GHK-Cu has decades of research behind it. The delivery format has almost none — and yet it's the decision that determines whether any of that research actually applies to your protocol.
Here's something most GHK-Cu content glosses over: the compound itself has decades of research. The delivery format you choose has almost none — and yet it's the variable that determines whether any of that research has any bearing on your protocol at all.
Think of GHK-Cu like a fresh strawberry. It has genuine biological value. But whether you actually get that value depends entirely on whether it survives the journey from the bottle to your bloodstream. Some delivery routes get it there intact. Others are essentially a blender set to liquify. The compound doesn't change. What changes is how much you absorb.
This content is published for research purposes only.
Why GHK-Cu Is Unusually Fragile
GHK-Cu is what's called a tripeptide — three amino acids connected in a chain. Think of it as a three-bead necklace. Your body actually makes this molecule naturally; it circulates in your blood throughout your life. Pickart and Thaler first pulled it from human serum in 1973 and published the finding in Nature New Biology (PMID 4349963).
The three-bead structure is the key detail here. It's linear, meaning the chain runs straight — no loops, no twists, nothing folded back on itself. Some peptides have cyclic structures (think of them as lasso-shaped) that are much tougher and harder to break down. Linear peptides don't have that protection. They're vulnerable to the enzymes your digestive system uses to cut proteins apart.
Research suggests plasma GHK levels sit at around 200 nanograms per millilitre when you're in your twenties. By the time you hit 60, that's declined to around 80 ng/mL — a 60% drop across four decades (Pickart & Margolina, Int J Mol Sci 2018, PMC6073405). The research rationale for GHK-Cu protocols starts right there: you're studying a signalling molecule your body produces less of as you get older.
What Happens When a Linear Peptide Hits Your Gut
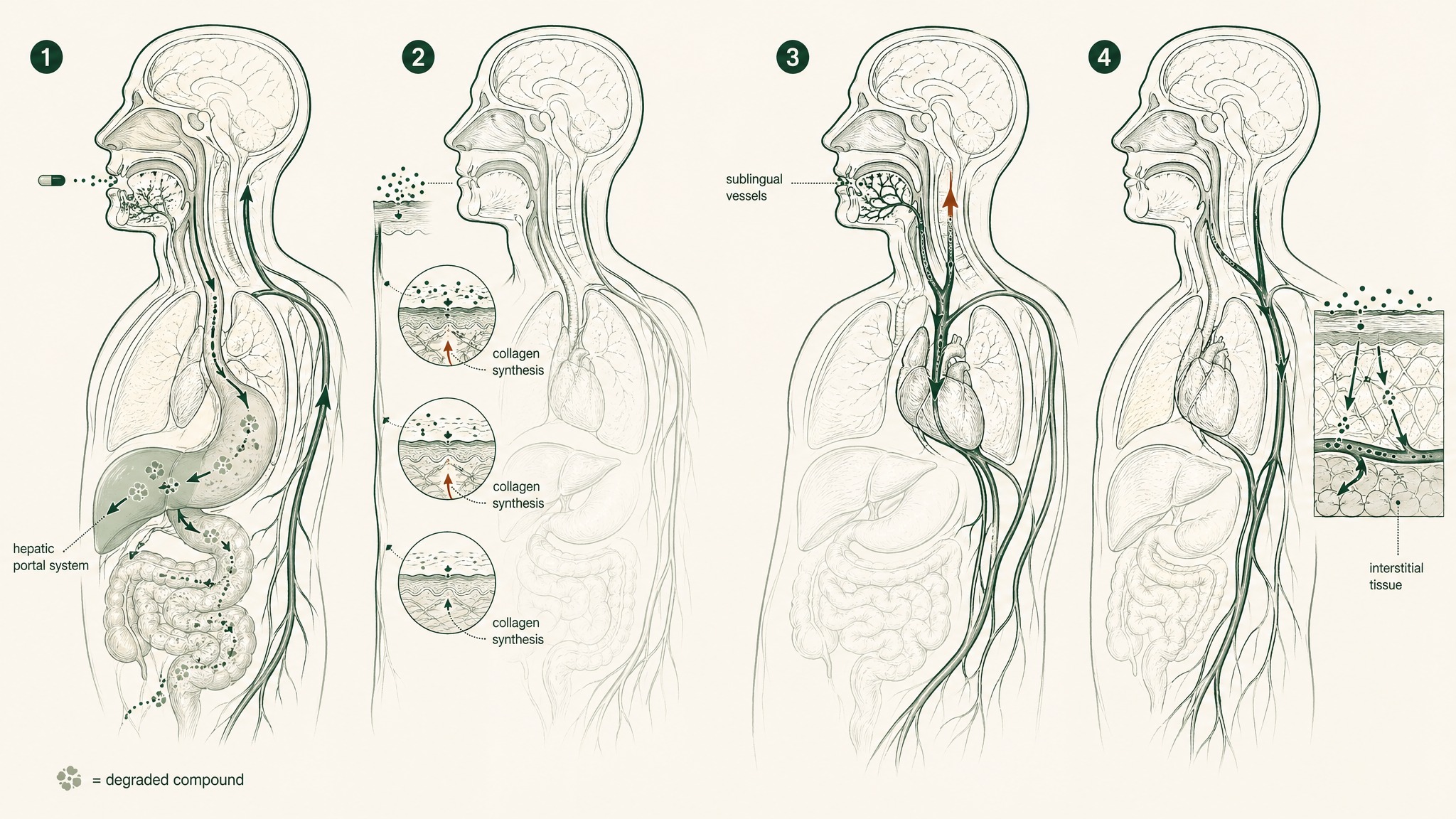
Your digestive system is essentially a security checkpoint with multiple layers of inspection — each one designed to break down what you swallow before it reaches your bloodstream.
First comes stomach acid, with a pH somewhere around 1 to 2. Then, as the partially digested material moves into your small intestine, brush-border peptidases (enzymes that line the gut wall and snip apart protein chains) take their turn. Anything that makes it past those two stages then gets routed through the hepatic portal system — meaning your liver gets first access before anything reaches your general circulation. That's "first-pass metabolism," the filter that processes a huge fraction of what you absorb before the rest of your body ever sees it.
For an unprotected linear tripeptide, this sequence is nearly terminal. Each checkpoint degrades the peptide a bit more. The cumulative result is negligible systemic bioavailability — meaning close to nothing makes it into your bloodstream intact where it could actually engage the biology you're researching.
Here's what matters: the biological effects documented in GHK-Cu research weren't observed using plain capsule administration. They were documented in cell culture, in nanoscale liposome delivery systems, and in ionic liquid microemulsion formulations. Engineered carrier systems. Delivery methods designed specifically to get the compound where it needed to go without losing it along the way.
Four Routes, Four Very Different Realities
Here's a plain-English breakdown of the four delivery routes people use for GHK-Cu, and what each one actually delivers biologically — not what sounds most appealing on a product page.
Swallowing a Capsule
Think of an unformulated GHK-Cu capsule like trying to mail a wet letter without an envelope. The message is real. But it's not going to survive the journey.
An unprotected oral capsule faces the full gastrointestinal sequence: stomach acid, intestinal enzyme activity, then hepatic first-pass processing before any systemic exposure is possible. For a linear tripeptide with no protective modifications, this produces very low systemic bioavailability. Most of what's in the capsule doesn't make it to your bloodstream.
You can engineer around this — enteric coating, nanoparticle encapsulation, and permeation-enhancing co-formulation can all change the pharmacokinetics (the way the compound moves through your body). But a standard capsule without those modifications doesn't connect to the published GHK-Cu biology in any meaningful way.
Applying It to Your Skin
Topical GHK-Cu actually has solid evidence — but specifically for localised skin effects, not for anything systemic.
Research published in 2015 found that skin creams containing GHK peptides increased collagen production in 70% of the women studied — outperforming vitamin C cream (50%) and retinoic acid (40%) in the same comparison group (Pickart, Vasquez-Soltero & Margolina, BioMed Res Int 2015, PMC4508379). At concentrations as low as 1 to 10 nanomolar — an extraordinarily small amount — GHK-Cu stimulated collagen synthesis and also modulated the enzymes responsible for regulated collagen turnover in fibroblast research.
Those skin effects are real. What they're not is systemic. Applying GHK-Cu to your skin surface doesn't produce the plasma concentrations linked to the broader gene modulation and antioxidant research. Topical is the right format for localised skin endpoints. It's the wrong format for anything requiring compound in your bloodstream.
Under Your Tongue
This is where the pharmacokinetics — how the compound moves through the body — get genuinely interesting.
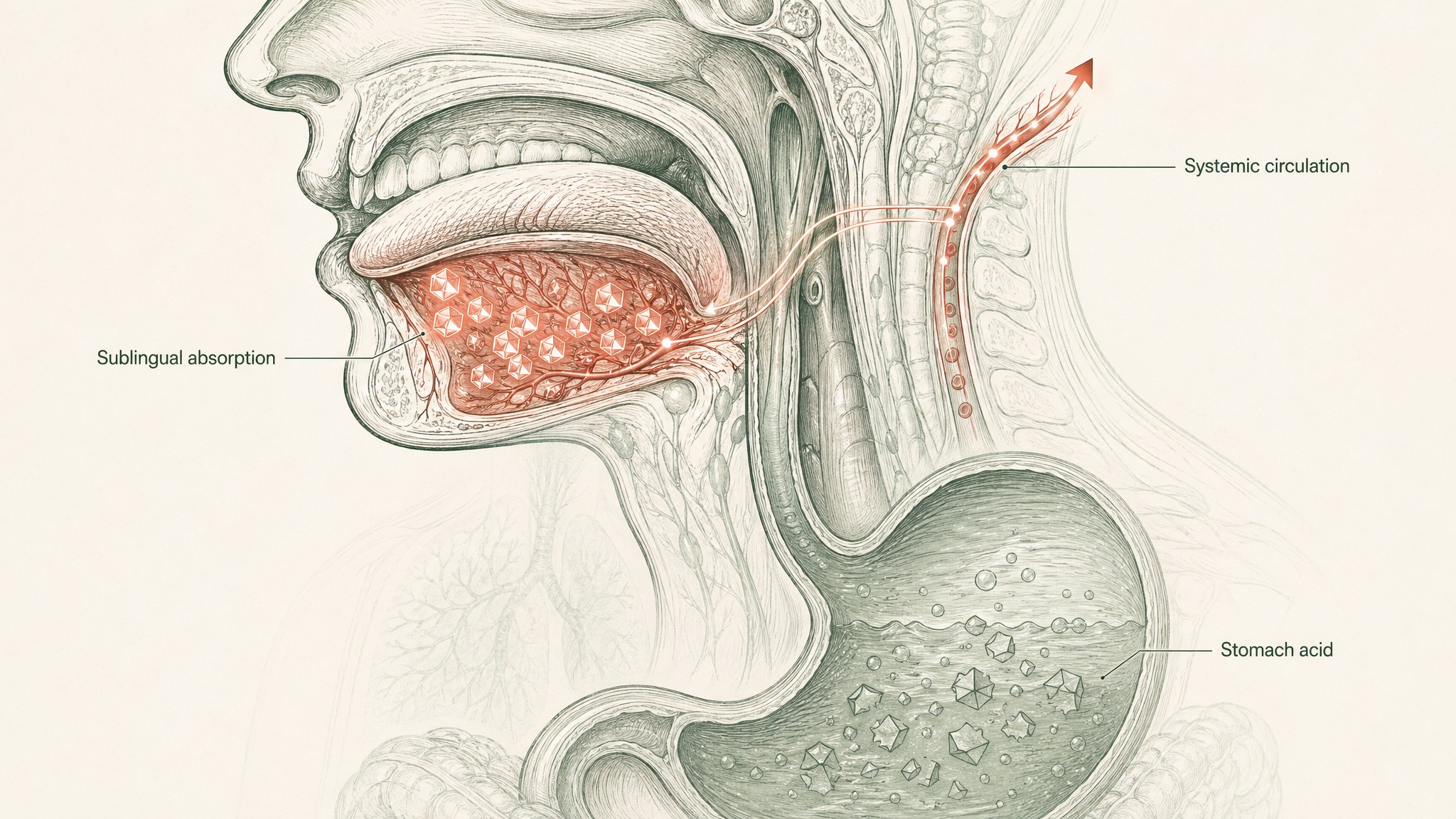
Sublingual absorption (holding something under your tongue rather than swallowing it) works by routing the compound through the dense network of blood vessels lining the floor of your mouth. Those vessels drain to the lingual veins, then to the internal jugular, and into systemic circulation — bypassing your gut and your liver entirely. The hepatic portal system, which normally routes anything absorbed from the gut through your liver before the rest of your body sees it, is completely sidestepped.
For a compound that gets degraded by both gut enzymes and hepatic processing, that's a meaningful advantage. You're removing the two largest degradation barriers in the oral absorption pathway.
The catch: holding something under your tongue doesn't automatically mean it absorbs. The sublingual membrane is permeable, but it isn't freely permeable to every compound — especially water-loving hydrophilic peptides like GHK-Cu. Without formulation engineering to extend contact time and facilitate membrane crossing, most of the compound gets swallowed before it has time to absorb.
Subcutaneous Injection
Subcutaneous injection (a small-gauge needle into the layer of tissue just beneath your skin) delivers compound directly into interstitial tissue, where it absorbs into capillaries and enters systemic circulation without passing through any digestive barriers at all. It's the most direct route to plasma-level exposure and the format used in most controlled GHK-Cu research settings.
For researchers who can manage the practical requirements — sterile reconstitution, refrigerated storage, injection technique and site rotation — it offers the most predictable path to systemic exposure. Members experience meaningful results this way, though it carries the highest practical burden of any format.
| Route | Systemic Exposure | Bypasses Liver | Injection? | Formulation Engineering Needed? |
|---|---|---|---|---|
| Oral capsule (plain) | Very low | No | No | Yes (to achieve any bioavailability) |
| Topical serum | Skin surface only | N/A | No | Partial |
| Sublingual (formulated) | High | Yes | No | Yes (critical) |
| Subcutaneous injection | High | Yes | Yes | No |
What's Actually Happening Under Your Tongue
Here's the physiology in plain terms.
The floor of your mouth is lined with a thin, highly vascular mucous membrane — more permeable than your skin, less barrier-heavy than your gut. Beneath it sits a dense bed of capillaries and small veins. When a compound crosses that membrane, it enters venous blood that drains to the internal jugular, into the superior vena cava, through the right side of your heart, across the pulmonary circulation, and out into systemic arterial flow.
Your liver never processes it at this stage. The problem the hepatic portal system creates for orally-administered peptides simply doesn't apply to sublingually-absorbed ones.
For GHK-Cu, this pharmacokinetic bypass removes the single most destructive step in the oral route: first-pass metabolism. A tripeptide that would lose a significant fraction of its intact mass passing through the liver arrives at your bloodstream without that processing step.
What it still has to do is cross the membrane. GHK-Cu is hydrophilic — water-attracted, which means it's less inclined to dissolve through the lipid-based mucosal barrier. That's the engineering problem.
Why "Sublingual" Is Only Half the Answer
Here's a distinction that gets lost in most product descriptions: the sublingual route and sublingual formulation engineering are two different things. The route gets you to the door. The engineering is what gets you through it.
Think of it like a secured building. Standing at the entrance is necessary. Having the right keycard is what actually opens it.
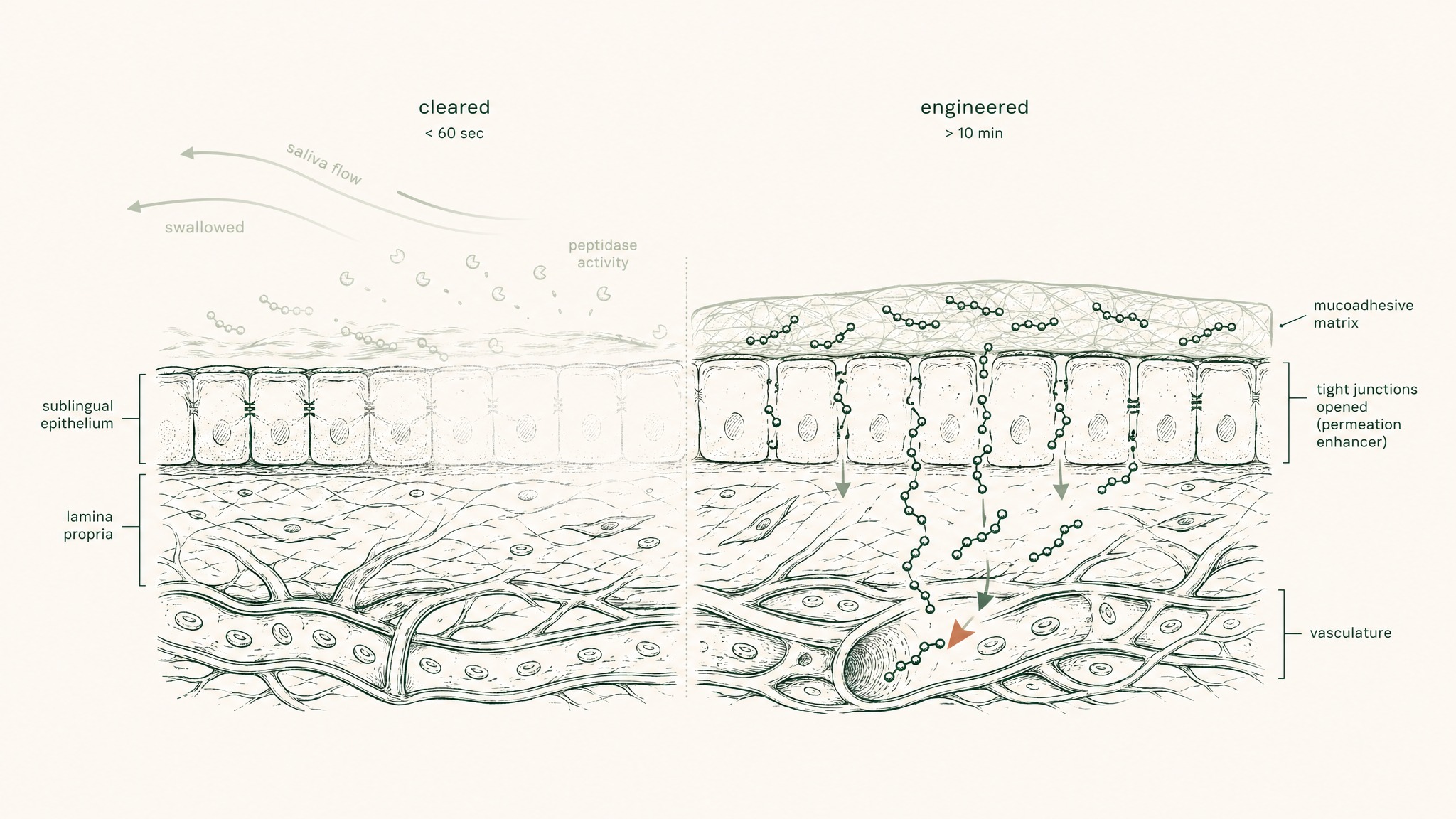
An unformulated sublingual drop — just GHK-Cu dissolved in liquid, placed under the tongue — faces a few specific problems:
Mucosal contact time. The average person swallows involuntarily every 30 to 60 seconds. An unformulated solution placed sublingually is mostly swallowed before meaningful absorption can occur. Mucoadhesive polymers — compounds that adhere to the mucosal surface and resist saliva-mediated clearance — extend that contact window from seconds to several minutes. That extended time is what creates the opportunity for absorption rather than swallowing.
Mucosal permeability. Adhesion extends contact. Permeation enhancers — excipients that transiently modify the mucous membrane to improve compound transit — actually facilitate the crossing. These are the keycard. Without them, GHK-Cu sits against the membrane surface but doesn't cross into the submucosal vasculature where absorption into venous drainage actually happens.
Salivary peptidase activity. Your saliva contains proteolytic enzymes — the same type that begin digesting proteins in your mouth when you eat. They start working on GHK-Cu on contact. A formulation that doesn't protect the peptide during the contact period allows degradation to compete with absorption throughout the entire absorption window. The compound can be in contact with the membrane and being destroyed at the same time. pH modification and matrix encapsulation strategies reduce that competitive degradation.
Users report that a well-engineered sublingual tablet feels noticeably different to an unformulated sublingual drop. The engineering isn't visible in the product format. But it's the variable that determines what actually happens at the mucosal level.
What the Leading Delivery Studies Chose
Research suggests the most instructive data point is what the researchers selected when they needed GHK-Cu to actually produce a measurable result.
Wang et al. (2017) didn't dissolve GHK-Cu in saline. They encapsulated it in nanoscale liposomes — spherical lipid carriers each smaller than a bacterium — specifically to protect the peptide in transit and enhance delivery to target tissue. Their mouse scald wound model recorded a 33.1% increased rate of human umbilical vein endothelial cell proliferation compared to controls. In the animal model, wound closure was shortened to approximately 14 days post-injury, with documented upregulation of VEGF and FGF-2 expression (Wang et al., Wound Repair Regen 2017, PMID 28370978). The liposomal delivery system was the variable that made the peptide's biology accessible.
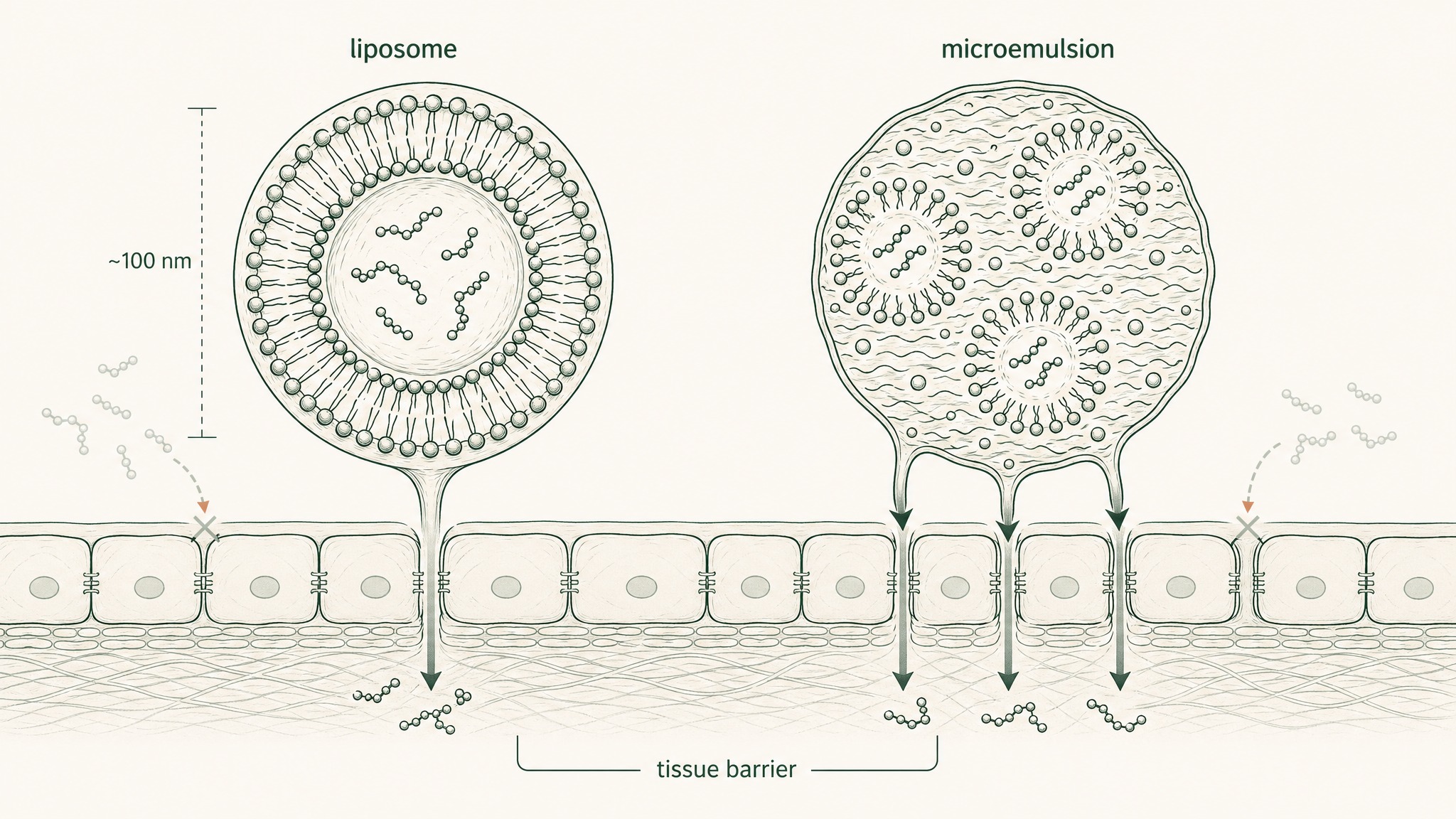
Liu et al. (2023) used thermodynamically stable ionic liquid microemulsions for hair follicle research. The engineered system achieved approximately threefold improvement in local peptide delivery compared to non-formulated controls, and activated the Wnt/beta-catenin signalling pathway — a key regulator of follicle cycling — in mouse models, with documented increases in VEGF and HGF expression (Liu et al., Bioactive Materials 2023, PMID 38026438).
Neither study used free peptide placed directly on cells or tissue. Both teams treated the delivery system as the critical variable that connected GHK-Cu's documented biology to a real-world outcome. Members experience the same principle in product selection: the carrier system is a protocol variable, not just packaging.
Why Getting This Right Is Worth the Effort
Here's the scope of what's at stake. Research suggests GHK modulates expression by at least 50% in 31.2% of human genes, covering ECM repair, antioxidant defence, anti-inflammatory signalling, and neurological maintenance pathways (Pickart & Margolina, Int J Mol Sci 2018, PMC6073405). A 2014 genomic analysis confirmed GHK changed expression by at least 50% in 32.1% of the 13,424 genes represented across 22,277 probe sets (Pickart et al., BioMed Res Int 2014, PMC4180391).
A 2012 review also documented GHK-Cu's role in antioxidant defence — specifically a 75% reduction in lipid peroxidation (cellular fat damage caused by oxidative stress) in gastric tissue samples, along with suppression of pro-inflammatory signalling proteins TGF-beta and TNF-alpha in human fibroblast culture, and identification of GHK-Cu as a strong HDAC inhibitor with potential relevance to cognitive and neurological research (Pickart et al., Oxid Med Cell Longev 2012, PMC3359723).
That's not a narrow-target compound. GHK-Cu sits at the intersection of tissue remodelling, oxidative stress, and neurological function simultaneously. It's one of the most broadly-researched peptides in longevity science.
But all of that documented activity is predicated on the compound reaching systemic circulation intact. GHK-Cu destroyed by stomach acid doesn't engage any of those pathways. The delivery format isn't a marketing choice. It's the variable that determines whether the research is relevant to your protocol at all.
RADIANCE and VERISORB: The Formulation Approach
RADIANCE is VERO's GHK-Cu research protocol, delivered via VERISORB — VERO's proprietary sublingual delivery platform. VERISORB applies mucoadhesive polymer and permeation-enhancer formulation engineering to the specific challenge of getting a hydrophilic linear tripeptide across the sublingual mucosa and into systemic circulation without injection.
The design directly addresses the three formulation problems above: extended mucosal contact time via polymer adhesion, facilitated mucosal crossing via permeation-enhancing excipients, and peptide protection during the contact window to reduce competitive salivary peptidase degradation. It isn't a plain sublingual drop with GHK-Cu dissolved in it. The formulation engineering is the product.
RADIANCE is designed for researchers whose protocol endpoint requires systemic GHK-Cu exposure across the ECM remodelling, antioxidant, anti-inflammatory, and longevity pathway research documented in the literature. It's not a topical product and not designed for localised dermal delivery. RADIANCE has not been evaluated by the FDA for any indication. Protocol decisions should be made in consultation with a qualified healthcare professional.
Key Takeaways
- GHK-Cu is a naturally occurring linear tripeptide first identified in human serum in 1973 (PMID 4349963). Its linear structure makes it vulnerable to gut enzyme sequences and hepatic first-pass metabolism.
- Research suggests plasma GHK declines roughly 60% between your twenties and sixties — from ~200 ng/mL to ~80 ng/mL (PMC6073405). That measurable decline in an endogenous signalling peptide is the basis for studying supplemental GHK-Cu.
- Topical GHK-Cu has documented skin effects: increased collagen production in 70% of women studied vs. 50% with vitamin C cream (PMC4508379). It doesn't produce systemic exposure.
- Sublingual delivery bypasses the hepatic portal system via lingual venous drainage — removing first-pass metabolism from the equation. But the route alone isn't enough: mucoadhesive polymers extend contact time and permeation enhancers facilitate membrane crossing. Both are required.
- Both leading GHK-Cu delivery studies used engineered carrier systems: liposomes in Wang 2017 (PMID 28370978) and ionic liquid microemulsions in Liu 2023 (PMID 38026438). Research suggests the carrier system is the protocol variable, not just the compound.
- GHK modulates expression by at least 50% in 31.2% of human genes (PMC6073405; PMC4180391). A 2012 review also confirmed GHK-Cu's role in antioxidant defence and anti-inflammatory signalling (PMC3359723). Getting the delivery format right is what determines whether any of that activity is accessible.
- RADIANCE via VERISORB is VERO's structured GHK-Cu sublingual research protocol. For research purposes only.
Ready to explore a structured GHK-Cu sublingual protocol? Explore RADIANCE →
This content is published for research and educational purposes only. It is not intended to diagnose, treat, cure, or prevent any disease, and it does not constitute medical advice. Readers should consult a qualified healthcare professional before making any decisions about their own health. Statements on this page have not been evaluated by the FDA. VERO's peptide products are supplied for research purposes only.
RADIANCE™
Coming SoonEngineered around 5mg GHK-Cu. Increases dermal thickness and systemic collagen synthesis
Clinical Context
Important Notice: VERO protocols are nutritional and systemic optimisation formats. They are not intended to diagnose, treat, cure, or prevent any medical condition. These statements have not been evaluated by the Food and Drug Administration.

