Choosing a peptide protocol starts with identifying your primary biological goal. This guide maps six research domains to their corresponding compound categories using verified peer-reviewed sources.
The short answer: start with your goal, not the compound. Identify the biological domain you want to research (recovery, skin integrity, metabolic function, cognition, longevity, or sexual wellness) then match it to the compound with the deepest published literature in that domain. This guide maps all six domains to their corresponding research compounds using verified peer-reviewed sources throughout.
This article is produced for research purposes only. The compounds described below are research chemicals with published preclinical literature. Nothing here constitutes medical advice or a treatment recommendation.
What a Peptide Protocol Actually Covers
Let's define the word before anything else. A peptide (a short chain of amino acids, the building blocks your body uses to make proteins) protocol covers four variables: which compound or compounds you're using, how it's being delivered into your body, how often you're administering it, and how long you're running the experiment. Change any one of those variables and you're running a different protocol.
Most people approach this backwards. They read about a specific compound, get interested in it, and then try to fit their goals to that compound's literature. Research suggests the more productive direction is goal-first: define what biological domain you're investigating, then identify which compound has the most relevant and consistent evidence base in that domain.
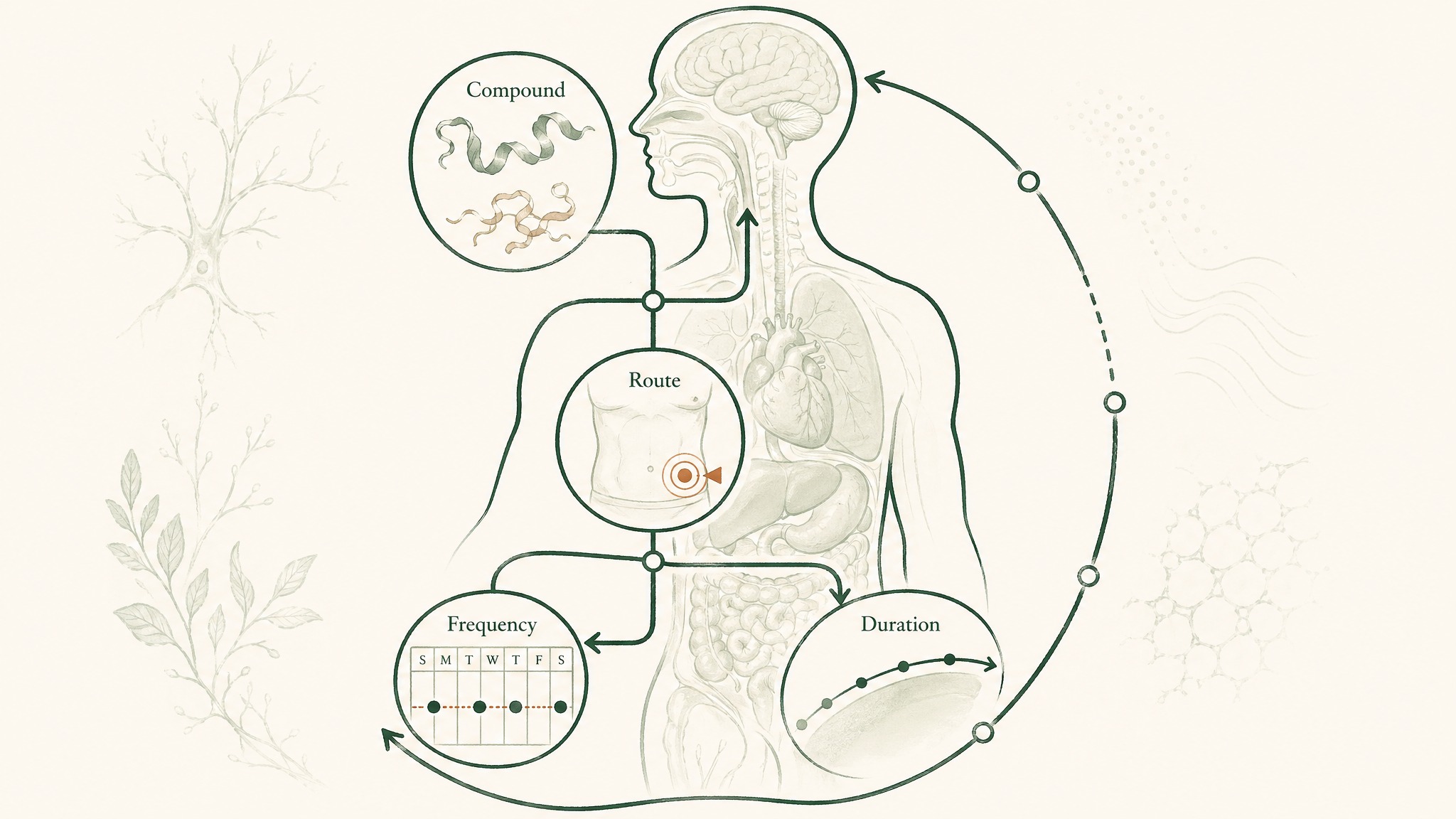
Peptides aren't interchangeable between domains. BPC-157 and Epithalon both surface in longevity discussions, but their mechanisms are entirely different. One targets soft tissue cytoprotection and new blood vessel formation; the other targets how many times your cells can divide before they stop. Treating them as substitutes misses the point of both.
Matching Your Biological Goal to the Research
Six research domains. Six compound categories. All backed by published preclinical literature, each cited below.
Musculoskeletal Recovery and Soft Tissue Support
BPC-157 is a synthetic pentadecapeptide, which means a chain of exactly 15 amino acids. It was derived from sequences found in human gastric juice, where your stomach produces related proteins as part of its protective lining.
A 2019 systematic review in Cell and Tissue Research (Gwyer, Wragg, and Wilson) examined the published BPC-157 research across multiple tissue injury types and found consistently positive outcomes across soft tissue categories, with particular strength in tendons and ligaments [1]. These are tissues that recover slowly because of their limited blood supply. The review was explicit that this evidence base is predominantly rodent model data, and human clinical confirmation does not yet exist.
Thymosin beta-4 (the active peptide fraction found in TB-500 research preparations) occupies the same broad domain through a different mechanism. A 2010 paper in the Annals of the New York Academy of Sciences (Philp and Kleinman) documented its role across animal models in "cell migration, blood vessel formation, cell survival, and stem cell maturation" in dermal, corneal, and cardiac tissues [2]. Where BPC-157 emphasises angiogenesis (building new blood vessels) and growth factor regulation at injury sites, thymosin beta-4 focuses on inflammatory pathway modulation and cellular movement. Different targets, same general domain, which is why they appear together frequently in research community discussions.
Skin Integrity and Connective Tissue Remodelling
GHK-Cu is a tripeptide (three amino acids: glycyl-L-histidyl-L-lysine) bound to copper. Think of it as a signalling molecule that can influence a surprisingly large number of biological processes simultaneously, with skin and connective tissue as its primary research domain.
A 2015 paper in BioMed Research International (Pickart, Vasquez-Soltero, and Margolina) found that GHK is capable of influencing at least 4,000 human genes involved in collagen production, glycosaminoglycan synthesis, immune cell recruitment, and antioxidant activity [3]. Collagen is the structural scaffolding of skin; glycosaminoglycans are what keeps that scaffolding hydrated and dense. GHK-Cu affects both simultaneously through multiple overlapping pathways, which contributes to both the breadth of its literature and the complexity of interpreting individual research outcomes.
Members experience gradual changes in skin texture and connective tissue density across multi-week protocols. Individual variation is significant, and results are context-dependent.
Metabolic Support and Body Composition
MOTS-c is unlike anything else in this list. It's a 16-amino-acid peptide encoded not in your cell's nucleus where most genetic information lives, but in your mitochondrial DNA (the separate genetic material inside your cells' power plants). That single structural fact separates it mechanistically from every other compound in this guide.
A landmark 2015 paper in Cell Metabolism (Lee et al.) found that MOTS-c administration in mouse models significantly reduced age-dependent and diet-induced insulin resistance and excess adiposity, acting through AMPK activation via the folate cycle and purine biosynthesis pathways [4]. AMPK functions as a master switch for cellular energy regulation. Research suggests MOTS-c works as a mitochondria-to-nucleus messenger, with the cells' power plants signalling to the genetic control room to adjust how the whole body handles fuel.
This mechanism is distinct from peptides that influence metabolic outcomes through growth hormone secretagogue pathways. The two categories aren't redundant.
Cognitive Performance and Neuroprotection
Semax is a synthetic seven-amino-acid peptide built from a fragment of ACTH, a signalling hormone your pituitary gland produces naturally. Its cognitive research centres on BDNF (brain-derived neurotrophic factor), which you can think of as fertiliser for brain cells. BDNF supports the growth and maintenance of the connections between neurons, the cells that carry information throughout your brain.
A 2006 paper in Brain Research (Dolotov et al.) found that a single administration of Semax measurably increased BDNF protein levels and trkB phosphorylation in rat hippocampal tissue, alongside improved conditioned learning performance in the same animals [5]. The hippocampus is the brain region most strongly associated with learning and memory formation. trkB is the receptor that BDNF binds to; its phosphorylation is the molecular event that activates the pathway.
Semax is typically administered intranasally because the tissue behind your nose, called the olfactory mucosa, gives compounds relatively direct access to brain tissue, bypassing most of the barriers that slow other delivery routes. Users report that cognitive effects from Semax tend to appear faster than most other peptides, likely because of this delivery route.
Sexual Wellness Research
Bremelanotide (commonly known as PT-141) is the one compound on this list with a completed FDA approval pathway. It was approved under the brand name Vyleesi for hypoactive sexual desire disorder (HSDD), a clinical diagnosis characterising significantly reduced sexual desire in premenopausal women, in June 2019.
A review article in Drugs (Dhillon and Keam, 2019) documents its mechanism as a "synthetic peptide analogue of the neuropeptide hormone alpha melanocyte-stimulating hormone (α-MSH)" acting at the melanocortin type 4 receptor to influence brain pathways involved in sexual response [6]. The key research distinction: bremelanotide acts centrally, through the brain and hypothalamus, rather than peripherally through vascular mechanisms. That's a fundamentally different research context from vasodilatory approaches, with different expected onset timelines and different protocol considerations.
Cellular Longevity and Telomere Biology
Epithalon is a synthetic four-amino-acid peptide originally developed at the St. Petersburg Institute of Bioregulation and Gerontology. Its research targets telomeres, the protective caps at the ends of your chromosomes. Picture the plastic tips on shoelaces: without them, the shoelace frays. Telomeres do the same job for chromosomes. Every time a cell divides, the telomeres get a little shorter. When they get short enough, the cell can no longer divide, a limit called the Hayflick limit.
A 2004 paper in the Bulletin of Experimental Biology and Medicine (Khavinson et al.) found that applying Epithalon to aging human fetal fibroblasts produced telomere elongation to lengths comparable to those in early-passage (young) cells, enabling roughly 10 additional cell divisions beyond the cells' normal limit [7]. Telomerase activation (what Epithalon appears to stimulate in this model) is an active research area in cellular longevity science.
Epithalon's literature is smaller than BPC-157 or GHK-Cu, with a significant portion in Russian-language sources. Its mechanism is entirely distinct from every other compound in this guide, which makes it non-redundant in a longevity-focused protocol.
Why Delivery Format Is Part of the Decision
Picking the right compound is only half the decision. How that compound gets into your bloodstream determines whether it arrives in usable form at all.
Here's the core problem with oral peptide administration: your digestive system treats peptides like food. Enzymes in your gut and stomach break down protein chains (which is what a peptide is) before they can be absorbed in meaningful quantities. Oral bioavailability for most short-chain peptides is below 2%. Out of every 100 milligrams in a capsule, 98 or more get broken down before reaching your bloodstream.
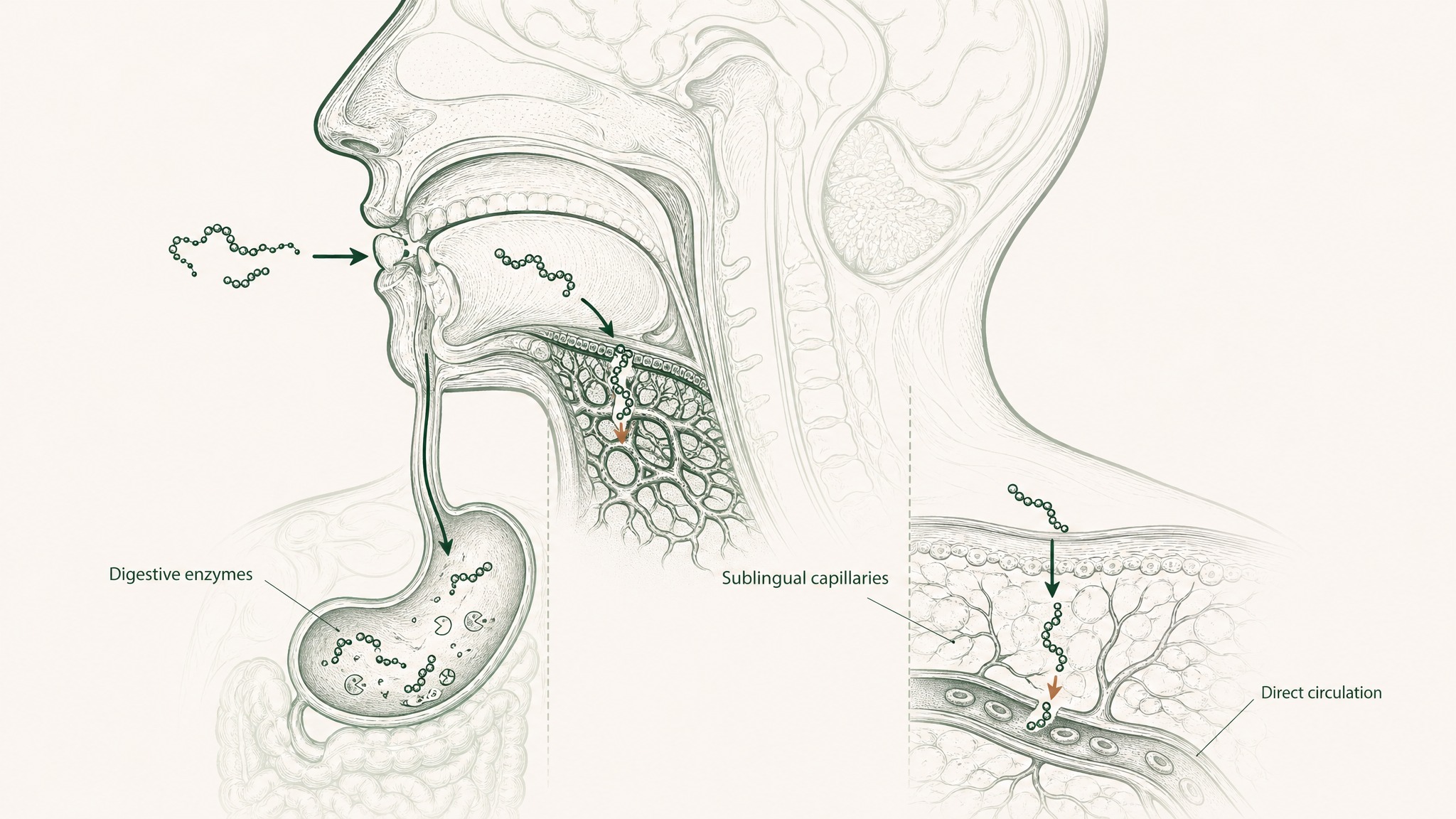
A 2022 review in the Journal of Controlled Release (Rawas-Qalaji, Thu, and Hussain) examined the challenges of delivering macromolecules (large biological molecules including peptides) across mucosal surfaces and confirmed that newer formulation strategies — penetration enhancers, mucoadhesive materials, and hybrid nanosystems — can substantially improve absorption compared to conventional oral delivery [8].
Sublingual delivery takes advantage of different anatomy. The tissue under your tongue is thin and densely supplied with blood vessels, so compounds can pass directly into systemic circulation without being processed by digestive enzymes or filtered by your liver on the first pass. VERO's VERISORB sublingual technology was developed specifically to address the enzymatic degradation barrier at the mucosal surface, using a formulation approach designed for this absorption mechanism.
Injectable administration eliminates all mucosal barriers entirely and provides the most predictable pharmacokinetics, the technical term for how a compound moves through and exits your body over time. It's the gold standard for getting a compound into circulation intact. A well-formulated sublingual system closes a significant part of that gap without the practical barrier to entry.
Not every compound is format-flexible. Semax has an intranasal delivery mechanism in its clinical literature; the access to brain tissue via the olfactory pathway is part of how it produces its effects in cognitive research. That route can't be replicated through sublingual or oral delivery.
Stacking: When One Compound Isn't Enough
Some research goals benefit from combining compounds with non-overlapping mechanisms rather than relying on a single compound to cover the whole biological domain.
The most examined combination in published animal research is BPC-157 paired with thymosin beta-4. BPC-157 literature emphasises angiogenesis and growth factor regulation at sites of tissue stress; thymosin beta-4 literature emphasises actin sequestration (how cells reorganise their structural proteins to facilitate movement), inflammatory modulation, and cell migration. The two target different parts of the same recovery domain. They're complementary, not redundant.
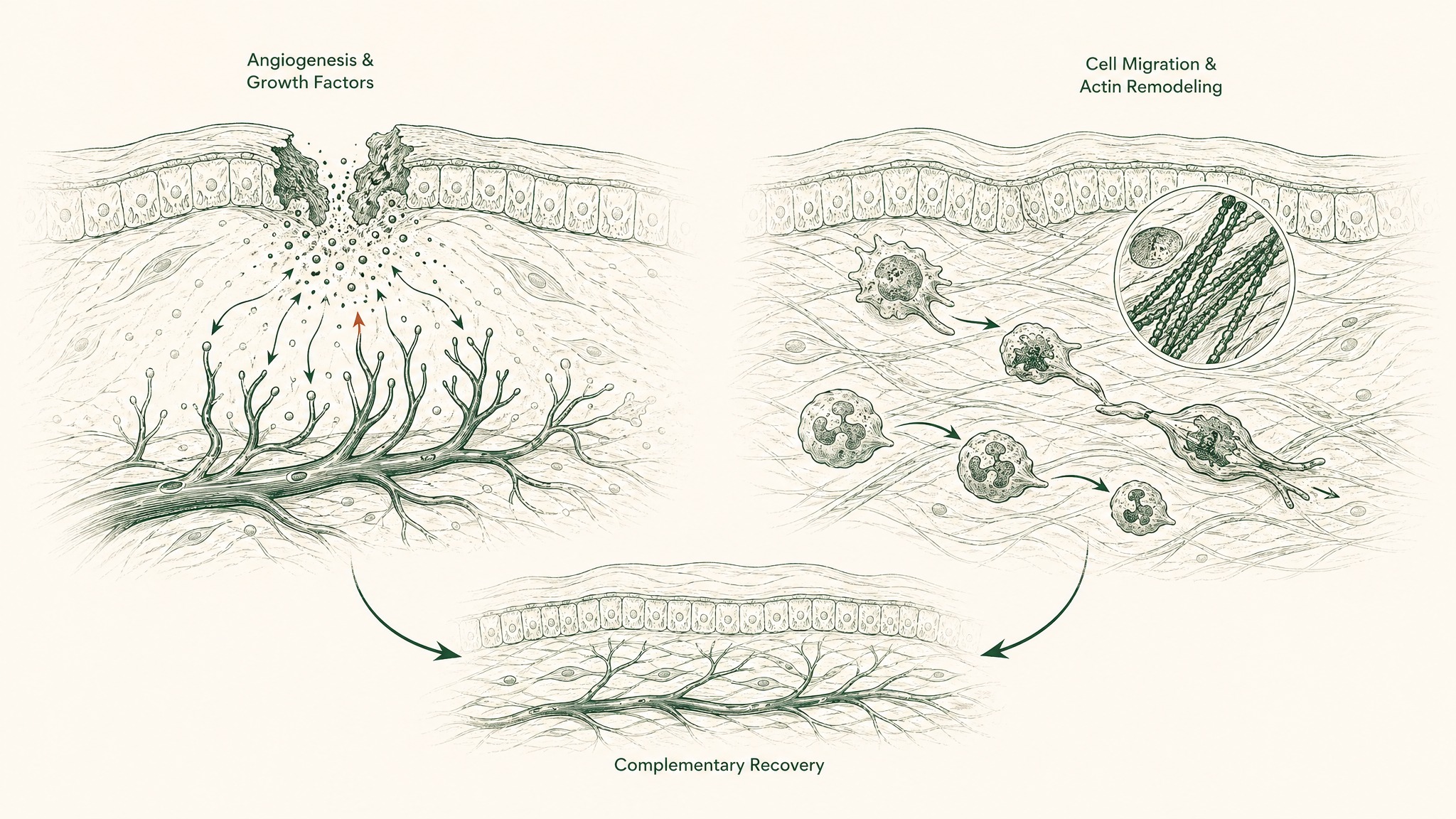
For longevity-oriented research, GHK-Cu and Epithalon represent a pairing with no mechanism overlap: GHK-Cu acts on extracellular matrix composition and broad gene expression; Epithalon acts on telomerase activity in dividing cells. Neither replaces the other.
The practical discipline for any stack is sequential introduction. Add one compound, establish a clear baseline response, then add a second. When two compounds go in at the same time, it's very difficult to attribute what you're observing to either one specifically. Members experience significantly better signal clarity from sequential introduction, even when it takes longer to get there.
Stacking doesn't multiply outcomes. It addresses non-overlapping mechanisms. The ceiling is set by biology.
Protocol Duration, Cycling, and Tracking
Published preclinical studies use highly variable timeframes depending on compound and endpoint. BPC-157 research typically uses two-to-four week intervention windows in acute tissue injury models. Epithalon aging studies use longer windows. Neither timeframe automatically translates to an optimal research duration for human protocols.
In community practice, most clear signals become measurable within two to six weeks of consistent administration. Members experience the best signal-to-noise ratio when the tracking metric was defined before the protocol started, not retrospectively.
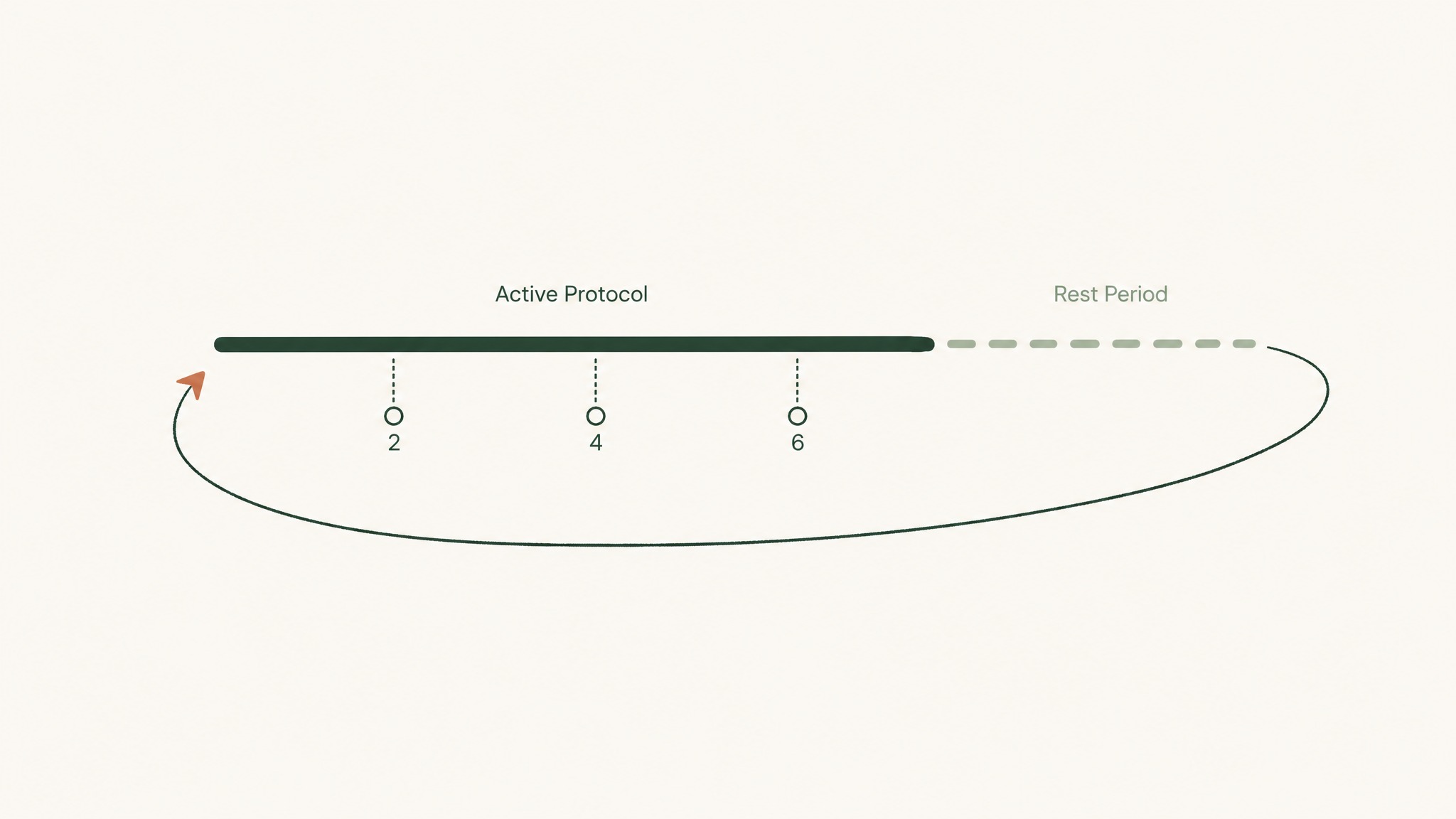
What that looks like in practice: consistent-lighting skin photography for GHK-Cu research. Specific recovery time benchmarks for BPC-157. Validated cognitive assessments for Semax. If you can't measure it before the protocol, you can't interpret what happens during it.
Cycling (periods of administration followed by off periods) appears consistently across research community practice. Most frameworks use four-to-twelve week administration cycles with one-to-four week breaks between them. Human clinical data on optimal cycling intervals is limited; the rationale varies by practitioner, but the structure is consistent.
Frequently Asked Questions
Can I run more than one peptide compound at the same time?
Compounds with non-overlapping mechanisms can be studied together, and some published animal research does exactly that. The practical recommendation from research community members is sequential introduction rather than simultaneous. One compound first, establish your baseline response, then add a second. Running two compounds from day one makes it very difficult to attribute any signal you observe to either one specifically.
How do VERO's protocols map to this goal framework?
VERO's protocol range was designed around the goal-first framework in this article. RESTORE maps to musculoskeletal recovery and soft tissue support. RADIANCE maps to skin integrity and connective tissue. SHIFT maps to metabolic support and body composition. CLARITY maps to cognitive performance and neuroprotection. All four use VERISORB sublingual delivery to address the bioavailability barrier described above.
What if my research goal spans more than one domain?
Start with your highest-priority goal and identify the compound with the deepest relevant literature for that domain. Secondary goals can be addressed with additional compounds after you've established a clear baseline response to the first. Trying to address every goal simultaneously produces noisier data and makes it harder to identify what's actually working.
Key Takeaways
- Start with goal clarity. Research compounds are domain-specific, not general-purpose tools.
- Recovery and soft tissue research maps to BPC-157 [1] and thymosin beta-4 [2]; skin and connective tissue to GHK-Cu [3]; metabolic to MOTS-c [4]; cognitive to Semax [5]; sexual wellness to bremelanotide [6]; cellular longevity and telomere biology to Epithalon [7].
- Delivery format determines whether a compound arrives bioavailable. Sublingual and injectable routes bypass the enzymatic degradation that makes oral peptide administration largely ineffective [8].
- Stack compounds with non-overlapping mechanisms, introduced sequentially (not simultaneously) to preserve interpretability.
- Define your tracking metric before the protocol starts. Retrospective measurement produces noise.
- VERO's protocol range maps directly to this framework: RESTORE, RADIANCE, SHIFT, and CLARITY.
This content is produced for educational and research purposes only. VERO products are not intended to diagnose, treat, cure, or prevent any disease. Compounds described are research chemicals and are not approved for human therapeutic use unless otherwise stated. Consult a qualified healthcare provider before making any health-related decisions.
Explore VERO's full research protocol range. View all protocols →
References
Gwyer D, Wragg NM, Wilson SL. Gastric pentadecapeptide body protection compound BPC 157 and its role in accelerating musculoskeletal soft tissue healing. Cell Tissue Res. 2019;377(2):153–159. PMID: 30915550.
Philp D, Kleinman HK. Animal studies with thymosin beta, a multifunctional tissue repair and regeneration peptide. Ann N Y Acad Sci. 2010;1194:81–86. PMID: 20536453.
Pickart L, Vasquez-Soltero JM, Margolina A. GHK peptide as a natural modulator of multiple cellular pathways in skin regeneration. Biomed Res Int. 2015;2015:648108. PMID: 26236730.
Lee C, Zeng J, Drew BG, et al. The mitochondrial-derived peptide MOTS-c promotes metabolic homeostasis and reduces obesity and insulin resistance. Cell Metab. 2015;21(3):443–454. PMID: 25738459.
Dolotov OV, Karpenko EA, Inozemtseva LS, et al. Semax, an analog of ACTH(4-10) with cognitive effects, regulates BDNF and trkB expression in the rat hippocampus. Brain Res. 2006;1117(1):54–60. PMID: 16996037.
Dhillon S, Keam SJ. Bremelanotide: first approval. Drugs. 2019;79(14):1599–1606. PMID: 31429064.
Khavinson VKh, Bondarev IE, Butyugov AA, Smirnova TD. Peptide promotes overcoming of the division limit in human somatic cell. Bull Exp Biol Med. 2004;137(5):503–506. PMID: 15455129.
Rawas-Qalaji M, Thu HE, Hussain Z. Oromucosal delivery of macromolecules: challenges and recent developments to improve bioavailability. J Control Release. 2022;352:726–746. PMID: 36334858.
RESTORE™
BPC-157Engineered around 500mcg BPC-157. Accelerates deep tissue and joint recovery
Clinical Context
Important Notice: VERO protocols are nutritional and systemic optimisation formats. They are not intended to diagnose, treat, cure, or prevent any medical condition. These statements have not been evaluated by the Food and Drug Administration.

