BPC-157 isn't FDA-approved, but its regulatory story is more nuanced than a simple yes or no. Here's what the current compounding rules, clinical trial gaps, and research landscape actually look like.
If you've spent any time looking into BPC-157, you've probably hit the same wall: vague answers, contradictory forum posts, and a regulatory picture that seems designed to confuse. The compound isn't banned. It isn't approved. It sits somewhere in between, in a genuinely complicated part of American drug regulation that most explainers gloss over.
This is a detailed walkthrough of where BPC-157 actually stands with the FDA in 2026 — what that means, why it got there, and what the underlying research looks like once you separate it from the noise.
What BPC-157 Actually Is
BPC-157 stands for Body Protection Compound 157. It's a synthetic peptide — a short chain of 15 amino acids — that researchers derived from a protein naturally present in human gastric juice. Think of amino acids as LEGO bricks: most proteins are enormous structures made of hundreds of them. BPC-157 is a very specific 15-brick arrangement that researchers isolated from a much larger protein found in your stomach.
The gastric juice origin matters for a few reasons. First, it suggests at least some biological precedent for the compound existing in human tissue. Second, it's part of why BPC-157 has attracted so much interest from researchers studying gut protection and tissue repair. And third, it's central to the patent problem that explains much of the regulatory gap — more on that shortly.
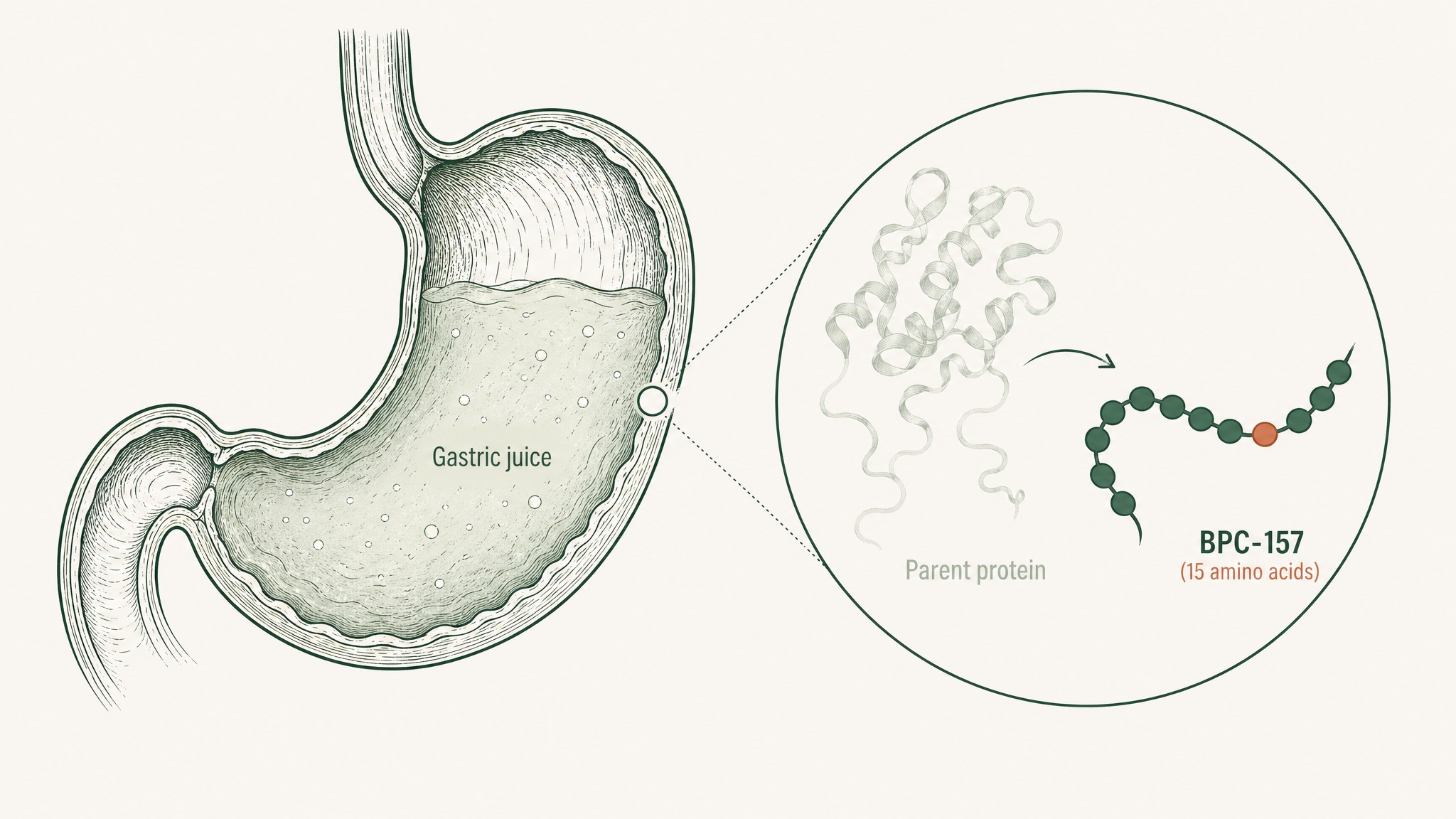
BPC-157 is not extracted from animals or plants. It's synthesised in a lab, which means what researchers work with is a precisely manufactured, chemically defined molecule.
FDA Approval — What It Actually Requires
Before getting into BPC-157 specifically, it's worth understanding what "FDA approval" actually means, because the term gets used imprecisely.
FDA approval for a drug requires completing three phases of human clinical trials. Phase 1 establishes safety in a small group of people. Phase 2 tests efficacy in a larger group. Phase 3 confirms efficacy and monitors side effects in a much larger randomised controlled trial — typically thousands of participants, run across multiple sites, over several years. The full process routinely costs $1–2 billion and takes a decade or more.
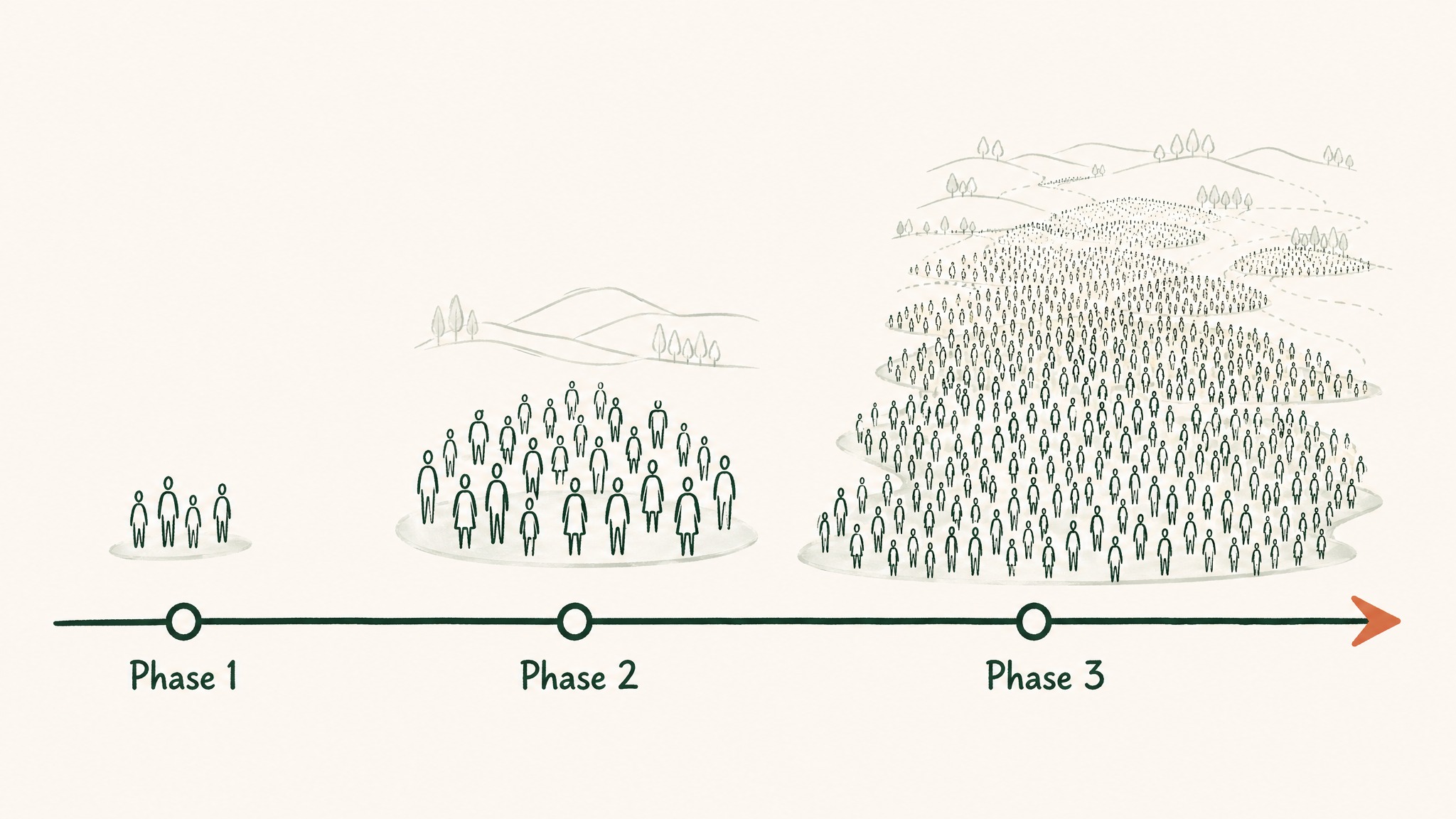
BPC-157 has not completed any of these phases. A 2025 review published in the journal Pharmaceuticals confirmed this directly: BPC-157 "has not been approved for use in standard medicine by the FDA and other global regulatory authorities due to the absence of sufficient and comprehensive clinical studies confirming its health benefits in humans." [PMID 40005999]
That's not a verdict that BPC-157 is dangerous. It's a statement about where the evidence sits in the clinical trial hierarchy. The FDA approval pathway requires human trial data. BPC-157 doesn't have it.
Why the Human Trials Haven't Happened
This is where the story gets structurally interesting — and where blaming regulatory obstruction misses the actual reason.
Pharmaceutical companies fund clinical trials when they can recoup the investment through patent protection. If you hold the patent on a drug and it gets approved, you have exclusive market rights for years, which is how you recover the cost of developing it.
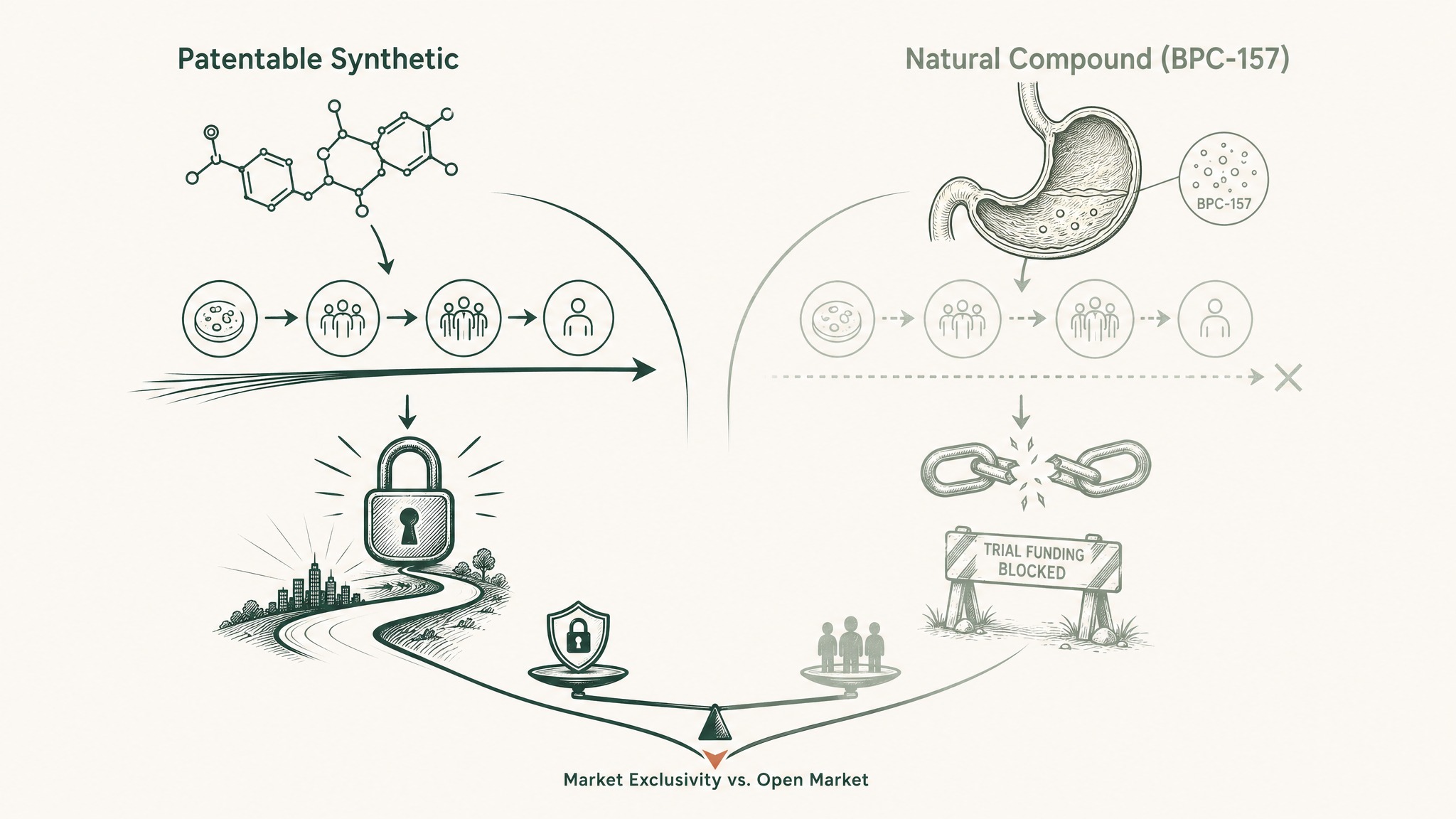
BPC-157 is a fragment of a protein that naturally occurs in human gastric juice. That makes it extremely difficult to patent in a way that gives a company exclusive commercial rights. Without patent protection, no pharmaceutical company has a financial incentive to spend a billion dollars on clinical trials. The compound isn't secret or dangerous — it's commercially unattractive for the entities with the resources to run trials.
This pattern isn't unique to BPC-157. It applies to many naturally occurring compounds that show interesting biological activity in research but never make it through clinical trials because the business case doesn't support the investment. The research sits in animal models, the compound stays in a grey zone, and curious researchers are left with preclinical data and limited human evidence.
The Compounding Pharmacy Question
There's a separate regulatory track that matters here: compounding pharmacies. These are specialised pharmacies that legally prepare custom drug formulations for individual patients — it's a bespoke service rather than mass production. They operate under their own set of FDA rules, governed by Section 503A of the Federal Food, Drug, and Cosmetic Act.
The FDA maintains a list of "bulk drug substances" — raw pharmaceutical ingredients — that compounding pharmacies are either allowed or prohibited from using. Being on the permitted list doesn't mean a substance is FDA-approved in the conventional sense; it means the FDA has evaluated it for use in this specific, more restricted context.
As of 2026, BPC-157 is actively being reviewed for inclusion on that 503A Bulks List. A Federal Register notice published in April 2026 (FR 2026-07361) confirmed that BPC-157 acetate and BPC-157 free base are on the agenda for the FDA's Pharmacy Compounding Advisory Committee, specifically under evaluation for a compounding use case relating to ulcerative colitis.
This is worth noting carefully. "Under evaluation" is not the same as approved, and it's not the same as prohibited. It means the formal conversation about BPC-157's regulatory legitimacy is ongoing and active — not settled.
What the Animal Research Actually Shows
Here's where things get genuinely interesting for researchers. While the human clinical record is limited, the preclinical data — studies in animal models and cell cultures — is substantial.
A 2019 narrative review published in Cell and Tissue Research [PMID 30915550] examined the body of BPC-157 research and found consistently positive results across multiple tissue types, from tendons and ligaments to muscle and bone. The authors were honest about the limits: "the efficacy of BPC 157 is yet to be confirmed in humans." But they noted that every animal model study they reviewed pointed in the same direction.
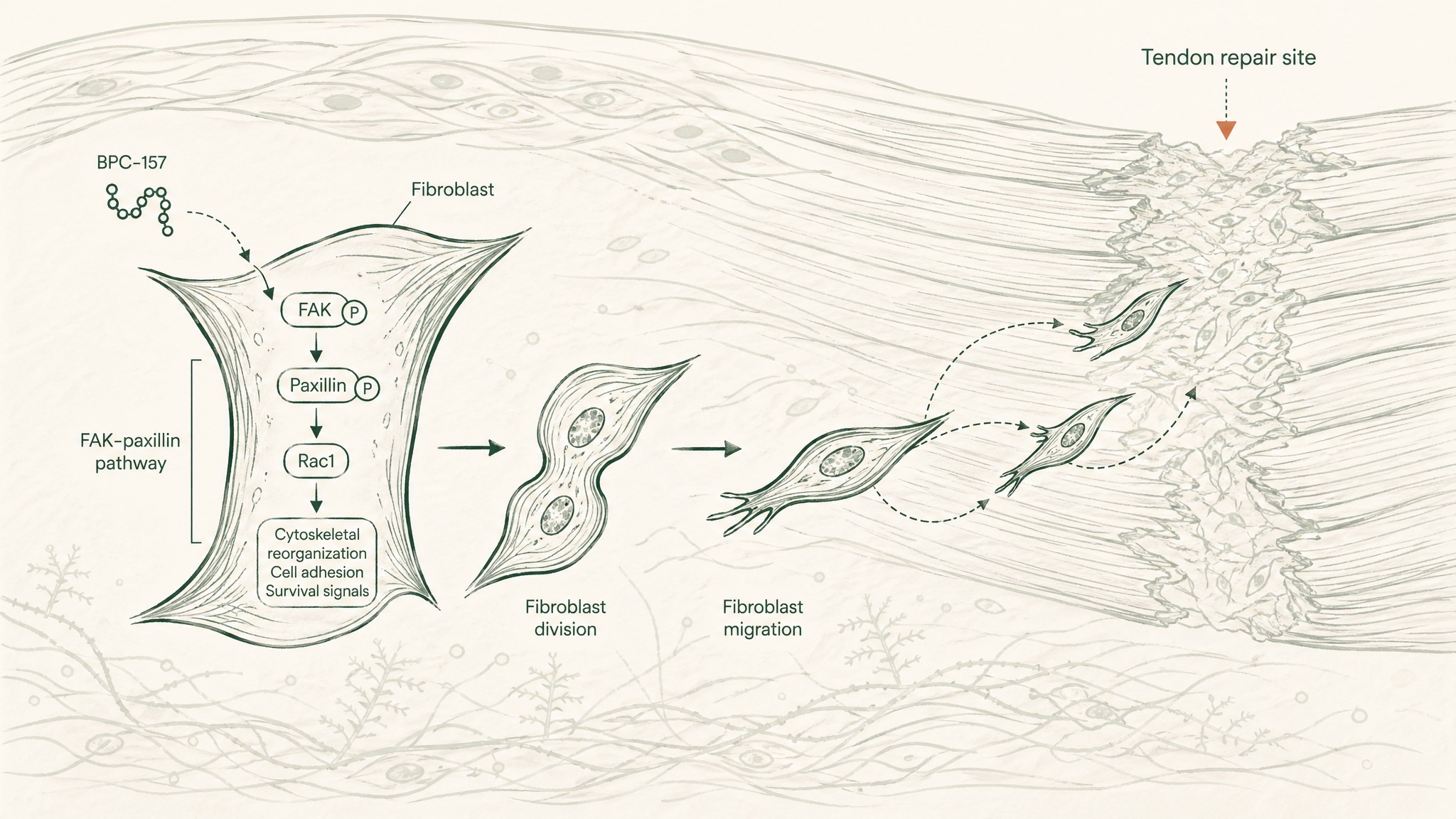
Research suggests the mechanism involves several pathways simultaneously. A 2011 study in the Journal of Applied Physiology [PMID 21030672] looked specifically at tendon fibroblasts — the cells responsible for building and maintaining tendons. BPC-157 significantly accelerated tendon tissue outgrowth and cell migration in rat models, working through what researchers call the FAK-paxillin pathway. In plain terms: a signalling chain that tells fibroblast cells to divide and move toward a site that needs repair.
On the gut side, a 2020 review in Current Pharmaceutical Design [PMID 32445447] documented BPC-157's cytoprotective effects — its apparent ability to preserve the integrity of the stomach lining against damage from NSAIDs like ibuprofen and aspirin. The connection between BPC-157's natural origin in gastric juice and its apparent protective effects on gastric tissue is something researchers find mechanistically coherent.
A 2021 review in Frontiers in Pharmacology [PMID 34267654] covered wound healing across skin, muscle, tendon, and connective tissue. It also noted something that doesn't always make it into general discussions: BPC-157 had previously been used in clinical trials for ulcerative colitis and multiple sclerosis, with no reported toxicity even at high experimental doses in animal safety studies. The lethal dose threshold — the dose at which 1% of animal subjects die, which researchers call LD1 — was never reached in preclinical safety testing.
The Human Evidence Gap — And What Does Exist
It would be dishonest to write this without being clear: the human data on BPC-157 is thin. The animal research is interesting, but it's animal research.
One retrospective chart review published in 2021 in Alternative Therapies in Health and Medicine [PMID 34324435] is the most cited piece of human data currently available. Researchers at a private clinic looked at 16 patients who received intra-articular injections of BPC-157 — meaning the compound was injected directly into the knee joint — to address various types of knee pain. Of the 12 patients who received BPC-157 alone without other compounds, 91.6% reported significant improvement. Across the full group of 16, 87.5% reported relief.
Users report meaningful experiences in research contexts, but the authors of that study were explicit: this was a small retrospective chart review, not a randomised controlled trial. They called for future studies. Self-reported outcomes outside a controlled setting are a data point, not a conclusion.
The UC and MS trials referenced in the Frontiers in Pharmacology review indicate BPC-157 has been administered to humans before, and the safety profile didn't produce red flags. But safety observations from early-stage trials are a long way from the efficacy evidence the FDA requires for drug approval.
The WADA Episode — What It Actually Tells You
One part of the BPC-157 story that comes up consistently: the World Anti-Doping Agency added BPC-157 to its prohibited substances list in 2022. The 2025 Pharmaceuticals review [PMID 40005999] documents this. It's also worth noting that BPC-157 is not currently on the WADA prohibited list — it was removed.
What this episode reflects is global regulatory uncertainty rather than settled science. Sports anti-doping bodies tend to prohibit substances ahead of full clinical evidence when they suspect performance-relevant effects. The removal suggests the initial concern didn't hold up under review. Neither the addition nor the removal tells you much about safety or efficacy — what it tells you is that governing bodies are watching, reacting, and revising as the research evolves.
What the Regulatory Picture Means for Researchers
The honest summary of BPC-157's regulatory status in 2026 looks like this:
It is not a scheduled controlled substance under US federal law. It is not prohibited for purchase as a research chemical. It has not passed through FDA clinical trial phases and cannot legally be sold or marketed for human therapeutic use. Compounding pharmacies operate in a regulatory grey zone around BPC-157 that is actively being reviewed by the FDA — with a formal evaluation process underway as of this year.
Members experience ongoing research interest in this compound across multiple biological domains, and the preclinical data has held up across independent research groups over more than a decade. But the clinical gap is real and shouldn't be papered over.
Any engagement with BPC-157 by researchers today is for research purposes only. That framing isn't regulatory boilerplate — it's an accurate description of where the compound sits in the evidence hierarchy. The interesting question isn't whether BPC-157 is "approved" or "banned." It's whether the clinical trial investment will ever materialise, and what drives that timeline.
The 2026 Federal Register evaluation for 503A compounding status is one small indicator that the conversation is moving, not stalling. Whether it moves toward broader access or further restriction depends on the committee process, the submitted evidence, and how the FDA weighs the existing preclinical record against the human data gap.
That process will be worth following.
Key Takeaways
- BPC-157 is not FDA-approved for any therapeutic use — this is confirmed by a 2025 peer-reviewed review that cites the absence of comprehensive human clinical trial data as the reason.
- FDA approval requires completing Phase 1, 2, and 3 human clinical trials. None of these have been completed for BPC-157, primarily because the patent landscape makes it commercially unattractive for pharmaceutical companies to fund the trials.
- BPC-157 is not a scheduled controlled substance in the US. It is legally purchasable as a research chemical.
- The FDA's compounding pharmacy evaluation (503A Bulks List) is actively ongoing for BPC-157 as of 2026 — specifically for a ulcerative colitis use case under committee review.
- Animal model research across tendon, gut, wound healing, and connective tissue domains is consistent and positive. Human data is limited to one small retrospective chart review (n=16) showing 87.5% self-reported improvement in knee pain.
- BPC-157 was temporarily added to the WADA prohibited list in 2022 and subsequently removed — reflecting global regulatory uncertainty rather than a settled safety or efficacy verdict.
- The compound's origin in human gastric juice protein is mechanistically relevant to the gut protection research, where it shows the most plausible biological rationale.
Exploring a research protocol built around recovery and cellular resilience? The RESTORE Protocol is where VERO members start. Explore RESTORE →
RESTORE™
BPC-157Engineered around 500mcg BPC-157. Accelerates deep tissue and joint recovery
Clinical Context
Important Notice: VERO protocols are nutritional and systemic optimisation formats. They are not intended to diagnose, treat, cure, or prevent any medical condition. These statements have not been evaluated by the Food and Drug Administration.

