Your cells run a maintenance system that slows down as you get older — not because ageing is inevitable, but because a key fuel supply runs dry. Here's what the NAD+–sirtuin cascade actually is, and what the research shows.
NAD+ and sirtuins are two parts of the same biological maintenance loop. NAD+ (nicotinamide adenine dinucleotide) is a molecule your cells need to power repair and energy work. Sirtuins are a family of proteins that carry out that repair, but only when NAD+ is available. Both decline measurably as you age, and research suggests this combined drop sits at the centre of how cells accumulate damage over decades. That's the longevity cascade: one molecule fuels the other, and when the first runs short, the whole system quietens.
What NAD+ Actually Does in Your Cells
Here's the simplest way to picture it: your cells are running two completely separate jobs at once, and NAD+ shows up for both of them.
The first job is energy production. When your body converts food into usable fuel, NAD+ acts as a shuttle, picking up electrons from one reaction and dropping them off at another. Without that shuttle, the whole energy chain grinds down. The second job is signalling. NAD+ gets consumed (not just borrowed) by a class of proteins called sirtuins and by enzymes called PARPs (which patch damaged DNA), and that consumption triggers repair and maintenance work across your genome.
Researchers at Harvard's Sinclair lab describe NAD+ as controlling "hundreds of key processes from energy metabolism to cell survival" — it's not a single-use molecule (Rajman, Chwalek & Sinclair, Cell Metab 2018, PMID 29514064). It fluctuates based on food intake, exercise, and even the time of day. And it declines with age in ways that now have a substantial body of research behind them (Verdin, Science 2015, PMID 26785480).
Meet the Sirtuins: Your Cellular Maintenance Crew
Think of sirtuins as a quality-control team stationed throughout your cell. Your body makes seven of them (SIRT1 through SIRT7), each sitting in a different compartment: the cell nucleus, the cytoplasm (the fluid inside the cell), and the mitochondria (the energy-generating structures inside every cell).
Their job is deacylation (a complicated word for a simple idea). Proteins in your cells collect chemical tags over time. Some tags are instructions; others are errors that build up like sticky notes left on the wrong file. Sirtuins strip those tags off, resetting proteins back toward their normal function. This has downstream effects on DNA repair, inflammation signalling, and how efficiently your mitochondria generate energy.
The catch is that sirtuins can't do any of this without NAD+. Every time a sirtuin does its job, it consumes one molecule of NAD+ in the process. No fuel, no maintenance. This is why sirtuin activity in tissues drops alongside NAD+ as both decline with age (Imai & Guarente, Trends Cell Biol 2014, PMID 24786309).
The NAD+–Sirtuin Loop: How One Powers the Other
To understand why this matters, you need to see how tightly the two are connected.
Your body doesn't just absorb NAD+ directly from food. It has to build it from raw materials via an enzyme called NAMPT (nicotinamide phosphoribosyltransferase). Think of NAMPT as the factory that makes the fuel. That fuel, NAD+, then powers the quality-control team (sirtuins). The sirtuins, when active, regulate the factory's output by influencing gene expression upstream. It's a loop: NAD+ enables sirtuins, sirtuins support the systems that keep NAD+ production running.
When that loop is intact, you get something like cellular coordination: energy production, DNA repair, and inflammation control all talking to each other. A 2013 Cell study from David Sinclair's lab at Harvard demonstrated this connection directly: when researchers raised NAD+ levels in old mice, mitochondrial function was "restored to that of a young mouse in a SIRT1-dependent manner" — meaning the restoration required sirtuin activity to work (Gomes et al., Cell 2013, PMID 24360282). They also showed that deleting SIRT1 accelerated the age-related decline, confirming that sirtuin activity is the mechanism, not just a bystander.
What Happens When NAD+ Falls With Age
This is where it gets concrete. Researchers at the Imai lab have described declining NAD+ as potentially "an Achilles' heel, causing defects in nuclear and mitochondrial functions" as you age (Imai & Guarente, Trends Cell Biol 2014, PMID 24786309). The wording is careful, but the implication is significant: if NAD+ is the rate-limiting factor in sirtuin activity, and sirtuin activity governs cellular maintenance, then NAD+ decline doesn't just slow your energy production. It slows your cells' ability to keep themselves in order.
The Gomes 2013 study showed one specific consequence of this. When NAD+ drops, a protein called HIF-1α, normally a sensor that responds to low oxygen, starts accumulating even when oxygen is plentiful. This creates a false emergency signal in the mitochondria, a "pseudohypoxic state" that disrupts the communication between the cell nucleus and the mitochondria. The mitochondria start acting like they're starved of oxygen when they aren't. The result is deteriorating energy output and accelerating cellular dysfunction.
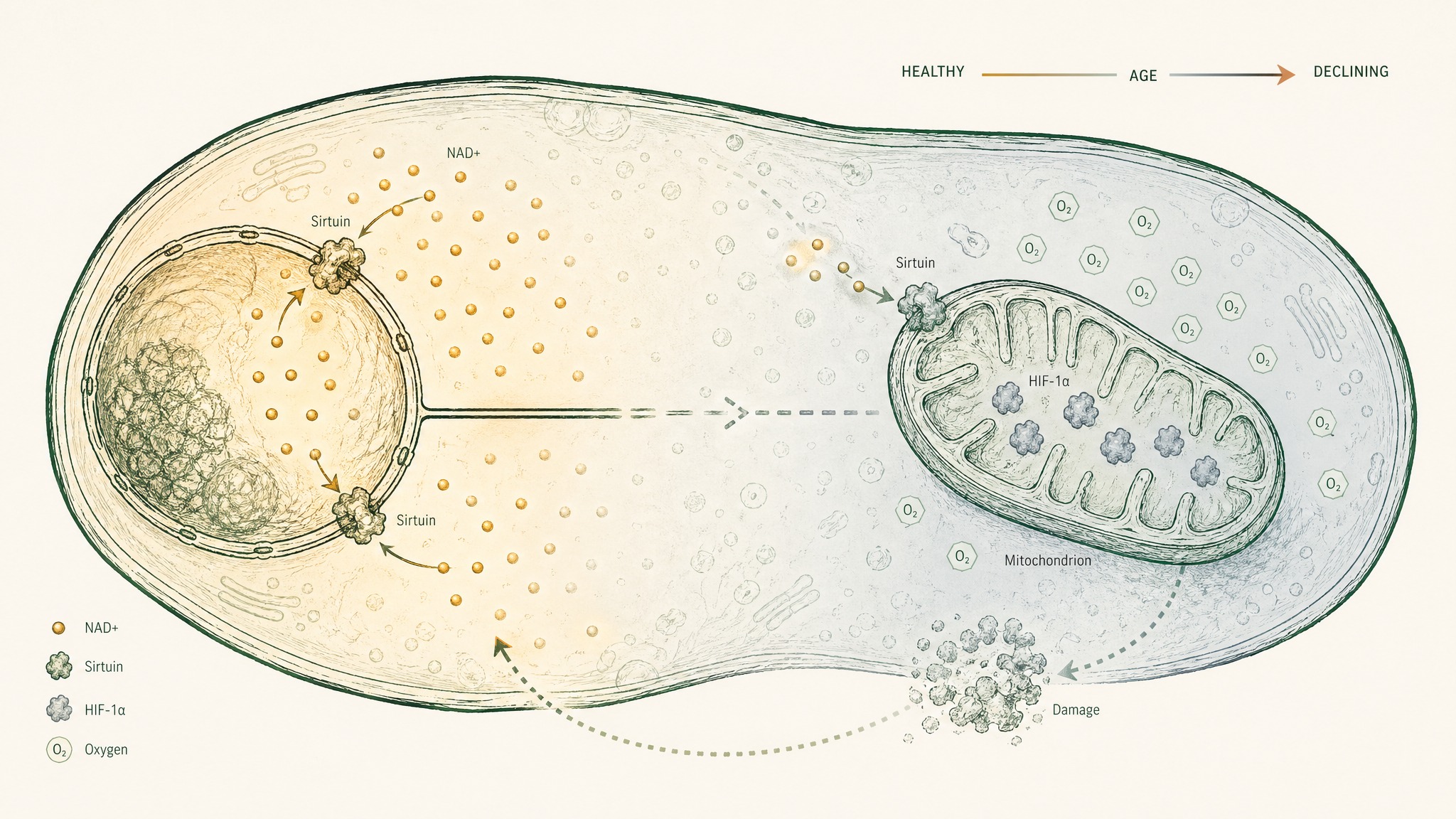
This isn't a gradual slide. It's a cascade. The NAMPT enzyme that builds NAD+ slows down with age. Less NAD+ means less sirtuin activity. Less sirtuin activity means more accumulated cellular damage. More damage means less efficient mitochondria. The loop runs in reverse.
NMN and NR: The Building Blocks Researchers Are Studying
Since raising NAD+ directly is impractical (NAD+ doesn't survive digestion intact), researchers turned to its immediate precursors: NMN (nicotinamide mononucleotide) and NR (nicotinamide riboside). These are the building blocks your body uses to assemble NAD+ from scratch.
NMN is one step away from NAD+ in the biosynthetic chain. NR is two steps away: it first converts to NMN via enzymes called NR kinases, and then to NAD+ via a separate class of enzymes called NMNATs. Both routes arrive at the same destination, but through different pathways in different tissues (Yoshino, Baur & Imai, Cell Metab 2018, PMID 29249689).
This is for research purposes only. Neither NMN nor NR is approved as a pharmaceutical agent for treating any condition. What the research is investigating is whether supplying these precursors in sufficient quantities can meaningfully restore NAD+ concentrations in tissues where it's fallen, and whether that restoration produces the downstream effects the animal data suggests.
What the Animal Research Found
The animal data is where the mechanism gets tested at scale. A 2016 Cell Metabolism study gave mice NMN daily for 12 months (a substantial portion of a mouse's lifespan) and measured outcomes across multiple systems (Mills et al., Cell Metab 2016, PMID 28068222). The results covered several distinct areas:
Metabolic function. Treated mice showed suppressed age-related weight gain, improved energy metabolism, better insulin sensitivity, and improved blood lipid profiles, without any changes to food or water consumption.
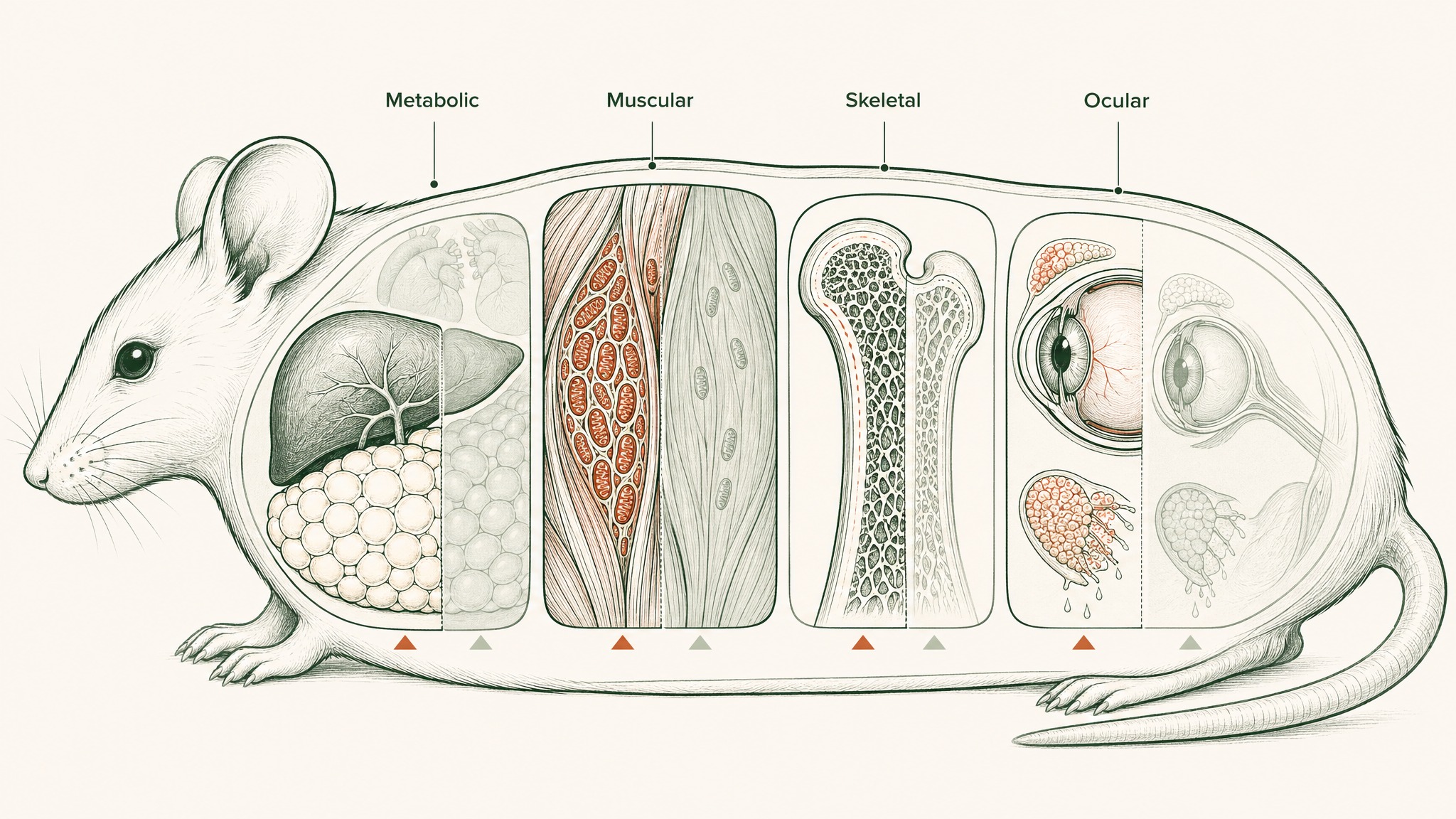
Physical function. Skeletal muscle showed enhanced mitochondrial oxidative metabolism, and physical activity levels increased during the mice's active periods.
Structural health. Bone mineral density increased. Eye function and tear production improved significantly compared with controls.
Gene expression. The researchers observed that NMN "prevented age-associated gene expression changes in key metabolic organs," with approximately 70% of ageing-related genetic changes reversed in muscle and fat tissues.
None of this came with observable toxicity across the 12-month window. A separate 2018 review by Rajman, Chwalek, and Sinclair summarised the broader in vivo evidence: "restoration of NAD+ levels in old or diseased animals can promote health and extend lifespan" across multiple models and disease contexts (Rajman, Chwalek & Sinclair, Cell Metab 2018, PMID 29514064). The consistency across different animal studies is what moved the field toward human trials.
What the Human Research Shows So Far
Human data on NMN is more recent and smaller in scale than the animal literature, but it's accumulating. Members experience and interpret these results differently depending on their baseline health, but the clinical signals point in consistent directions.
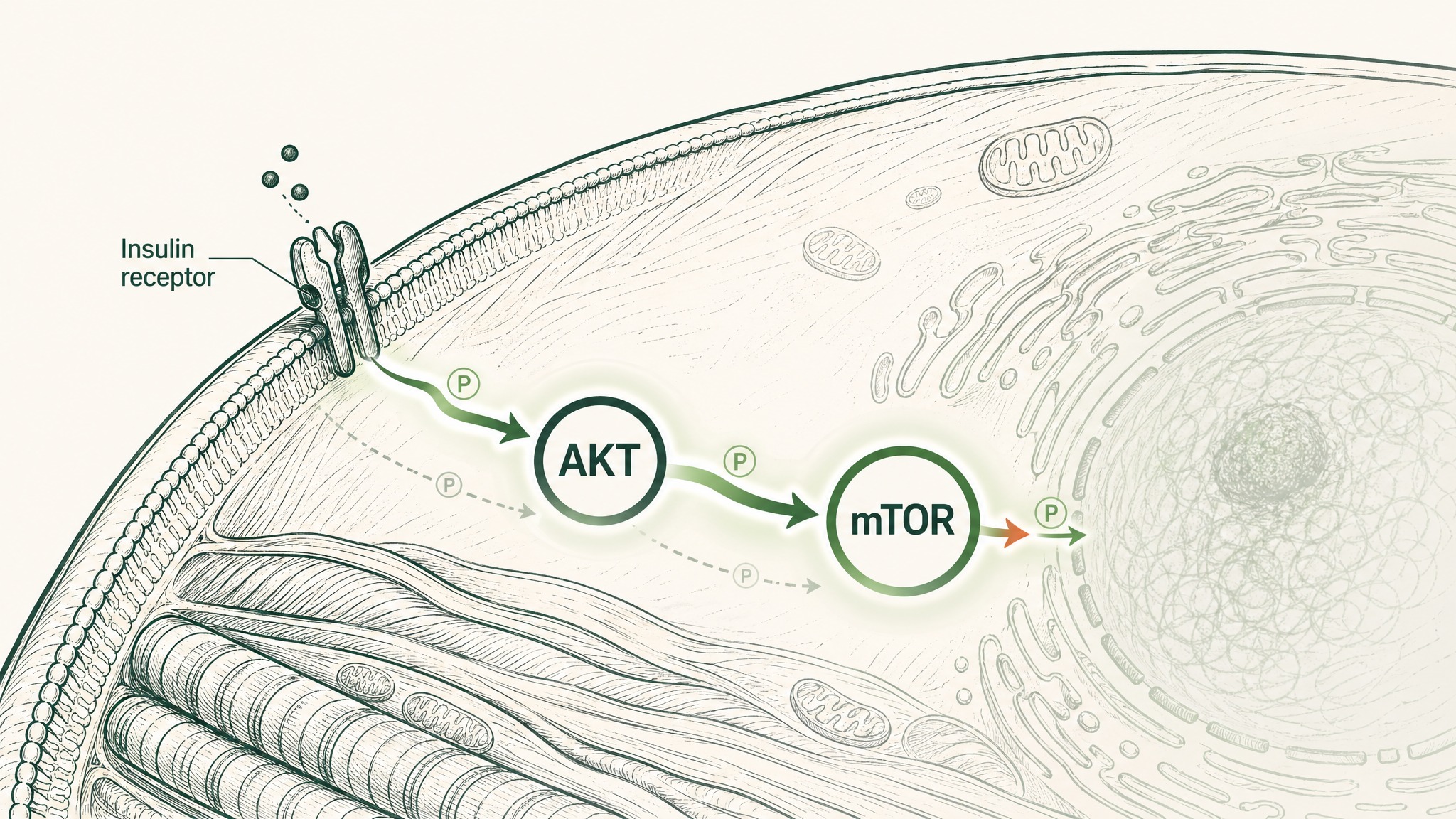
A 2021 Science paper from Washington University School of Medicine ran a 10-week randomised, placebo-controlled trial in postmenopausal women with prediabetes. The primary finding: NMN supplementation improved skeletal muscle insulin sensitivity, measured via a hyperinsulinemic-euglycemic clamp (a precise method for assessing how well cells respond to insulin). The mechanism appeared to be increased phosphorylation of AKT and mTOR proteins, which are key nodes in the insulin signalling pathway inside muscle cells (Yoshino et al., Science 2021, PMID 33888596).
A 2023 GeroScience trial took a different approach: 80 healthy middle-aged adults, randomised to placebo, 300 mg, 600 mg, or 900 mg NMN daily for 60 days. Blood NAD concentrations increased significantly in all treatment groups versus both placebo and baseline at day 30 and day 60. On physical performance, walking distance during a six-minute test improved across all treated groups, with the largest gains at 600–900 mg. On biological age (assessed using epigenetic markers), the placebo group's measured biological age increased over the 60 days, while the treated groups remained stable. The 600 mg dose produced the best overall balance of outcomes, and no safety issues emerged throughout (Yi et al., GeroScience 2023, PMID 36482258).
Users report a range of subjective changes (energy, recovery, sleep quality), but the controlled trial data focuses on measurable metabolic and molecular markers rather than subjective experience, and those distinctions matter when reading the evidence.
In 2024, Leonard Guarente, David Sinclair, and Guido Kroemer reviewed the landscape of human anti-ageing trials in Cell Metabolism, identifying NAD+ precursors (including NMN) among eight of the most promising compounds now entering clinical evaluation, tested against age-associated diseases including diabetes, cardiovascular conditions, and neurodegeneration (Guarente, Sinclair & Kroemer, Cell Metab 2024, PMID 38181790).
The Bigger Picture: NAD+ as Cellular Infrastructure
What makes the NAD+–sirtuin axis different from most single-compound research is its scope. Most molecules studied in longevity science hit one target. NAD+ operates as infrastructure: it sits upstream of dozens of downstream effects simultaneously.
When NAD+ is restored and sirtuins become active again, the effects aren't isolated to one tissue or one pathway. They span mitochondrial function, DNA damage response, inflammation regulation, and metabolic efficiency at the same time. That's what a "longevity cascade" means in practice: one upstream change produces coordinated effects downstream across multiple biological systems simultaneously, because all of those systems were depending on the same upstream fuel.
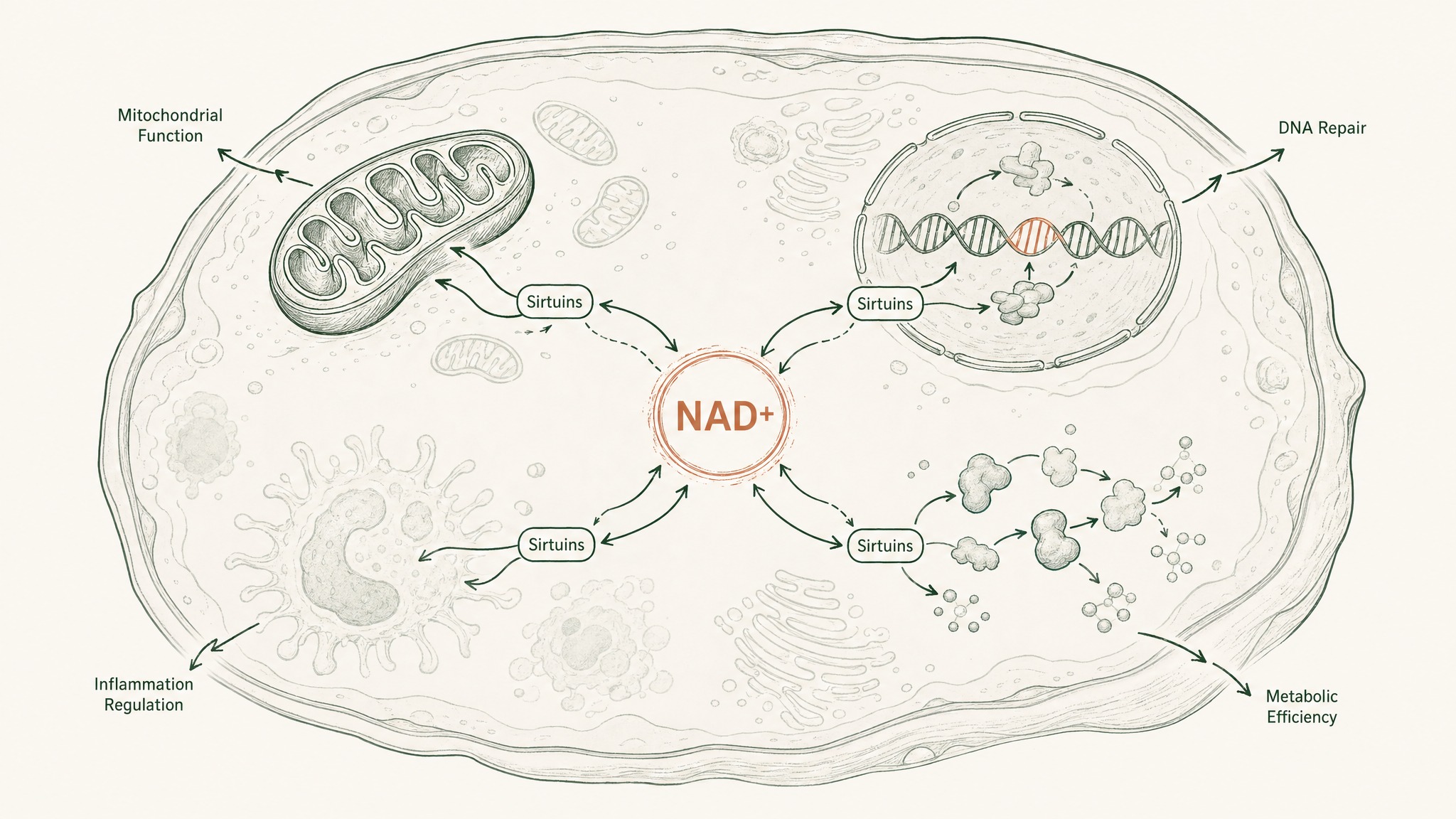
The Verdin 2015 Science review notes that modulating NAD+ production or usage can affect both healthspan (how long you stay healthy) and lifespan across organisms, with particular implications for neurodegeneration where NAD+ decline appears especially significant (Verdin, Science 2015, PMID 26785480). The cardiovascular and metabolic dimensions appear separately in the Rajman 2018 review: the same molecule, different disease contexts, consistent direction of effect.
This is an evolving field. The human trials are still early. The animal data is compelling but doesn't translate automatically to humans. What the research has established clearly is the mechanism: NAD+ falls with age, sirtuin activity falls with it, and restoration of NAD+ in animal models reverses measurable aspects of that decline through a sirtuin-dependent process. The human data is beginning to show whether the same cascade responds in people.
Key Takeaways
- NAD+ is a signalling and energy molecule that declines measurably with age. It's not just for energy production; it's also consumed by sirtuins and DNA-repair enzymes during cellular maintenance work.
- Sirtuins are a family of seven proteins (SIRT1–7) that carry out quality-control work across your cells. They deactivate without NAD+: no fuel, no maintenance.
- The NAD+–sirtuin connection is mechanistically established. A 2013 Harvard study showed that raising NAD+ in old mice restored mitochondrial function to that of young mice, but only when SIRT1 was present (PMID 24360282).
- NMN and NR are the direct precursors used to raise NAD+ in research contexts. NMN is one biosynthetic step from NAD+; NR is two steps, via NR kinases and NMNATs (PMID 29249689).
- Animal studies of long-term NMN showed improvements across metabolic function, bone density, physical activity, and eye health over 12 months without observed toxicity (PMID 28068222).
- Human trials are now producing controlled data. A 2021 RCT showed NMN improved muscle insulin sensitivity in prediabetic women (PMID 33888596). A 2023 trial in healthy middle-aged adults showed increased blood NAD, improved walking performance, and stable biological age at 600 mg daily (PMID 36482258).
- The NAD+–sirtuin cascade is infrastructure, not a single-target fix. Restoring NAD+ activates downstream effects across energy metabolism, DNA repair, inflammation, and mitochondrial function simultaneously, which is why researchers consider it a foundational longevity pathway rather than a niche compound.
Explore VERO's longevity-focused protocols, built around the delivery and bioavailability science that matters most. View protocols →
References
Rajman L, Chwalek K, Sinclair DA. Therapeutic Potential of NAD-Boosting Molecules: The In Vivo Evidence. Cell Metab. 2018;27(3):529–547. PMID 29514064
Verdin E. NAD⁺ in aging, metabolism, and neurodegeneration. Science. 2015;350(6265):1208–1213. PMID 26785480
Imai S, Guarente L. NAD+ and sirtuins in aging and disease. Trends Cell Biol. 2014;24(8):464–471. PMID 24786309
Gomes AP, Price NL, Ling AJY, et al. Declining NAD(+) induces a pseudohypoxic state disrupting nuclear-mitochondrial communication during aging. Cell. 2013;155(7):1624–1638. PMID 24360282
Mills KF, Yoshida S, Stein LR, et al. Long-Term Administration of Nicotinamide Mononucleotide Mitigates Age-Associated Physiological Decline in Mice. Cell Metab. 2016;24(6):795–806. PMID 28068222
Yoshino J, Baur JA, Imai SI. NAD+ Intermediates: The Biology and Therapeutic Potential of NMN and NR. Cell Metab. 2018;27(3):513–528. PMID 29249689
Yoshino M, Yoshino J, Kayser BD, et al. Nicotinamide mononucleotide increases muscle insulin sensitivity in prediabetic women. Science. 2021;372(6547):1224–1229. PMID 33888596
Yi L, Maier AB, Tao R, et al. The efficacy and safety of β-nicotinamide mononucleotide (NMN) supplementation in healthy middle-aged adults: a randomized, multicenter, double-blind, placebo-controlled, parallel-group, dose-dependent clinical trial. GeroScience. 2023;45(1):29–43. PMID 36482258
Guarente L, Sinclair DA, Kroemer G. Human trials exploring anti-aging medicines. Cell Metab. 2024;36(2):354–376. PMID 38181790
RESTORE™
BPC-157Engineered around 500mcg BPC-157. Accelerates deep tissue and joint recovery
Clinical Context
Important Notice: VERO protocols are nutritional and systemic optimisation formats. They are not intended to diagnose, treat, cure, or prevent any medical condition. These statements have not been evaluated by the Food and Drug Administration.

