categories explains why GHK-Cu consistently outperforms more narrowly acting compounds — and why single-ingredient protocols often underdeliver even when the ingredient is correct.
GHK-Cu: Why It Has Five Decades of Research
GHK-Cu (glycyl-L-histidyl-L-lysine copper complex) is the compound with the deepest research base in skin biology. Unlike most ingredients in consumer skin products, it isn't synthetic. It's a molecule your body already makes.
Pickart and Thaler first isolated it from human serum and published the finding in Nature New Biology in 1973 (PMID 4349963). Your blood carries it throughout your life — just less of it as you get older.
Research suggests GHK-Cu plasma levels run at around 200 nanograms per millilitre in your twenties. By age 60, that's dropped to roughly 80 ng/mL — a 60% decline across four decades (Pickart & Margolina, Int J Mol Sci 2018, PMC6073405). The dermal changes that occur across the same period — thinner skin, slower wound healing, declining collagen density — may not be coincidental.
At concentrations as low as 1 to 10 nanomolar (an extremely small amount by pharmacological standards), GHK-Cu stimulated collagen production in fibroblast cultures, along with dermatan sulfate, chondroitin sulfate, and decorin — the structural proteoglycans that fill and support the ECM (extracellular matrix, the collagen-and-elastin scaffold of the dermis) (Pickart, Vasquez-Soltero & Margolina, Biomed Res Int 2015, PMC4508379). It also modulated both MMPs and their natural inhibitors TIMP-1 and TIMP-2, coordinating both sides of the turnover cycle rather than just pushing production higher.
A Connectivity Map genomic analysis found GHK-Cu changed expression by at least 50% in 32.1% of the 13,424 human genes across 22,277 probe sets examined (Pickart, Vasquez-Soltero & Margolina, Biomed Res Int 2014, PMC4180391). A 2018 update confirmed 31.2% of human genes modulated, covering repair, antioxidant defence, and anti-inflammatory signalling (PMC6073405). Antioxidant effects include documented superoxide dismutase support and a 75% reduction in lipid peroxidation in experimental tissue samples (Pickart, Vasquez-Soltero & Margolina, Oxid Med Cell Longev 2012, PMC3359723).
GHK-Cu isn't a single-target compound. It's a broad regulator of the biology your skin uses to build and maintain itself.
The Skin Doesn't Run on One Compound
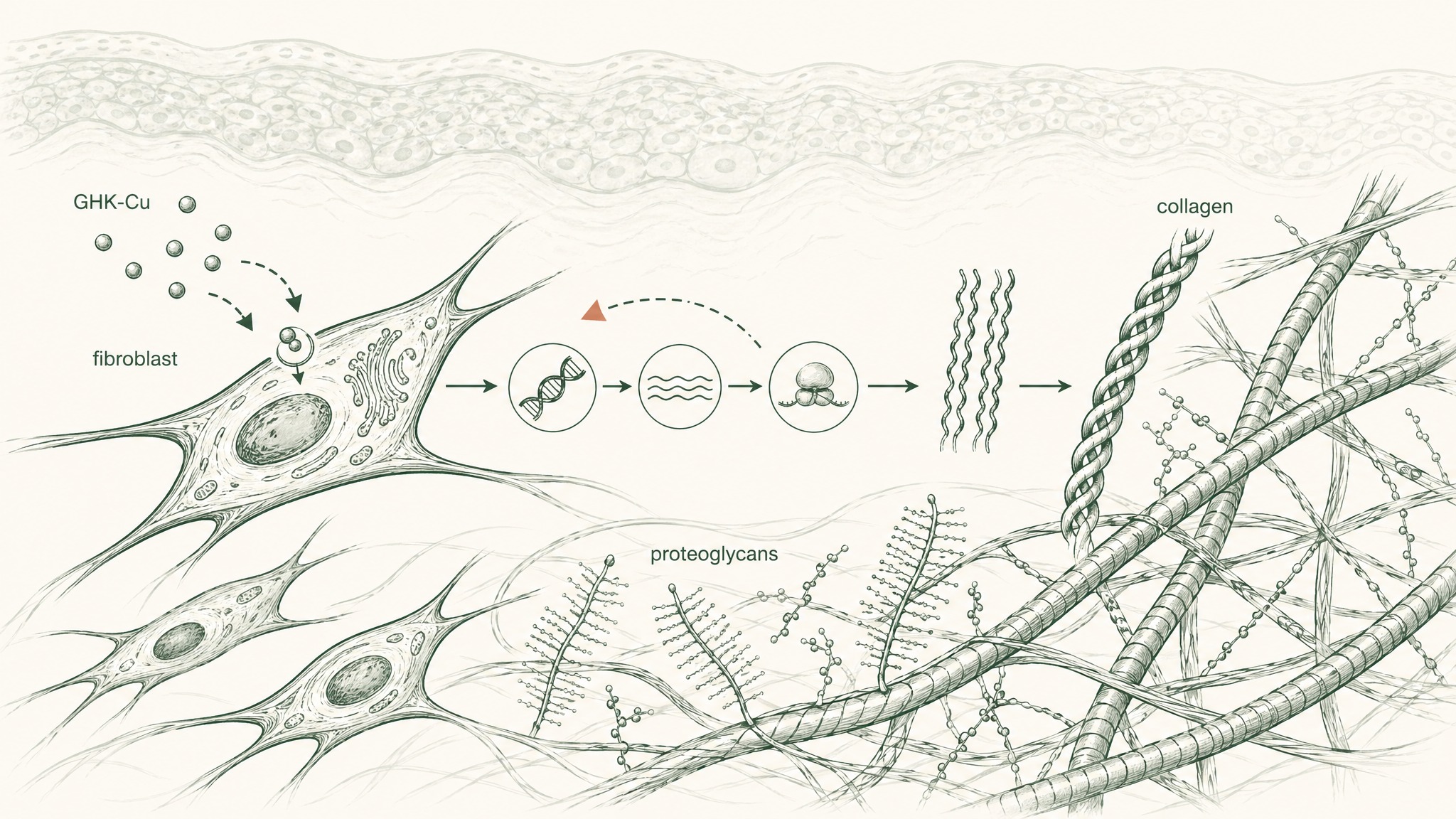
Here's where most GHK-Cu content stops short. The compound signals fibroblasts to synthesise collagen — but those cells still have to complete the synthesis. And they need other inputs to do so.
Think of it like a factory getting a production signal. The signal is necessary. But you also need raw materials, the right tools, and a functional work environment. Missing any one of those and the output falls short, regardless of how clear the signal is.
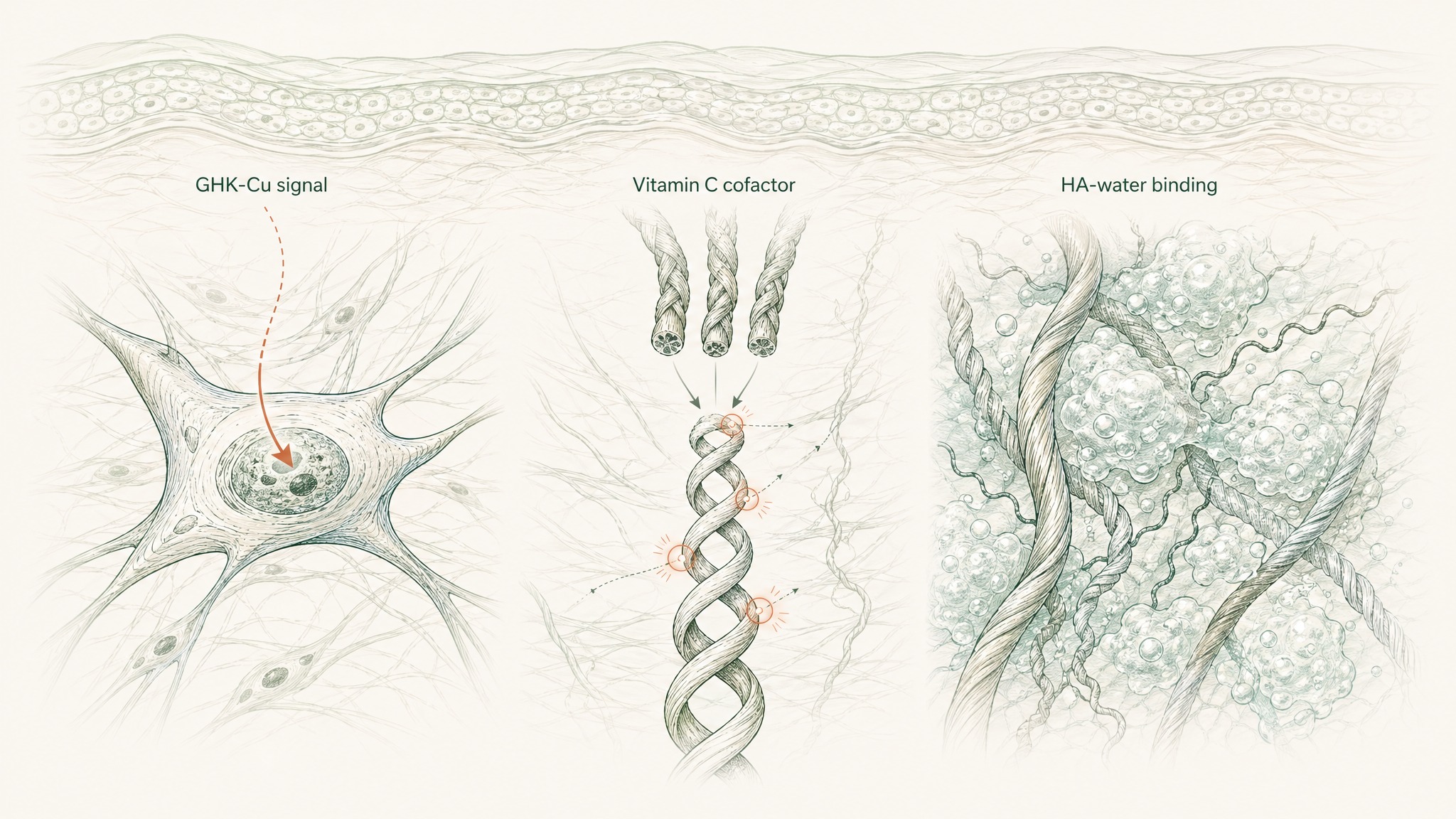
Hyaluronic acid (HA) is a glycosaminoglycan — a structural molecule that occupies the extracellular space between collagen and elastin fibres. It binds water at a remarkable rate, up to a thousand times its own molecular weight, and maintains the volumetric density that gives healthy skin its cushioned feel and visible plumpness. Research has documented measurable HA decline with age in the dermis, correlating directly with the flattening and volume loss visible in ageing skin (Papakonstantinou, Roth & Karakiulakis, Dermatoendocrinology 2012, PMC3583886). When the HA between collagen and elastin fibres depletes, the structural environment those fibres need to maintain their architecture starts to deteriorate.
Vitamin C (ascorbic acid) plays a different, non-negotiable role in the same system. Collagen biosynthesis requires hydroxylation of proline and lysine residues at two specific steps in the pathway. Without vitamin C as an enzymatic cofactor at both steps, procollagen chains can't complete the triple-helix structure that gives mature collagen fibres their tensile strength (Pullar, Carr & Vissers, Nutrients 2017, PMC5579659). GHK-Cu tells the fibroblast to produce collagen. Vitamin C is what the cell needs to actually complete that production.
VERO's RADIANCE Complex pairs GHK-Cu (5mg), hyaluronic acid (100mg), and vitamin C (500mg) because each one addresses a distinct layer: signalling input, structural hydration environment, and enzymatic synthesis capacity. Each targets something the others don't. That's the logic of the stack.
Why Topical Products Have a Hard Biological Ceiling
The vast majority of peptide-for-skin products are topical — serums, creams, and masks applied to the skin surface. Most of the GHK-Cu research that generates all that interest was conducted in cell cultures and in specially engineered delivery systems, not in serum jars on a bathroom shelf. That gap matters.
Your stratum corneum — the outermost skin layer, which functions like a tightly packed brick wall built from dead cells and lipids — is a serious biological barrier. The pharmaceutical rule of thumb for skin penetration is called the 500 Dalton rule: compounds above 500 Daltons in molecular weight generally can't cross intact skin in meaningful quantities (Bos & Meinardi, Exp Dermatol 2000, PMID 10839713). GHK-Cu at approximately 341 Daltons falls below that threshold — which sounds promising.
But that's only half the equation. GHK-Cu is hydrophilic — it's attracted to water and dissolves readily in it. The stratum corneum is lipophilic, built of lipids (fats). Water-soluble molecules partition poorly into fat-based barriers regardless of size. A GHK-Cu molecule applied to the skin surface encounters a fat wall standing between it and the fibroblasts in the dermis below. Most of it doesn't cross.
Users report noticing this distinction in practice: topical peptide products do produce localised effects at the epidermal level — surface texture, immediate hydration, barrier-layer changes. What they don't reproduce is the systemic exposure the gene modulation research, the plasma restoration rationale, and the broader ECM repair biology reflects. The dermis is a different biological target than the skin surface.
For endpoints that involve structural dermal change rather than surface-level hydration, the delivery route is the primary variable — not the compound concentration in the jar.
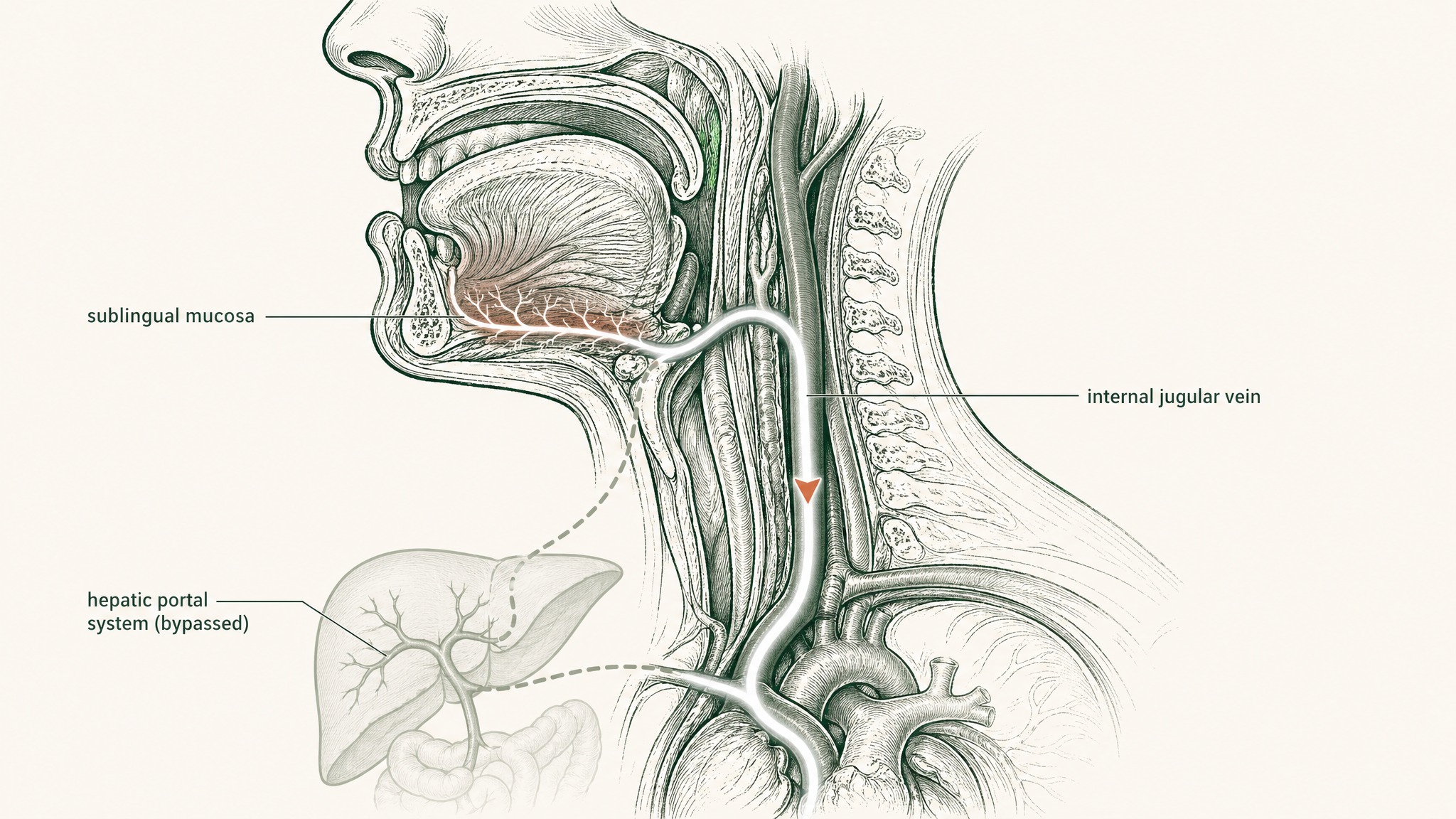
Sublingual Delivery and the VERISORB System
Sublingual delivery places the compound under your tongue, where mucosal membranes absorb it directly into venous drainage that bypasses the digestive system entirely. That drainage connects to the internal jugular and into systemic circulation without routing through the hepatic portal system. For a compound susceptible to both gut-enzyme degradation and first-pass liver metabolism, that bypass is a significant pharmacokinetic advantage.
The engineered carrier matters as much as the route. An unformulated drop of GHK-Cu placed under the tongue gets swallowed within seconds — before meaningful mucosal absorption can occur. Effective sublingual delivery of GHK-Cu requires mucoadhesive polymers to extend the contact window between compound and mucosal membrane, permeation-enhancing excipients to help a hydrophilic molecule cross into the submucosal vasculature, and protection from the salivary peptidases present even on the sublingual surface.
The published GHK-Cu literature consistently used engineered delivery systems to achieve its results — not unformulated free peptide. Wang et al. 2017 used liposome encapsulation in a scald wound model and showed a 33.1% increase in endothelial cell proliferation, upregulated VEGF and FGF-2, and wound closure at approximately 14 days post-injury (Wang et al., Wound Repair Regen 2017, PMID 28370978). Liu et al. 2023 demonstrated that an ionic liquid microemulsion improved GHK-Cu delivery approximately threefold versus unformulated peptide (Liu et al., Bioact Mater 2023, PMID 38026438). The delivery engineering is a core variable in the protocol, not optional packaging.
VERO's VERISORB system applies mucoadhesive and permeation-enhancer formulation principles specifically to GHK-Cu's pharmacokinetic profile. VERISORB is the mechanism by which RADIANCE achieves systemic compound exposure from a sublingual format without requiring an injectable — targeting the dermal and systemic endpoints the literature documents.
What to Observe and When
Structural changes in the dermis don't happen overnight. Your fibroblasts respond to GHK-Cu signalling at the cellular level within days, but the changes those cells produce — new collagen fibres, rebuilt proteoglycan matrix, increased ECM density — accumulate over weeks, not days.
Here's the general timeline the biology supports for a RADIANCE protocol:
Weeks 1–3. No visible structural change is normal in this window. GHK-Cu is establishing signalling input at the fibroblast level. Some members experience improved surface hydration during this phase, consistent with the hyaluronic acid component beginning to affect the extracellular environment.
Weeks 4–6. Early changes in skin firmness and texture may start becoming apparent, particularly for those tracking against a defined baseline — photographs, tactile assessment, hydration measurements. Research suggests this is when collagen synthesis rates are actively responding to the ongoing GHK-Cu signalling.
Weeks 8–12. The primary observational window for structural dermal change. Members experience the most consistent and quantifiable results during this phase. The GHK-Cu evidence base reflects protocols of at least 10 to 12 weeks for connective tissue and skin endpoints.
The off-period after an active cycle is as informative as the active phase itself. Changes that persist after stopping suggest actual structural ECM modification. Changes that fade suggest cellular response without lasting architectural change — and that distinction tells you exactly what a second cycle should target.
Key Takeaways
- Skin peptides are short amino acid chains that signal fibroblasts — the cells that build and maintain your skin's structural scaffold. Signal, carrier, and enzyme-inhibitor peptides work at different stages of the same collagen turnover cycle.
- GHK-Cu was first isolated from human serum in 1973 (PMID 4349963). Its plasma concentration declines roughly 60% between age 20 and 60 (PMC6073405).
- At 1–10 nM concentrations, GHK-Cu stimulates collagen, dermatan sulfate, chondroitin sulfate, and decorin, while co-ordinating MMP regulation on both sides of ECM turnover (PMC4508379).
- GHK-Cu modulates 31–32% of human genes by 50% or more across repair, antioxidant, and anti-inflammatory pathways (PMC4180391; PMC6073405).
- Topical peptide products are limited by the stratum corneum's lipid barrier and GHK-Cu's hydrophilicity. The dermal layer where fibroblasts live isn't accessible via most topical formulations (PMID 10839713).
- Engineered delivery systems are the key variable in achieving published GHK-Cu outcomes. Ionic liquid microemulsions improve delivery approximately threefold versus unformulated peptide (PMID 38026438).
- RADIANCE pairs GHK-Cu (collagen signalling), hyaluronic acid (structural hydration environment), and vitamin C (collagen synthesis cofactor) — three distinct layers of dermal support (PMC3583886; PMC5579659).
- Structural dermal changes accumulate over 8–12 weeks. The off-period determines whether results are structural (lasting) or transient — that distinction is the point of running a defined protocol.
Ready to build your skin research protocol? Explore the RADIANCE Complex →
This content is published for research and educational purposes only. It is not intended to diagnose, treat, cure, or prevent any disease, and it does not constitute medical advice. Consult a qualified healthcare professional before making any decisions about your own health. Statements on this page have not been evaluated by the FDA. VERO's peptide products are supplied for research purposes only.
References
Pickart L, Thaler MM. Tripeptide in human serum which prolongs survival of normal liver cells and stimulates growth in neoplastic liver. Nature New Biology. 1973;243(122):85–87. PMID 4349963
Pickart L, Margolina A. Regenerative and protective actions of the GHK-Cu peptide in the light of the new gene data. Int J Mol Sci. 2018;19(7):1987. PMC6073405
Pickart L, Vasquez-Soltero JM, Margolina A. GHK peptide as a natural modulator of multiple cellular pathways in skin regeneration. Biomed Res Int. 2015;2015:648108. PMC4508379
Pickart L, Vasquez-Soltero JM, Margolina A. The human tripeptide GHK-Cu in prevention of oxidative stress and degenerative conditions of aging: implications for cognitive health. Oxid Med Cell Longev. 2012;2012:324832. PMC3359723
Pickart L, Vasquez-Soltero JM, Margolina A. GHK and DNA: resetting the human genome to health. Biomed Res Int. 2014;2014:674168. PMC4180391
Wang X, Liu B, Xu Q, Sun H, Shi M, Wang D, Guo M, Yu J, Zhao C, Feng B. GHK-Cu-liposomes accelerate scald wound healing in mice by promoting cell proliferation and angiogenesis. Wound Repair Regen. 2017. PMID 28370978
Liu T, Liu Y, Zhao X, Zhang L, Wang W, Bai D, Liao Y, Wang Z, Wang M, Zhang J. Thermodynamically stable ionic liquid microemulsions pioneer pathways for topical delivery and peptide application. Bioact Mater. 2023. PMID 38026438
Papakonstantinou E, Roth M, Karakiulakis G. Hyaluronic acid: a key molecule in skin aging. Dermatoendocrinology. 2012;4(3):253–258. PMC3583886
Pullar JM, Carr AC, Vissers MCM. The roles of vitamin C in skin health. Nutrients. 2017;9(8):866. PMC5579659
Bos JD, Meinardi MM. The 500 Dalton rule for the skin penetration of chemical compounds and drugs. Exp Dermatol. 2000;9(3):165–169. PMID 10839713
RADIANCE™
Coming SoonEngineered around 5mg GHK-Cu. Increases dermal thickness and systemic collagen synthesis
Clinical Context
Important Notice: VERO protocols are nutritional and systemic optimisation formats. They are not intended to diagnose, treat, cure, or prevent any medical condition. These statements have not been evaluated by the Food and Drug Administration.

