PT-141 is a synthetic peptide that targets the brain's own desire circuitry — not hormone levels in the bloodstream. But like most peptides, what you take and how you take it are two completely different questions.
Most conversations about low desire blame hormones. And hormones play a role. But they're not where the story ends.
There's a small region of your brain — the medial preoptic area of the hypothalamus, roughly the size of an almond — where desire actually gets switched on. The neurotransmitter that flips that switch is dopamine, the brain's motivation-and-drive chemical. Sitting in that region is a receptor called MC4R. When it's activated, dopamine flows. When it isn't, the signal stays quiet.
PT-141 is a synthetic peptide designed to activate MC4R. This content is published for research purposes only.
What PT-141 Actually Is
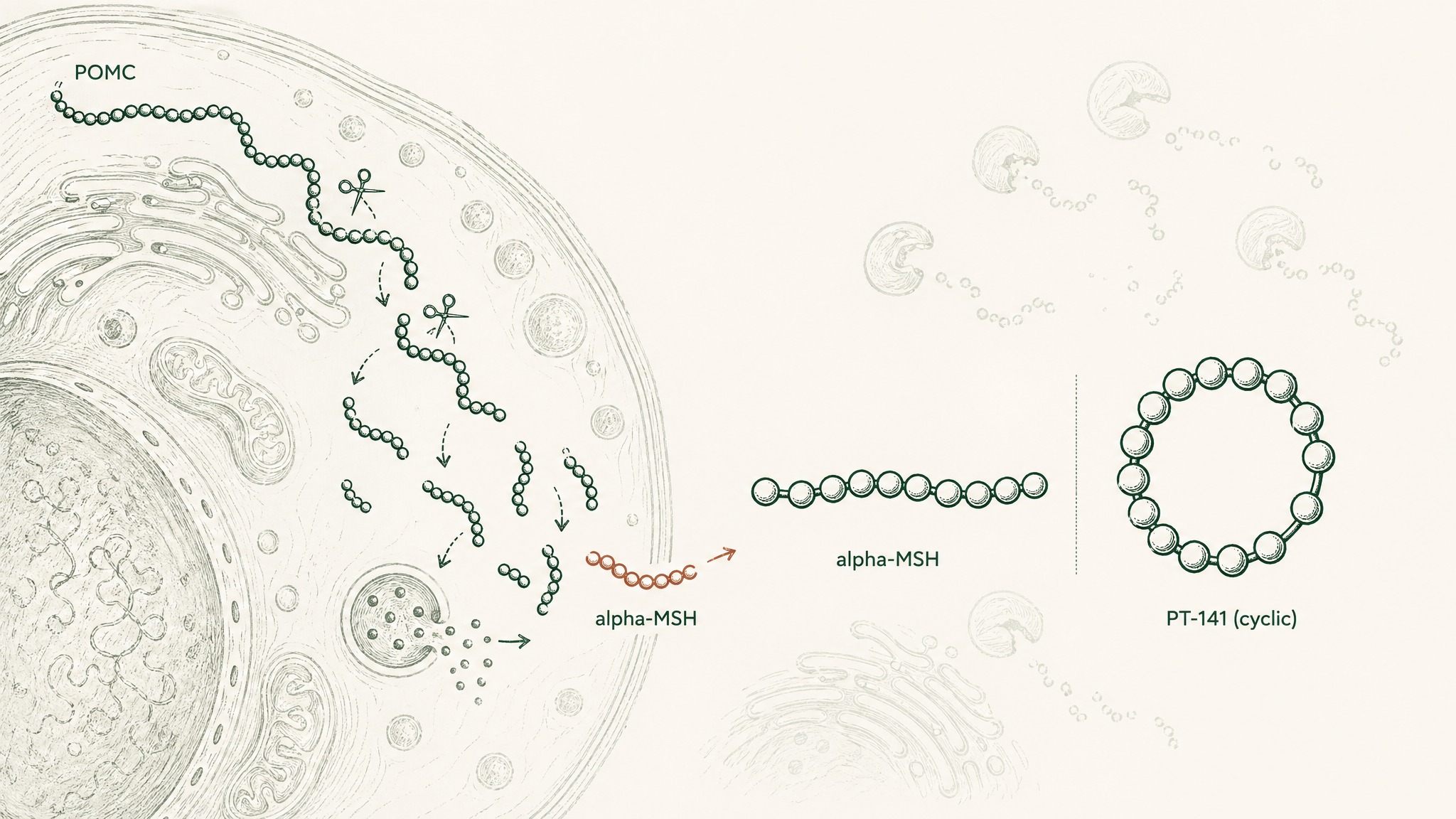
Your brain produces a natural signalling molecule called alpha-MSH — alpha-melanocyte-stimulating hormone. It's made by cleaving a larger protein called POMC (proopiomelanocortin), think of POMC as a molecular chain your cells cut into pieces, with each piece having its own specific job. Alpha-MSH is one of those pieces, and it acts across the melanocortin receptor system — a network of five different receptor types, each found in different tissues, each doing different things when activated (Gantz & Fong, Am J Physiol 2003, PMID 12556347).
PT-141 is a synthetic, cyclic analog of alpha-MSH. Cyclic means the molecule's ends are joined to form a ring structure, making it more resistant to the enzymes that would normally break it down. It was engineered to be more stable and to have a longer-lasting effect than the natural hormone. In 2019, the FDA granted approval for bremelanotide (the generic name) under the brand name Vyleesi, for use in premenopausal women with acquired, generalized hypoactive sexual desire disorder (HSDD) — persistently reduced sexual desire that causes real personal distress (Dhillon & Keam, Drugs 2019, PMID 31429064).
PT-141 was the second FDA-approved pharmaceutical for HSDD, and the only one that works by targeting the central nervous system rather than hormones in the blood.
The Brain Chemistry Behind Desire
Picture the melanocortin receptor system as a network of five different locks scattered throughout your body — MC1R through MC5R. MC1R sits in your skin cells and governs pigmentation. MC3R and MC4R are found primarily in your brain. And MC4R, specifically, is concentrated in the medial preoptic area (mPOA) — the part of your hypothalamus most involved in regulating sexual behaviour.
When PT-141 binds to MC4R receptors in the mPOA, it activates presynaptic neurons that release dopamine — the brain's motivation and desire chemical, the same one that makes food taste rewarding and goals feel worth pursuing. This is the mechanism: not hormones in the bloodstream, but a direct signal at the receptor layer inside your brain (Pfaus, Sadiq, Spana & Clayton, CNS Spectr 2022, PMID 33455598).
This is what separates PT-141 from hormone-based approaches. Testosterone and oestrogen therapies operate at the level of the bloodstream and influence many different systems at once. PT-141 works upstream, at the receptor level in the CNS (central nervous system — your brain and spinal cord). Research suggests approximately 10% of women in the United States experience HSDD (PMID 33455598).
What the Phase 3 Trials Found
The pivotal research came from two large, parallel Phase 3 trials called RECONNECT, published in Obstetrics & Gynecology in 2019. Combined, they enrolled 1,267 women with diagnosed HSDD. The studies used two validated measurement tools: the Female Sexual Function Index desire domain (FSFI-D), which scores the level of sexual desire, and the Female Sexual Distress Scale (FSDS-DAO), which measures how much the lack of desire is causing personal distress.
Both endpoints reached statistical significance. Across the integrated trials, desire scores improved by +0.35 on the FSFI scale (p<0.001) and distress scores improved by -0.33 on the FSDS scale (p<0.001) versus placebo (Kingsberg, Clayton, Portman et al., Obstet Gynecol 2019, PMID 31599840).
The effect is on-demand, not daily. Bremelanotide is dosed roughly 45 minutes before sexual activity, and the maximum frequency studied was 8 doses per month. Members experience the timing flexibility as one of the more practical features of this research compound compared to daily-dose options.
Long-term data came from a 52-week open-label extension following the core trial. Of the 684 women who continued into the extension, FSFI desire scores increased by 1.25–1.30 points from baseline by end of year, with no new safety signals emerging across the full year of use (Simon, Kingsberg, Portman et al., Obstet Gynecol 2019, PMID 31599847).
That's meaningful. Sustained effect over 12 months without the adverse event picture worsening — both are important signals when evaluating a research compound's longer-term profile.
Why Delivery Format Is the Deciding Variable
Here's the part most content about PT-141 glosses over: the clinical trial data above was generated using subcutaneous injection — a small-gauge needle into the tissue just beneath the skin of the abdomen or thigh. That route delivers bremelanotide directly into interstitial tissue, where it absorbs into capillaries and enters systemic circulation without passing through any digestive barriers at all.
What happens when you try to deliver the same compound orally instead? Think of your gut as a dissection lab with one assignment: break down proteins and peptides into their smallest building blocks before letting anything into your bloodstream. A melanocortin peptide like PT-141 faces stomach acid first, then intestinal enzyme activity, then hepatic first-pass metabolism — the step where your liver processes everything absorbed from the gut before the rest of your body sees it. By the time a peptide has navigated all three stages, the amount reaching your bloodstream intact is negligible.
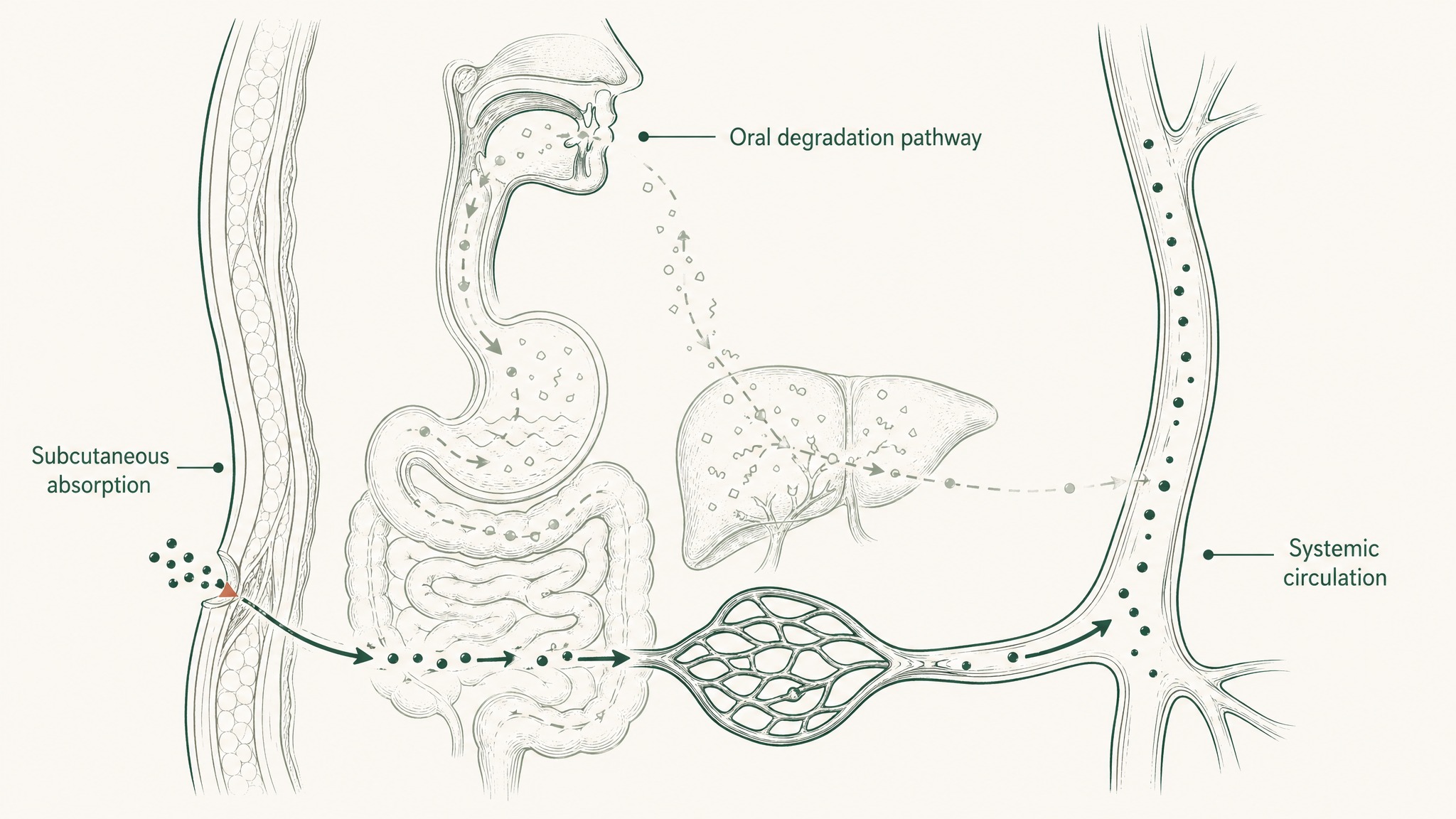
Research on oral bremelanotide in beagle dogs found exactly what you'd expect: minimal oral absorption (Sauter, Uhl, Burhenne & Haefeli, J Pharm Biomed Anal 2020, PMID 32353679). Separate research on Melanotan-II — a related cyclic melanocortin peptide — documented oral bioavailability of just 4.6% in rats (Lan, Ugwu, Blanchard et al., J Pharm Sci 1994, PMID 7983590).
You can't meaningfully study what you can't absorb.
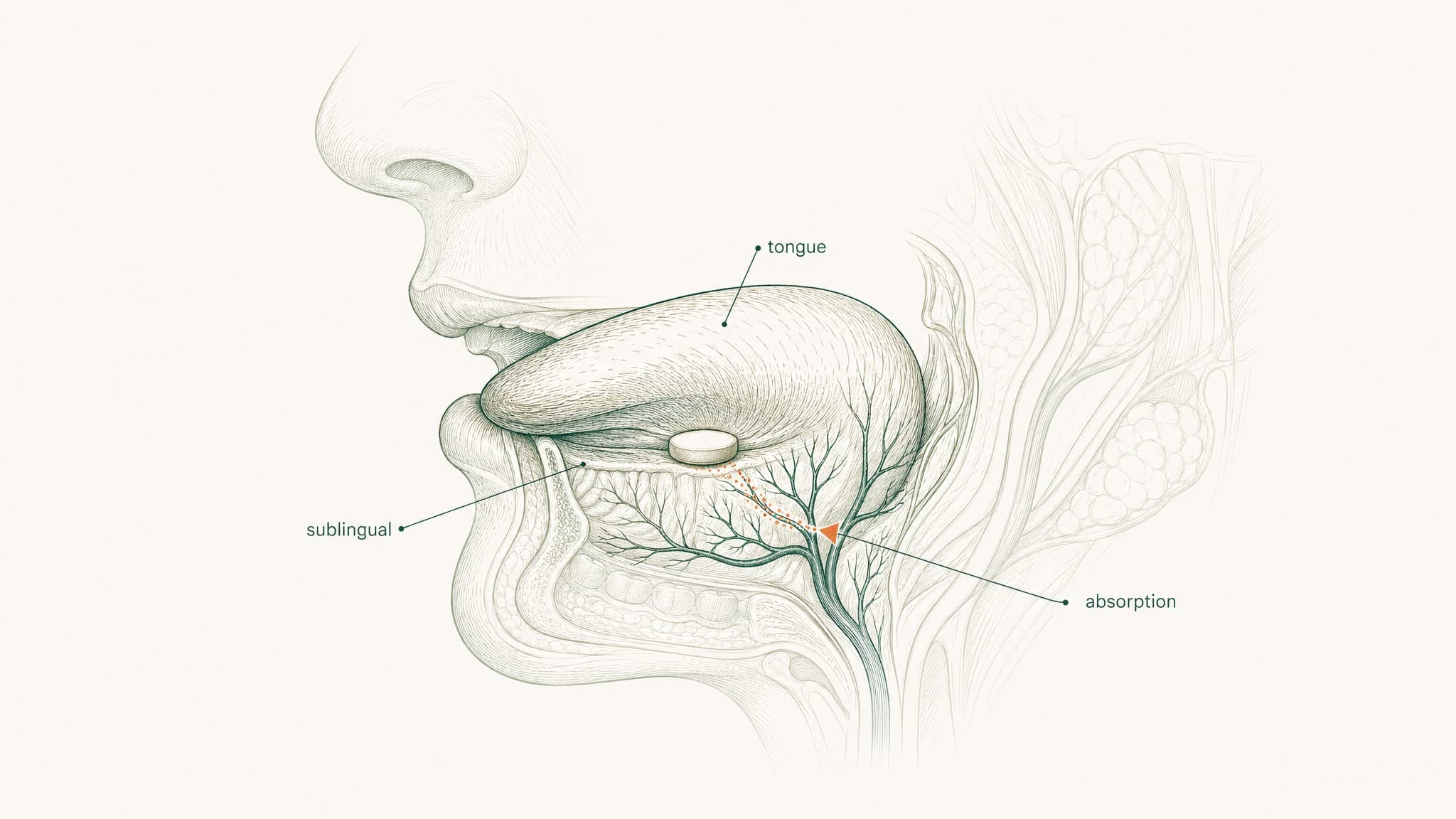
Where Sublingual Delivery Fits In
There are two ends of the spectrum: subcutaneous injection (high bioavailability, requires needles and sterile preparation) and swallowing a capsule (near-zero bioavailability, no practical burden but no useful outcome). Sublingual delivery — absorbing through the tissue under your tongue rather than swallowing — is the middle path, but only if the formulation is right.
Here's why the route is interesting. The floor of your mouth is lined with a highly vascular mucous membrane. It's more permeable than your skin, and it doesn't have the enzymatic barrier-loading of your gut wall. Beneath that membrane sits a dense network of capillaries and small veins. When a compound crosses that membrane, it drains to the lingual veins, then the internal jugular, and into systemic circulation — bypassing your gut and your liver entirely.
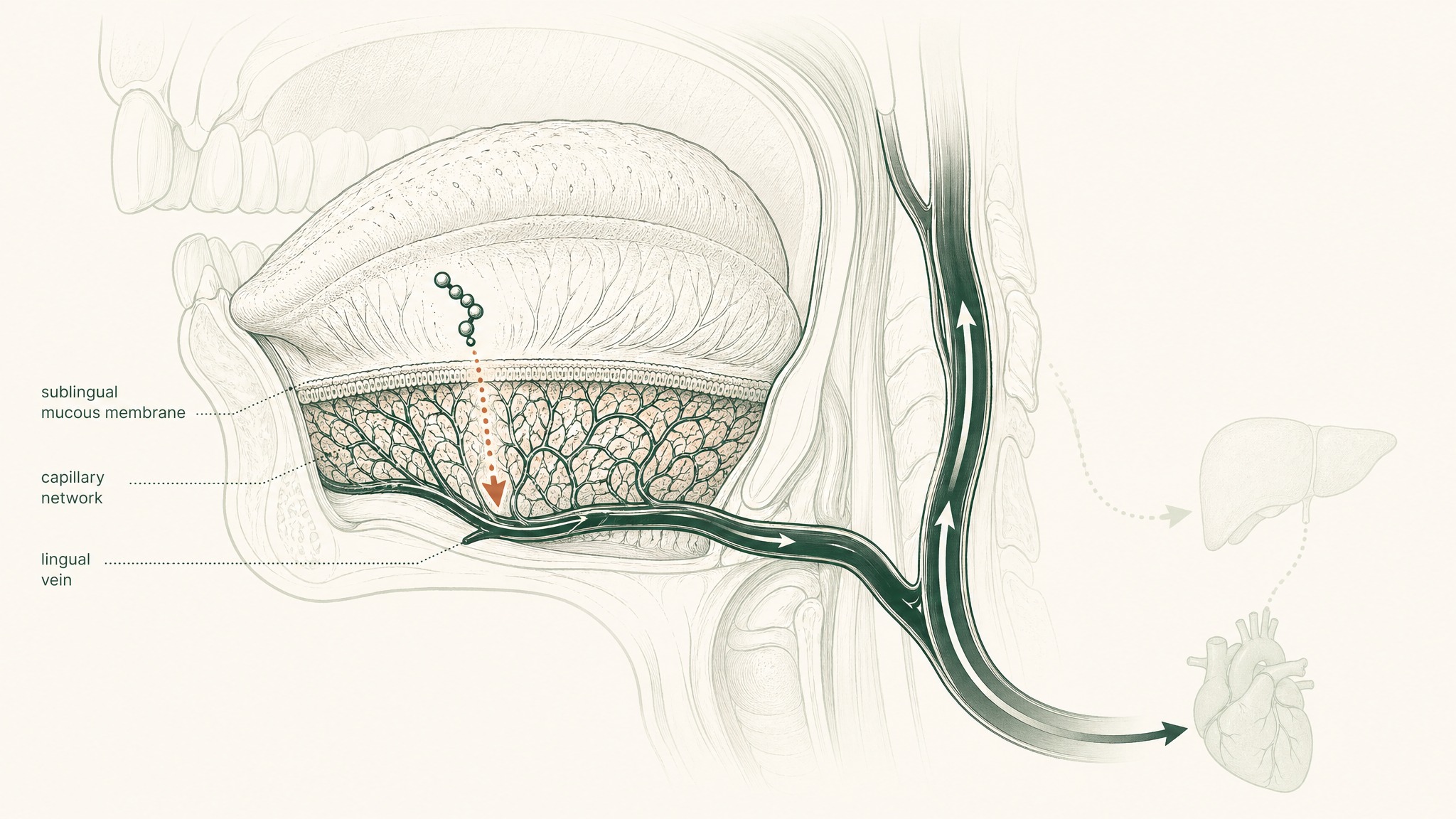
Your liver's portal system, the route that processes anything absorbed from your digestive tract before the rest of your body sees it, simply doesn't apply to sublingually absorbed compounds. The hepatic first-pass step that destroys most orally administered peptides is removed from the equation.
The catch is the membrane itself. It's permeable, but not freely permeable to every compound. Hydrophilic peptides — water-loving molecules that struggle to dissolve through the lipid-based mucosal barrier — don't cross efficiently on their own. Without formulation engineering, most of the compound gets swallowed involuntarily before meaningful absorption can happen. The route gets you to the door; the engineering is what gets you through it.
What the Research Shows About Mucosal Delivery of Bremelanotide
Research suggests mucosal delivery of bremelanotide isn't theoretical. A 2025 study by Krupke et al. published in Journal of Controlled Release developed a biodegradable patch for transbuccal (inner cheek) peptide delivery — applying compound directly to buccal tissue with a chemical permeation enhancer to facilitate membrane crossing. When tested against subcutaneous injection as the reference standard, buccal delivery of bremelanotide achieved 26% relative bioavailability (Krupke, Zoratto, Rabut et al., J Control Release 2025, PMID 40513668).
That's not injection-level exposure. But it's orders of magnitude beyond what oral delivery achieves — and it was accomplished without a needle.
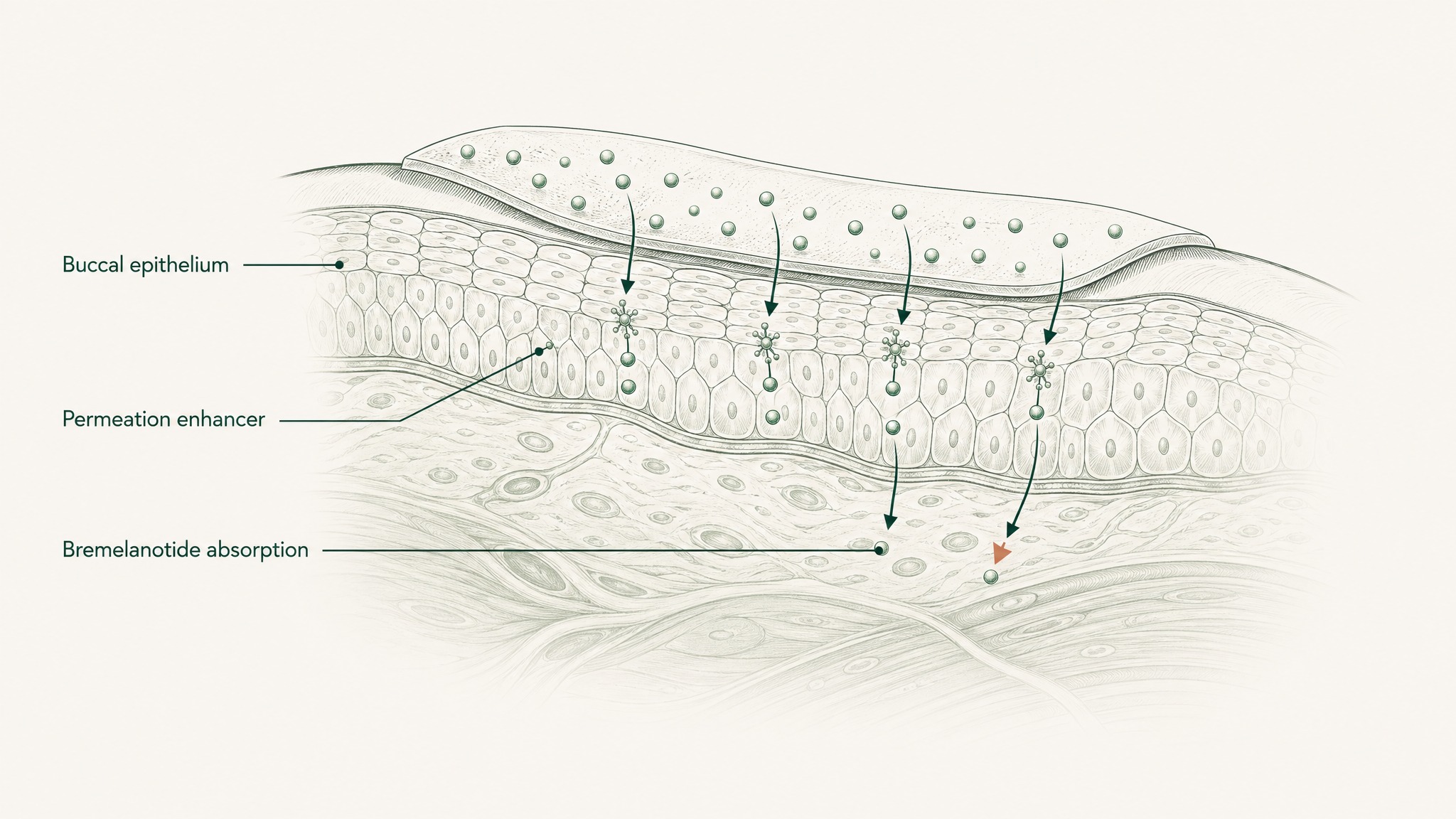
The critical variable in that result was the combination of extended tissue contact time and chemical permeation enhancement. The study confirmed enhanced permeation of the mucosal barrier when the enhancer was present, compared to formulations without it. Extended contact plus membrane-crossing facilitation — both components were required to produce meaningful absorption.
This mirrors the principle behind VERO's other sublingual research protocols: the delivery format you choose determines whether the biology you're trying to study is even accessible. For bremelanotide specifically, the 2025 data puts a concrete number on what well-formulated mucosal delivery can achieve.
The Safety Profile You Should Know
Users report that the most common side effect of bremelanotide is nausea — and the long-term extension data backs that up: nausea was reported by 40.4% of participants, flushing (warmth or skin redness) by 20.6%, and headache by 12.0% (Simon et al. 2019, PMID 31599847). The majority of these were mild to moderate in intensity, and they didn't worsen over 52 weeks of use.
There's also a transient blood pressure effect worth knowing about. A dedicated pharmacodynamic study in 397 premenopausal women found dose-dependent increases in systolic blood pressure of 2.4–3.2 mmHg in the 0–4 hour post-dose window, with peak increases typically lasting less than 15 minutes (White, Myers, Jordan & Lucas, J Hypertens 2017, PMID 27977473). These increases resolved on their own, but anyone with cardiovascular considerations should be aware of them before starting any protocol involving this compound.
One notable positive in the safety picture: research found no clinically significant interaction between bremelanotide and alcohol, which separates it from some other pharmacological approaches to sexual dysfunction (Mayer & Lynch, Ann Pharmacother 2020, PMID 31893927).
CLARITY has not been evaluated by the FDA for any indication. Any protocol decision should involve a qualified healthcare professional.
CLARITY and VERISORB: VERO's PT-141 Sublingual Research Protocol
CLARITY is VERO's PT-141 research protocol, delivered via VERISORB — VERO's proprietary sublingual delivery platform. VERISORB addresses the formulation gap between the established subcutaneous pharmacology of bremelanotide and the practical alternative of sublingual absorption.
The formulation applies mucoadhesive polymers and permeation-enhancing excipients to the specific challenge of getting a cyclic melanocortin peptide across the sublingual mucosa. The mucoadhesive component extends contact time with the mucosal surface — slowing the involuntary swallowing that would otherwise clear the compound before absorption begins. The permeation-enhancing excipient facilitates membrane crossing. Without both, the sublingual route is just a way to swallow something more slowly.
CLARITY is designed for researchers whose protocol endpoint involves the central melanocortin system's documented role in desire — specifically the MC4R–dopamine pathway in the medial preoptic area that the published literature has mapped in detail. It's formulated for sublingual administration and is not designed for injection or oral ingestion.
CLARITY is for research purposes only and has not been evaluated by the FDA. Protocol decisions should be made in consultation with a qualified healthcare professional.
Key Takeaways
- PT-141 (bremelanotide) is a synthetic, cyclic analog of alpha-MSH — a signalling peptide your body produces naturally from POMC. Cyclic structure makes it more stable than the linear natural hormone (Gantz & Fong 2003, PMID 12556347; Dhillon & Keam 2019, PMID 31429064).
- Its mechanism is brain-based, not hormonal. It activates MC4R in the hypothalamus's medial preoptic area, triggering dopamine release — the neurochemistry of motivation and desire (Pfaus et al. 2022, PMID 33455598).
- The RECONNECT Phase 3 trials (N=1,267) showed statistically significant improvements in desire scores (+0.35, p<0.001) and reduction in desire-related distress (-0.33, p<0.001) versus placebo. Effect was on-demand, dosed ~45 minutes before activity (Kingsberg et al. 2019, PMID 31599840).
- A 52-week extension study (N=684) showed sustained improvements and no new safety signals. Primary adverse events: nausea (40.4%), flushing (20.6%), headache (12.0%) (Simon et al. 2019, PMID 31599847).
- Oral delivery produces minimal useful absorption. Research found near-zero oral bioavailability for bremelanotide in animal models (Sauter et al. 2020, PMID 32353679) and just 4.6% oral bioavailability for the related cyclic melanocortin peptide Melanotan-II in rats (Lan et al. 1994, PMID 7983590).
- Formulated buccal delivery achieved 26% relative bioavailability versus subcutaneous injection when combined with a chemical permeation enhancer — demonstrating that mucosal absorption of bremelanotide is achievable and meaningful without injection (Krupke et al. 2025, PMID 40513668).
- Transient blood pressure increases of 2.4–3.2 mmHg (lasting <15 minutes) have been documented. Consult a healthcare professional before beginning any protocol (White et al. 2017, PMID 27977473).
- CLARITY via VERISORB is VERO's structured PT-141 sublingual research protocol. For research purposes only.
Ready to explore a structured PT-141 sublingual protocol? Explore CLARITY →
This content is published for research and educational purposes only. It is not intended to diagnose, treat, cure, or prevent any disease, and it does not constitute medical advice. Readers should consult a qualified healthcare professional before making any decisions about their own health. Statements on this page have not been evaluated by the FDA. VERO's peptide products are supplied for research purposes only.
CLARITY™
Coming SoonEngineered around 10mg Dihexa. Drives advanced synaptic connectivity and neurogenesis
Clinical Context
Important Notice: VERO protocols are nutritional and systemic optimisation formats. They are not intended to diagnose, treat, cure, or prevent any medical condition. These statements have not been evaluated by the Food and Drug Administration.