Most peptide pills deliver under 2% of their dose to your bloodstream. Here's what bioavailability means, how it's measured, and what actually gets through.
Here's the number that changes how you read every peptide supplement label: the bioavailability of a standard oral peptide capsule — meaning how much of what you swallow actually reaches your bloodstream — sits below 2 percent. Out of 100 milligrams in a capsule, roughly 98 milligrams never make it into your blood. They don't sit unused somewhere; they get systematically dismantled in transit. This content is published for research purposes only.
Understanding why that number is where it is, and what it means for any research protocol built around oral peptide pills, is the whole point of this piece.
What Bioavailability Actually Means for a Peptide Pill
Bioavailability (how much of what you take actually reaches your bloodstream in a form your body can use) is the most important number in peptide supplementation. It's almost never on the label.
Think of it like this: the label tells you what's in the capsule, not what arrives in your blood. Those are two different numbers, and for oral peptide pills, the gap between them is enormous.
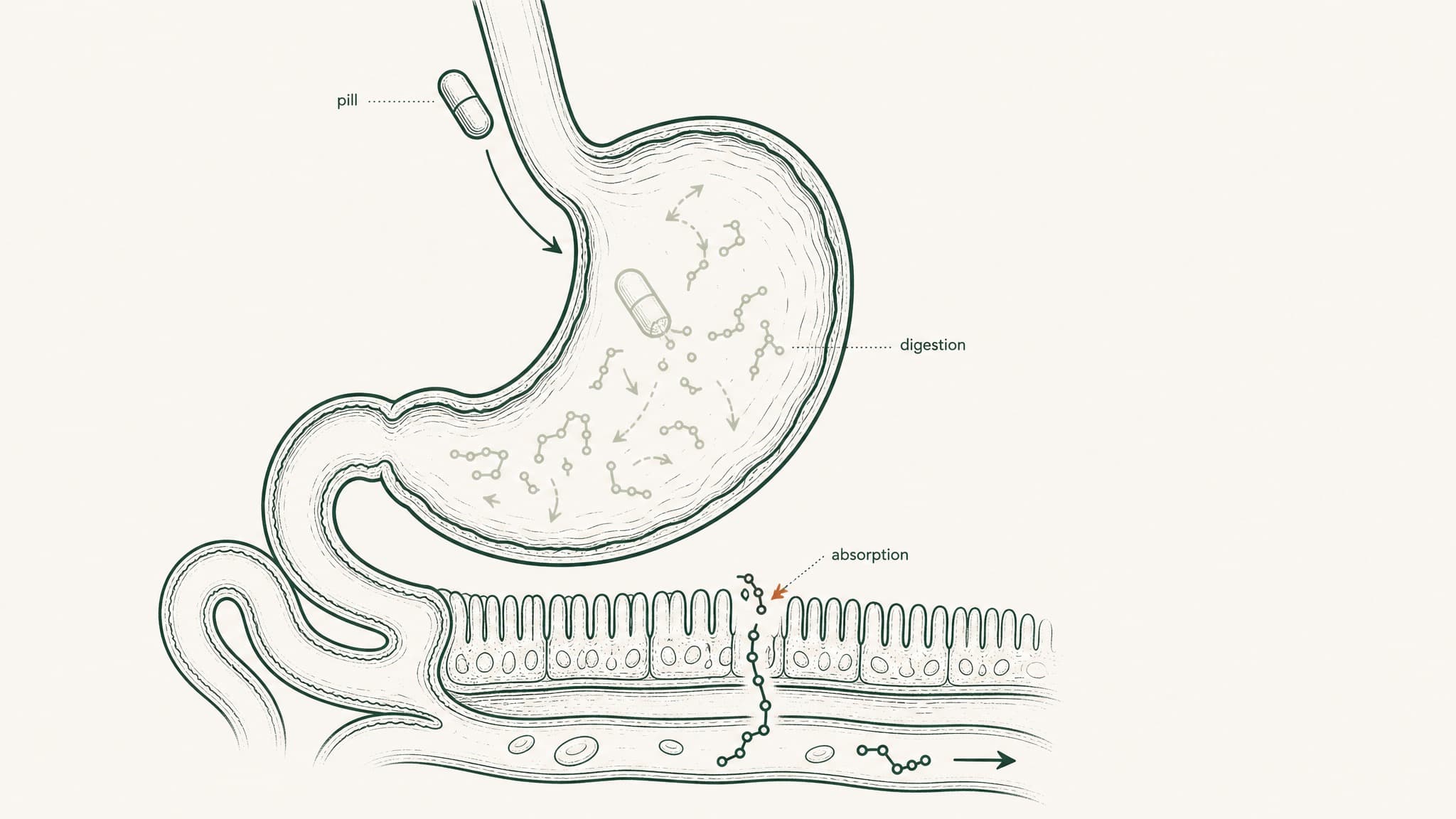
Why only 1–2% of an oral peptide pill reaches your bloodstream instead of the full labeled dose.
Here's what bioavailability actually measures:
- What's in the capsule: the milligram count on the label, what you paid for
- What survives digestion: the fraction not destroyed in your stomach and intestines
- What crosses into your bloodstream: the fraction that successfully passes through the intestinal wall
- What your liver didn't process first: the fraction that reaches systemic circulation before your liver takes its cut
Bioavailability is the number you get at the end of that chain. For standard oral peptide capsules, research documents it consistently below 1 to 2 percent (Renukuntla et al. 2013, Int J Pharm, PMC3680128). The label milligram count and the bioavailable milligram count are not the same thing. They're not even close.
Why Oral Capsules Deliver Under 2%: The Five Gut Barriers
Your digestive system is a tiered protein-dismantling machine. It wasn't designed to block peptide supplements; it was designed to process food. But peptides are chains of amino acids, and food protein is also chains of amino acids, and the gut runs both through the same destruction sequence without distinguishing between them.
Here's what that sequence does to a peptide pill from the moment you swallow it:
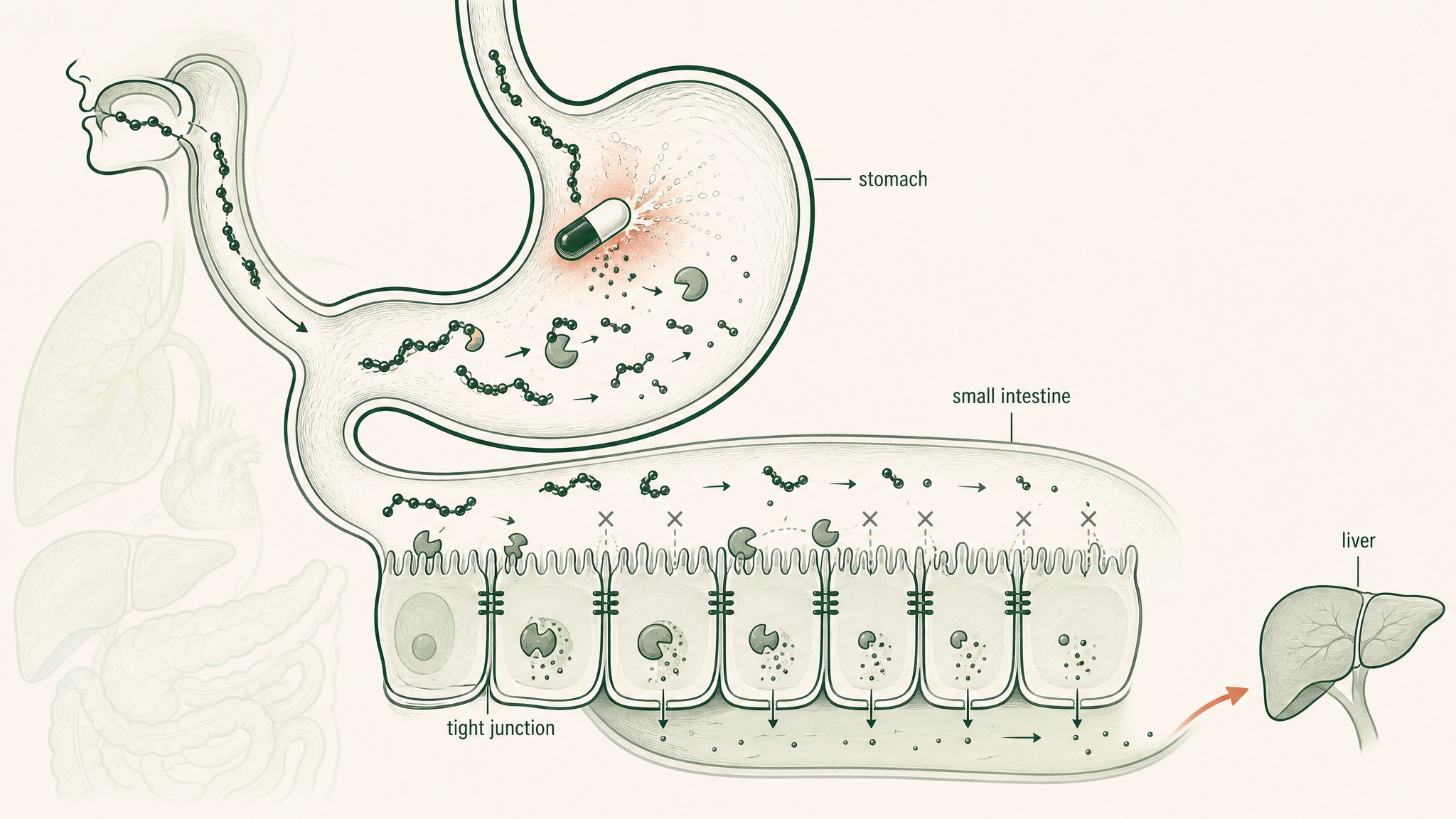
Why oral peptides get destroyed at five checkpoints before reaching your bloodstream.
- Stomach acid (pH 1.5–3.5, roughly battery-acid territory): begins unfolding molecular architecture and activates pepsin, an enzyme whose entire function is cutting peptide bonds
- Brush-border peptidases: enzymes coating the surface of your small intestine wall that continue dismantling whatever survived the stomach
- Epithelial barrier: a single continuous cell layer acting as both a physical wall and a biochemical checkpoint, including intracellular enzymes that process anything attempting to cross
- Tight junctions: protein seals between intestinal cells that block most molecules above a certain size; most research peptides don't fit through
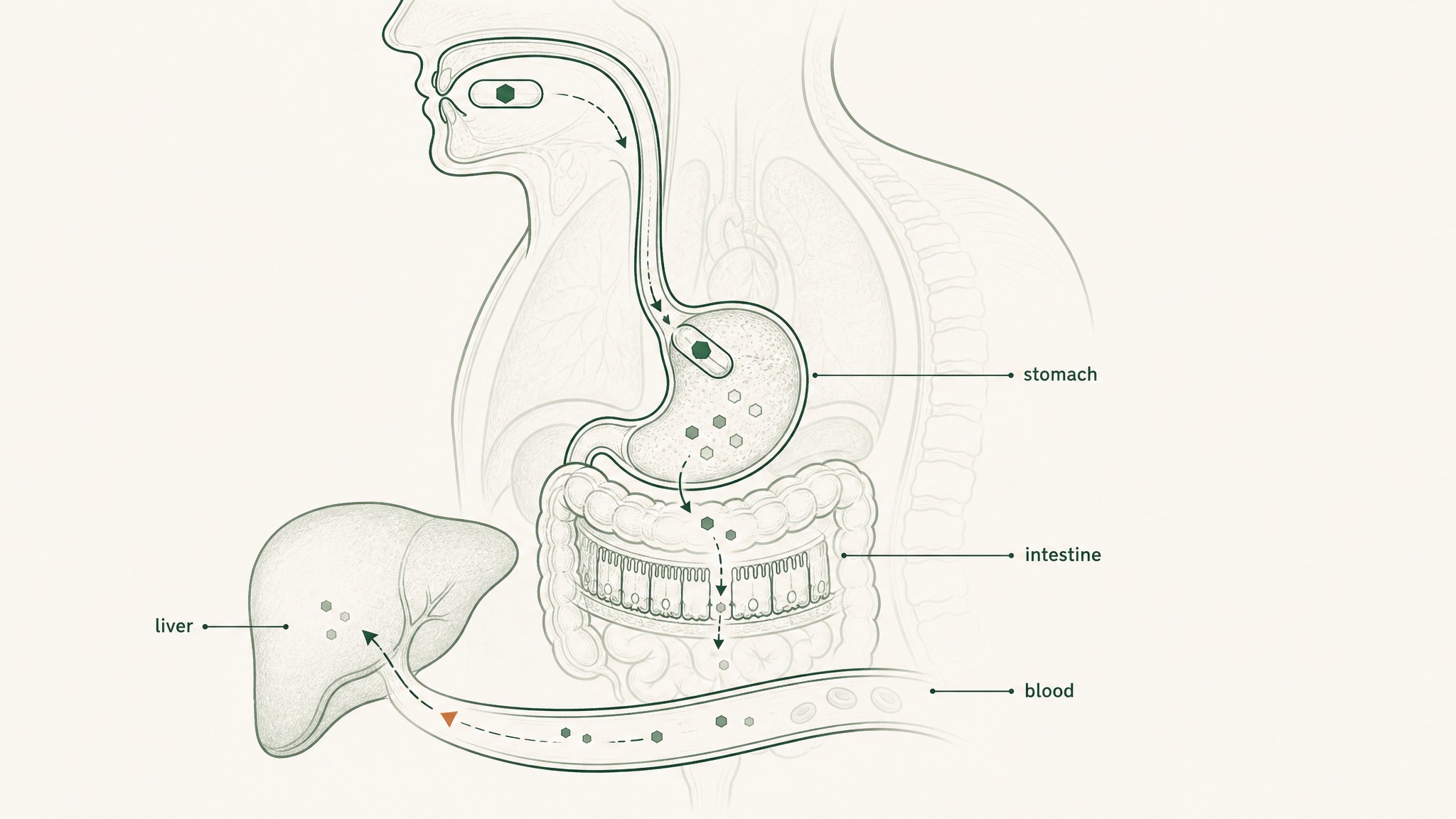
- Hepatic first-pass metabolism: everything absorbed from the gut routes to your liver before the rest of your body sees it; your liver runs its own protease enzymes and takes a final cut
These barriers don't each subtract a fixed percentage from the original dose. They compound, each one operating on whatever the previous stage left behind (Verma et al. 2021, Drug Dev Res, PMID 33988872). That compounding is how you arrive at sub-2-percent bioavailability even from a well-manufactured, pharmaceutical-grade capsule: not a pessimistic estimate, but the documented outcome across peptide classes.
The Real Cost of Sub-2% Bioavailability
Most people evaluate peptide products by milligram count and price per serving. The number that actually matters is effective delivery: how many milligrams reach your bloodstream per dollar spent.
Here's the arithmetic. A standard peptide capsule protocol at 100mg per day with 1.5% bioavailability delivers approximately 1.5mg of systemic peptide exposure daily. That's 45mg per month of actual bloodstream delivery from a label dose of 3,000mg per month.
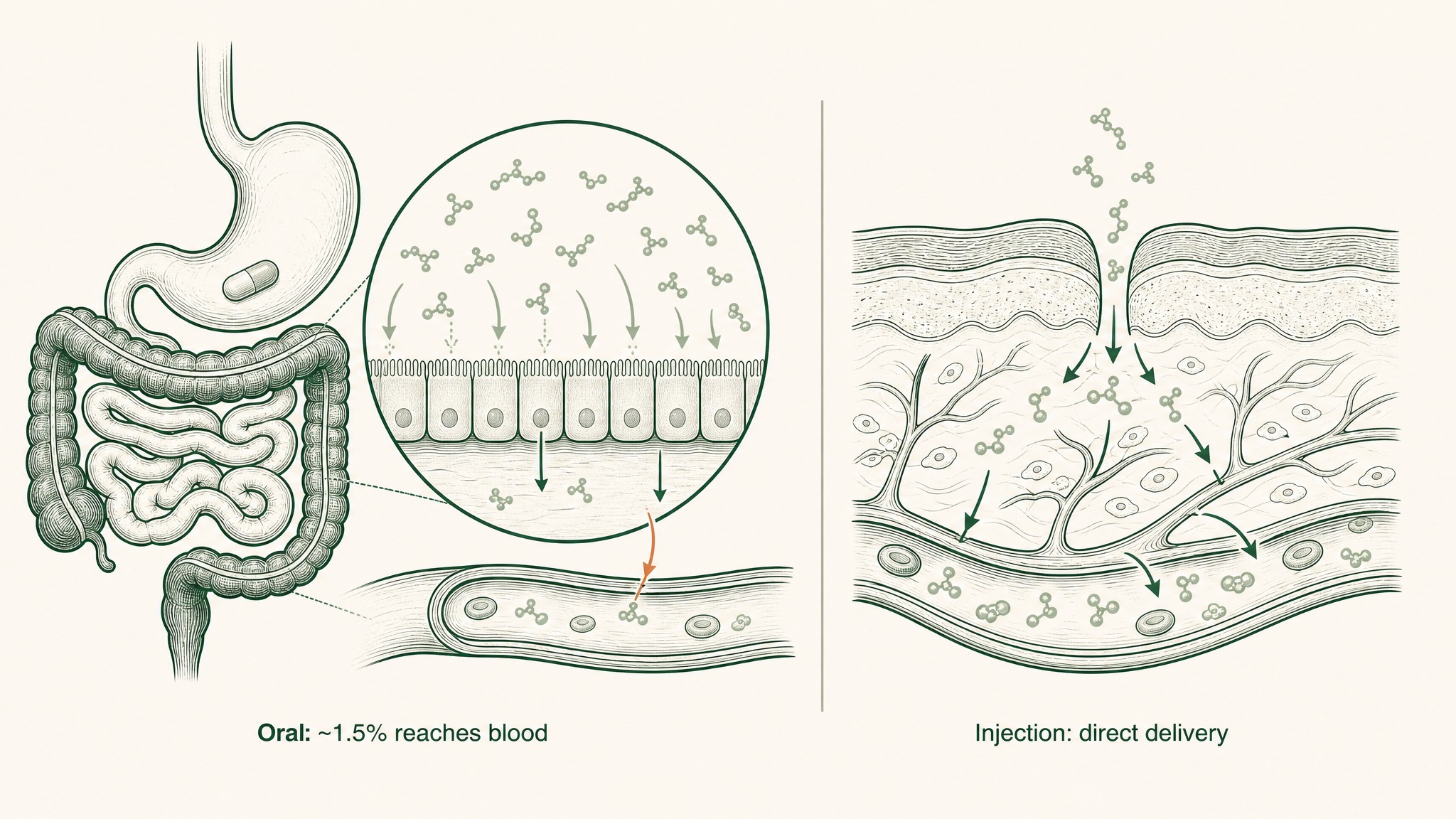
Why injected peptides reach your bloodstream in far greater amounts than swallowed pills.
You're paying for 3,000mg. Your blood receives roughly 45mg.
Subcutaneous injection, the reference standard for peptide delivery in research, deposits the compound directly into vascularised tissue beneath the skin, bypassing the gastrointestinal tract entirely (Zhang et al. 2020, Int J Pharm, PMID 32622810). The same compound at the same dose via injection produces fundamentally different systemic exposure: not marginal variation, but an order-of-magnitude difference.
Research suggests the effective-dose calculation matters most for compounds that require specific systemic concentrations to produce the outcomes being studied. A compound that needs a meaningful blood level to do anything useful doesn't get there at 1.5% bioavailability, regardless of the milligram count on the label.
High-Purity Capsules Don't Fix the Delivery Problem
This is the most common misconception in the peptide supplement space. Buyers assume that a high-purity, third-party-tested capsule delivers more peptide to the bloodstream than a cheaper product. Quality of ingredient and delivery of that ingredient are completely separate problems.
Purity is what's in the capsule. Bioavailability is what reaches your blood. These are different measurements.
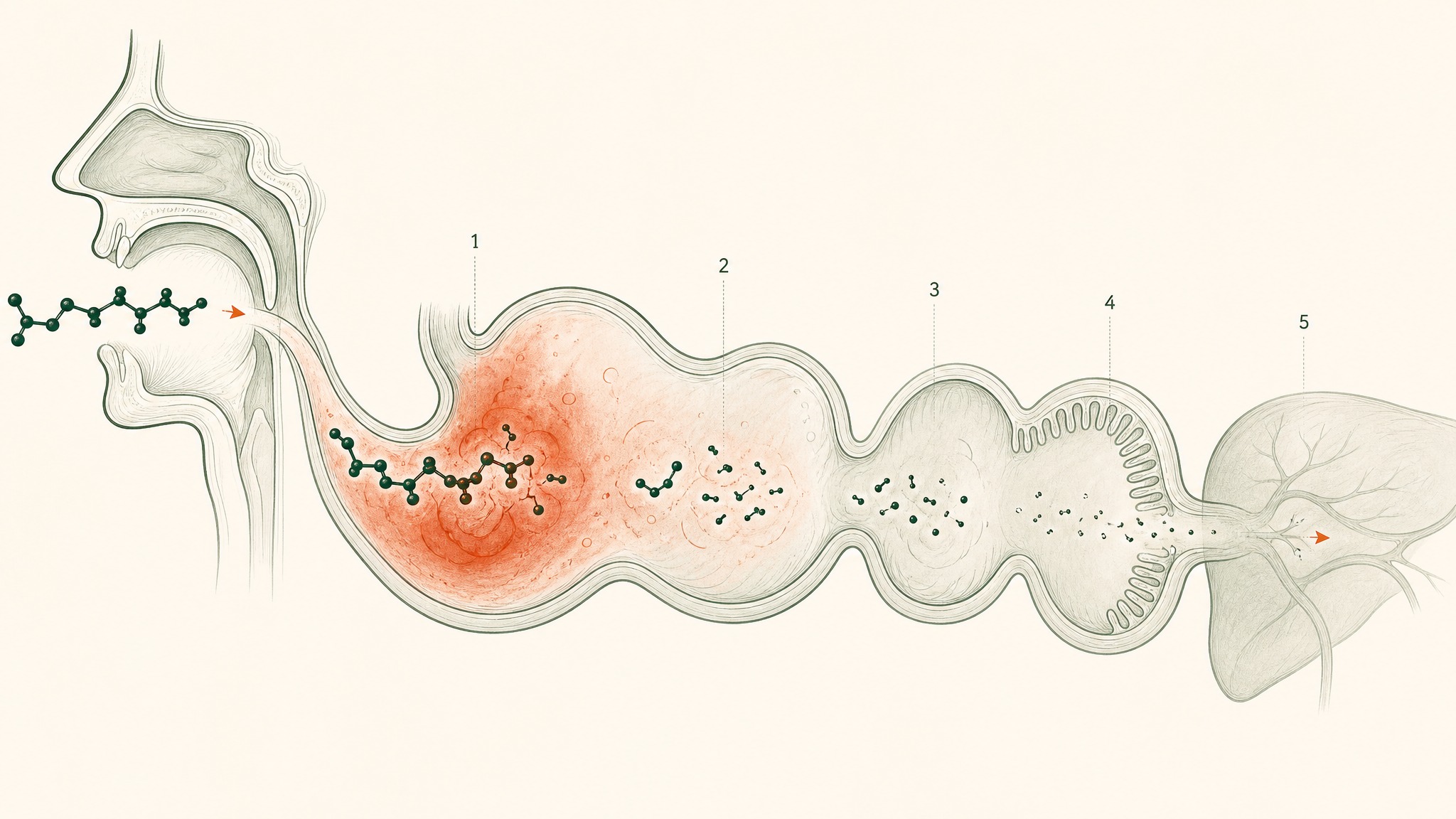
How stomach acid and enzymes destroy peptides before they reach the bloodstream.
Third-party certificates of analysis tell you what's in the capsule. They don't measure what reaches your blood after your gut is done with it. A 99.9%-pure peptide in a standard oral capsule faces the same five-stage destruction sequence as a lower-quality product. Purity affects what goes in, not how much gets through.
Users report disappointment when premium oral peptide products don't produce the results their marketing implies. That gap isn't surprising; it's exactly what the pharmacokinetics predict. The premium is on the synthesis quality. The barrier is on the delivery format.
The evidence on what serious pharmaceutical engineering achieves makes this concrete. Oral semaglutide (Rybelsus), developed with SNAC (a proprietary absorption enhancer engineered specifically for this one compound over years of pharmaceutical research), taken 30 minutes before food under strict dosing conditions, achieves absolute bioavailability of 0.8 percent (Overgaard et al. 2021, Clin Pharmacokinet, PMID 33969456). That's the pharmaceutical ceiling. A standard supplement capsule doesn't use SNAC technology, has no access to the gastric absorption pathway it creates, and faces the full five-stage destruction sequence from pH 1.5 onward.
The One Gut Transport Mechanism — and Why It Doesn't Apply
There is a transporter protein in your intestinal wall that actively absorbs intact peptides. It's called PepT1, encoded by the SLC15A1 gene. PepT1 shuttles peptide fragments across the intestinal wall via a hydrogen-ion co-transport mechanism (Freeman 2015, World J Gastrointest Pharmacol Ther, PMC4419090).
Here's why this doesn't rescue oral peptide pills:
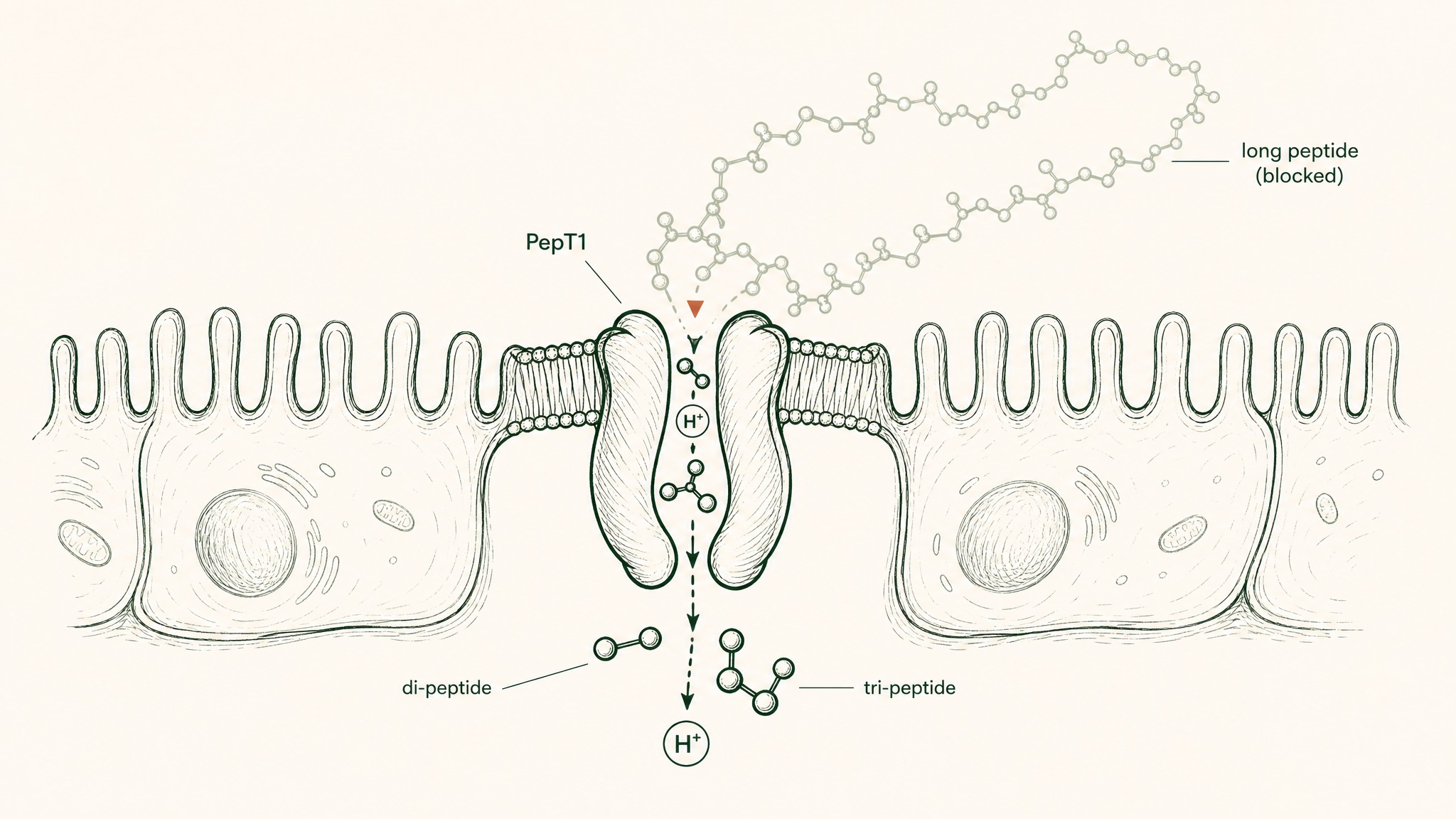
PepT1 transporter accepts only two- or three-amino-acid chains, not the longer peptides in supplements.
- PepT1 only transports di-peptides and tri-peptides, chains of exactly two or three amino acids
- Its biological function is absorbing the output of food digestion, not transporting intact research compounds
- Most research peptides are far longer than three amino acids; BPC-157 is 15 amino acids long
The one transporter that could theoretically help is designed for the wrong chain length.
PepT1 was built for food protein digestion. It works exactly as designed, just not in a way that benefits oral peptide supplementation. It's a small window in a very tall wall.
How Researchers Actually Measure Peptide Bioavailability
Bioavailability figures come from pharmacokinetic studies, research that measures a compound's concentration in blood plasma at multiple time points after administration. Here's how the measurement actually works:
- Give the subject the compound via the route being tested (oral capsule, sublingual, subcutaneous).
- Take blood samples at regular intervals after administration.
- Plot the concentration-time curve: how blood levels rise and fall over time.
- Calculate the area under that curve (AUC), a measure of total drug exposure over time.
- Compare this AUC to the AUC produced by intravenous administration of the same dose, which represents 100% delivery by definition.
- The ratio between the two gives you the bioavailability percentage.
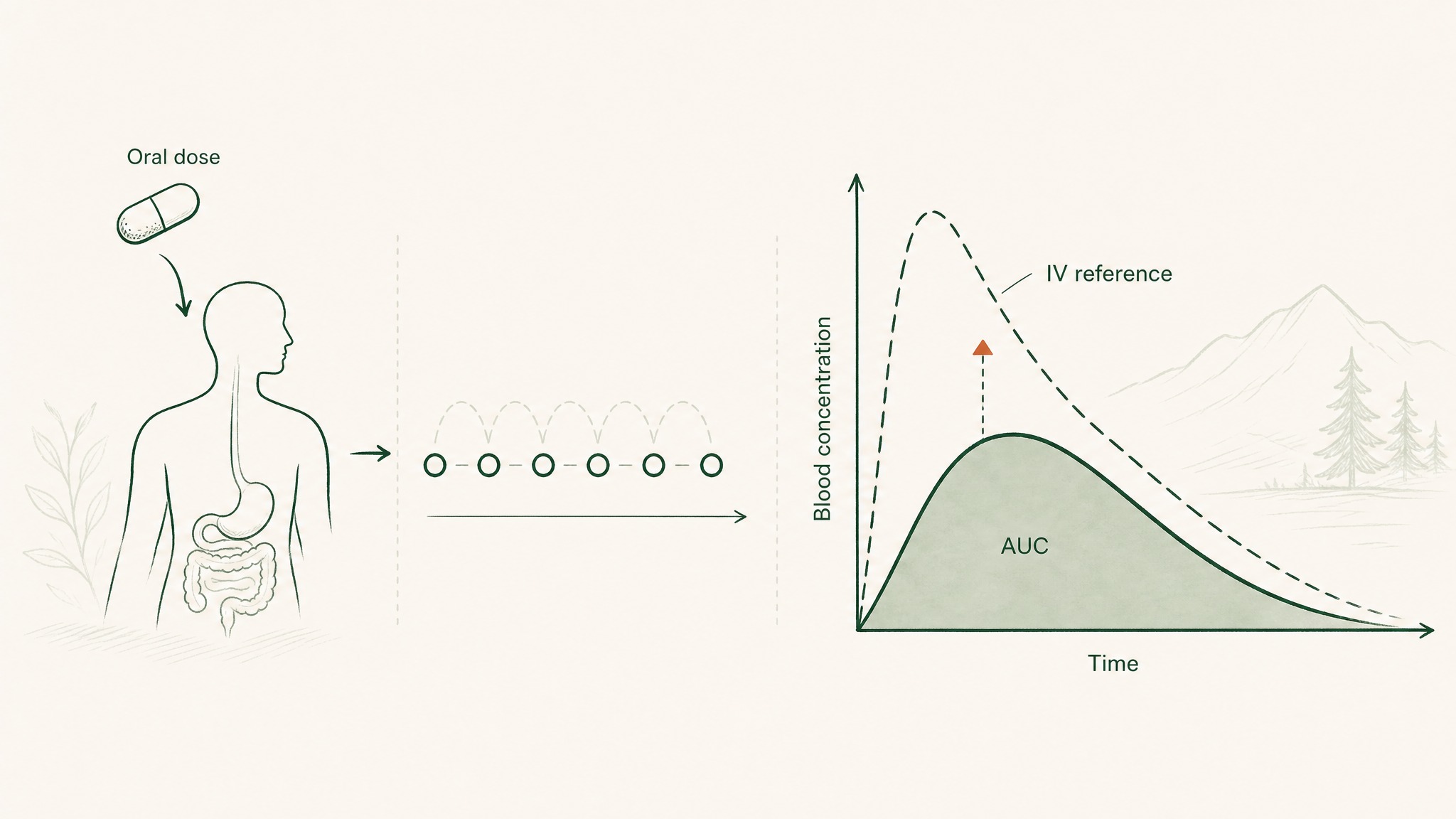
How researchers measure bioavailability: blood concentration rises and falls after a dose, and the area under that curve is compared to intravenous delivery.
For most research peptides, this data comes from animal models, primarily rodent studies. Human pharmacokinetic data for research compounds is sparse because most haven't gone through human clinical trial programmes.
The extrapolation runs in an uncomfortable direction. Rodents have proportionally faster gastrointestinal transit and more efficient absorption than humans for many compounds, so rat model bioavailability figures often come in above what humans achieve. The documented <2% figure isn't the worst case: it's the central tendency across the literature, already accounting for species differences.
Sublingual Delivery vs Oral Capsule: What the Bioavailability Data Shows
The pharmaceutical approach to improving oral peptide bioavailability is to engineer around individual barriers. Liposome formulations (lipid spheres that carry the peptide through the gut wall rather than exposing it directly to the enzyme environment) have achieved meaningful results for specific compounds in research: approximately 3.4 percent bioavailability for human growth hormone, and up to 19 percent for exendin-4 using glycocholic acid-conjugated chitosan-coated formulations (Cui et al. 2022, Pharmaceuticals, PMC9501131). These are compound-specific, formulation-specific outcomes that don't transfer between peptides.
A different approach sidesteps the gut entirely. The underside of your tongue is lined with non-keratinised epithelium, thinner and more permeable than any surface in your digestive tract, sitting directly over a dense venous network. Compounds absorbed there drain into the sublingual and lingual veins, reaching systemic circulation before your liver ever sees it — five gut barriers skipped entirely (Zhang, Zhang & Streisand 2002, Clin Pharmacokinet, PMID 12126458).
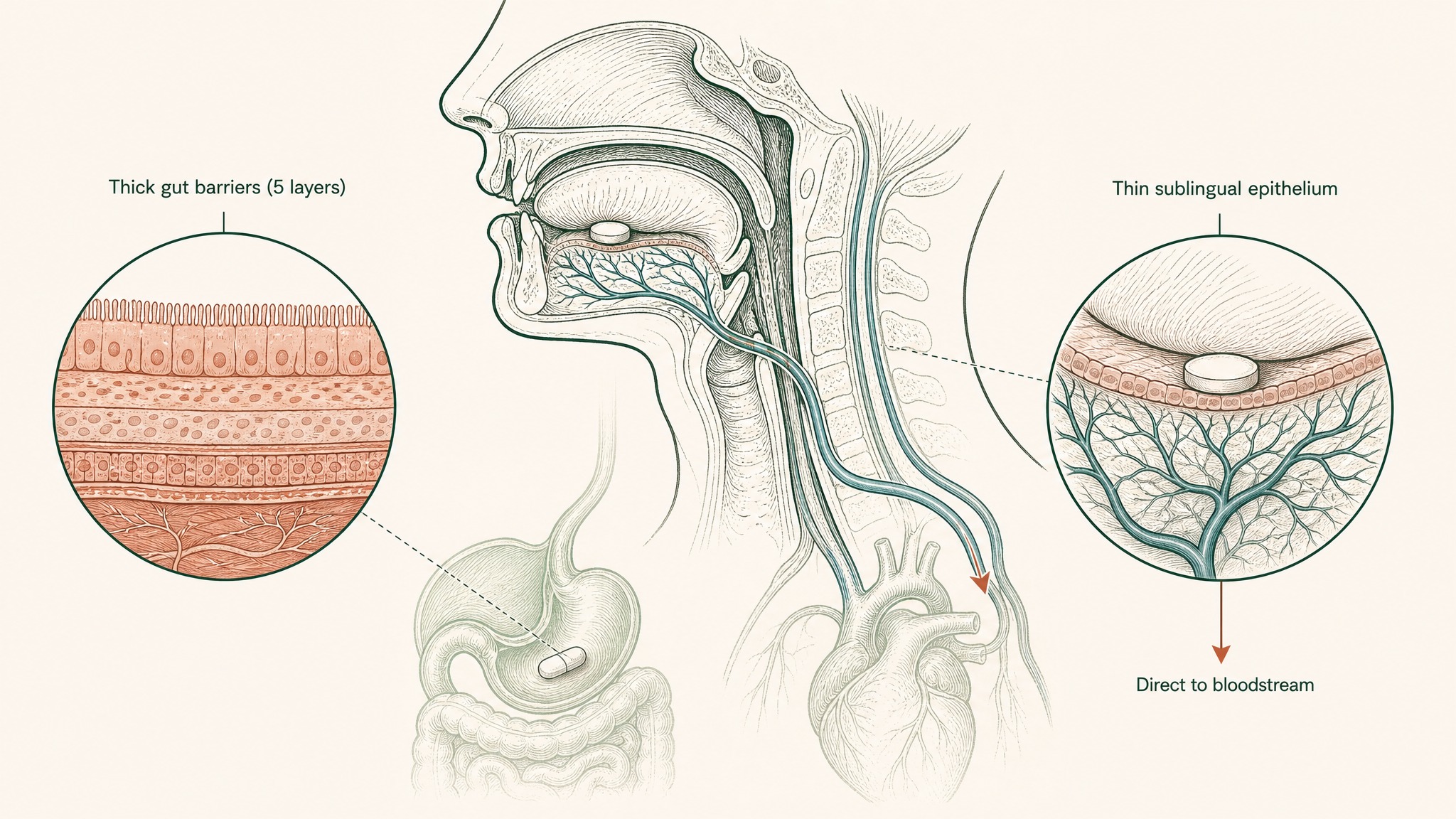
Why the thin tissue under your tongue absorbs peptides five times faster than your stomach does.
That's not one barrier removed. That's five.
Purpose-formulated sublingual peptide preparations have demonstrated bioavailability of up to 24.1 percent relative to a subcutaneous injection reference in formulation research (Patil & Devarajan 2016, Drug Deliv, PMID 24901208). These are compound-specific, formulation-specific figures that don't automatically transfer across peptide classes. What they establish is the order-of-magnitude shift the sublingual route produces when the formulation is specifically engineered for it.
An unformulated peptide under the tongue doesn't automatically absorb any more than a capsule in the stomach does. The sublingual mucosa is one barrier, not five. But it's still a barrier. What makes the route work is engineering: mucoadhesive polymers that hold the compound against the mucosal surface long enough to cross, permeation enhancers matched to each peptide's molecular characteristics.
Members experience the practical side of this most clearly in extended protocols. A delivery format that produces meaningful systemic exposure without requiring injection changes what actually gets followed through across six or twelve weeks consistently. That consistency is its own variable in any research protocol.
VERO's VERISORB platform is built specifically around the sublingual route: mucoadhesive polymer systems formulated compound-by-compound, addressing the one remaining barrier rather than fighting through five.
Key Takeaways
- Bioavailability (how much of a dose actually reaches your bloodstream) is below 2% for standard oral peptide capsules (Renukuntla et al. 2013, PMC3680128). The label milligram count and the bloodstream milligram count are not the same number.
- The gut's five-stage destruction sequence (gastric acid and pepsin, brush-border peptidases, epithelial barrier, tight junctions, hepatic first-pass) compounds across each stage, each one working on whatever the previous left behind (Verma et al. 2021, PMID 33988872).
- The PepT1 intestinal transporter, the gut's only active peptide uptake mechanism, accepts only di-peptides and tri-peptides. Most research peptides are far longer than that threshold (Freeman 2015, PMC4419090).
- High-purity capsules face the same delivery barriers as lower-quality ones. Purity of ingredient and delivery to bloodstream are separate variables.
- The best oral peptide bioavailability pharmaceutical engineering has achieved commercially is 0.8% (semaglutide with SNAC, strict dosing conditions — Overgaard et al. 2021, PMID 33969456). Standard supplement capsules don't have access to that absorption pathway.
- Sublingual delivery removes five gut barriers. Purpose-formulated preparations have reached up to 24.1% bioavailability relative to subcutaneous injection in formulation research (Patil & Devarajan 2016, PMID 24901208).
- Route determines systemic exposure as directly as the label dose. A protocol built on oral pill numbers is working against its own pharmacokinetics.
Ready to explore a delivery format built around the sublingual route? Start with the RESTORE Protocol →
This content is published for research purposes only and does not constitute medical advice. It is not intended to diagnose, treat, cure, or prevent any disease. Anyone considering a peptide research protocol should consult a qualified clinician before beginning.
RESTORE™
BPC-157Engineered around 500mcg BPC-157. Accelerates deep tissue and joint recovery
Clinical Context
Important Notice: VERO protocols are nutritional and systemic optimisation formats. They are not intended to diagnose, treat, cure, or prevent any medical condition. These statements have not been evaluated by the Food and Drug Administration.