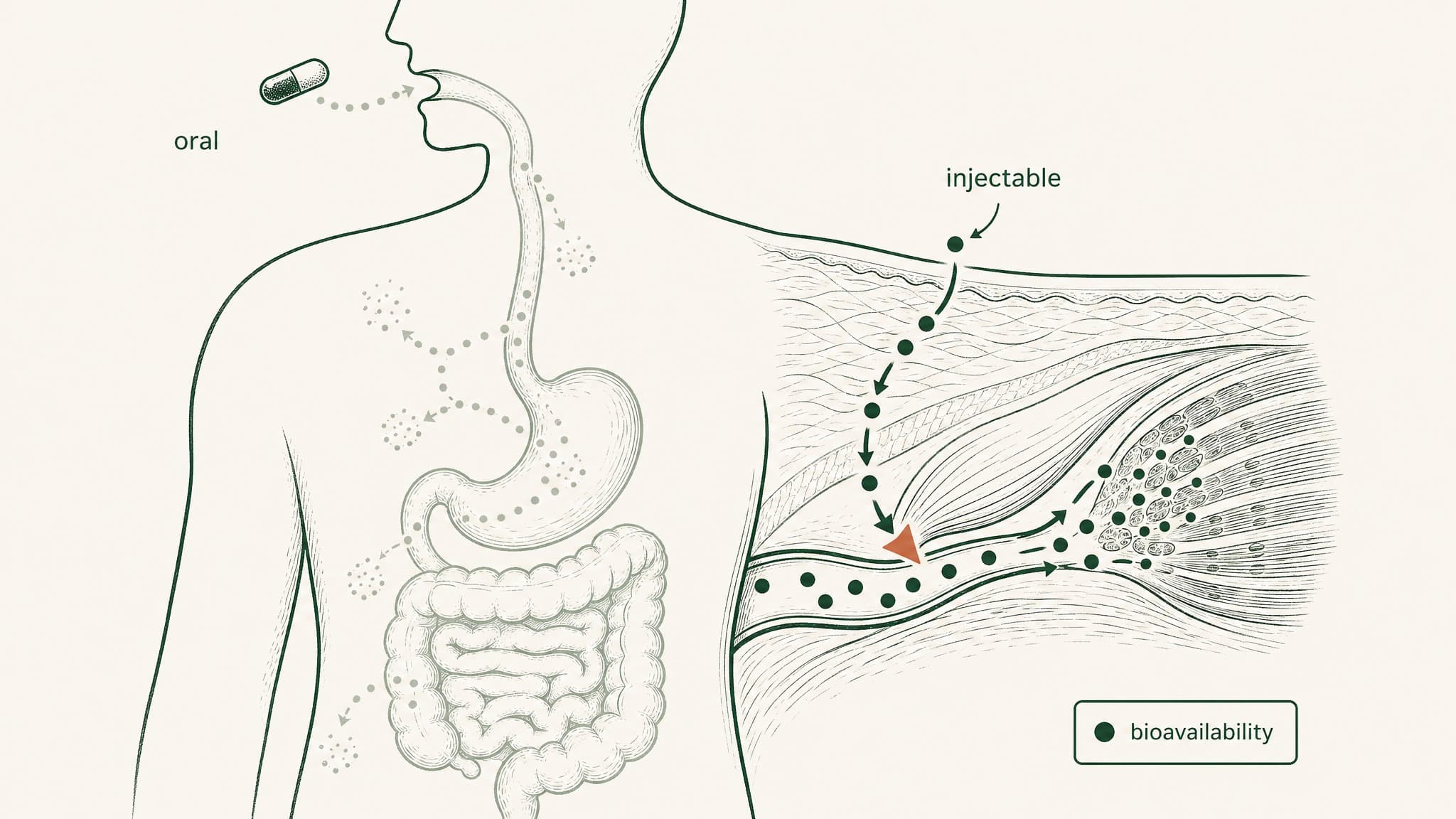
The gap between oral and injectable peptide delivery isn't a marketing claim — it's documented in the research. Here's what the bioavailability numbers actually show across all three routes.
There are three ways to get a peptide into your bloodstream: swallow it, inject it, or absorb it under your tongue. The bioavailability numbers — how much of what you take actually makes it into your blood where it can do something — differ by an order of magnitude across these three routes. This content is published for research purposes only.
Here's the clearest way to think about it. If you take an oral peptide capsule with 1.5% bioavailability, 98.5 milligrams out of every 100 milligrams never reach your bloodstream. They get dismantled in transit. Understanding exactly where each route sits in the documented data — not estimates, not marketing copy — is what makes it possible to design a peptide research protocol with any real precision.
The Oral Bioavailability Floor: What the Data Actually Shows
Your gut is a tiered security system. It wasn't designed to block peptide supplements — it was designed to process food protein. But peptides are chains of amino acids. Food protein is chains of amino acids. The gut doesn't distinguish between the two.
Your stomach operates at pH 1.5 to 3.5 — roughly the acidity of battery acid — and produces an enzyme called pepsin whose entire job is cutting the chemical bonds that hold peptide chains together. Anything that survives the stomach moves to the small intestine, where a second wave of enzymes called brush-border peptidases continues the dismantling at the intestinal wall surface. Below that wall is the epithelial barrier: a single continuous cell layer acting simultaneously as a physical wall and a biochemical checkpoint, complete with intracellular enzymes that process anything attempting to cross. The gaps between epithelial cells are sealed by structures called tight junctions — selective channels that allow small molecules through while blocking most of the larger ones.
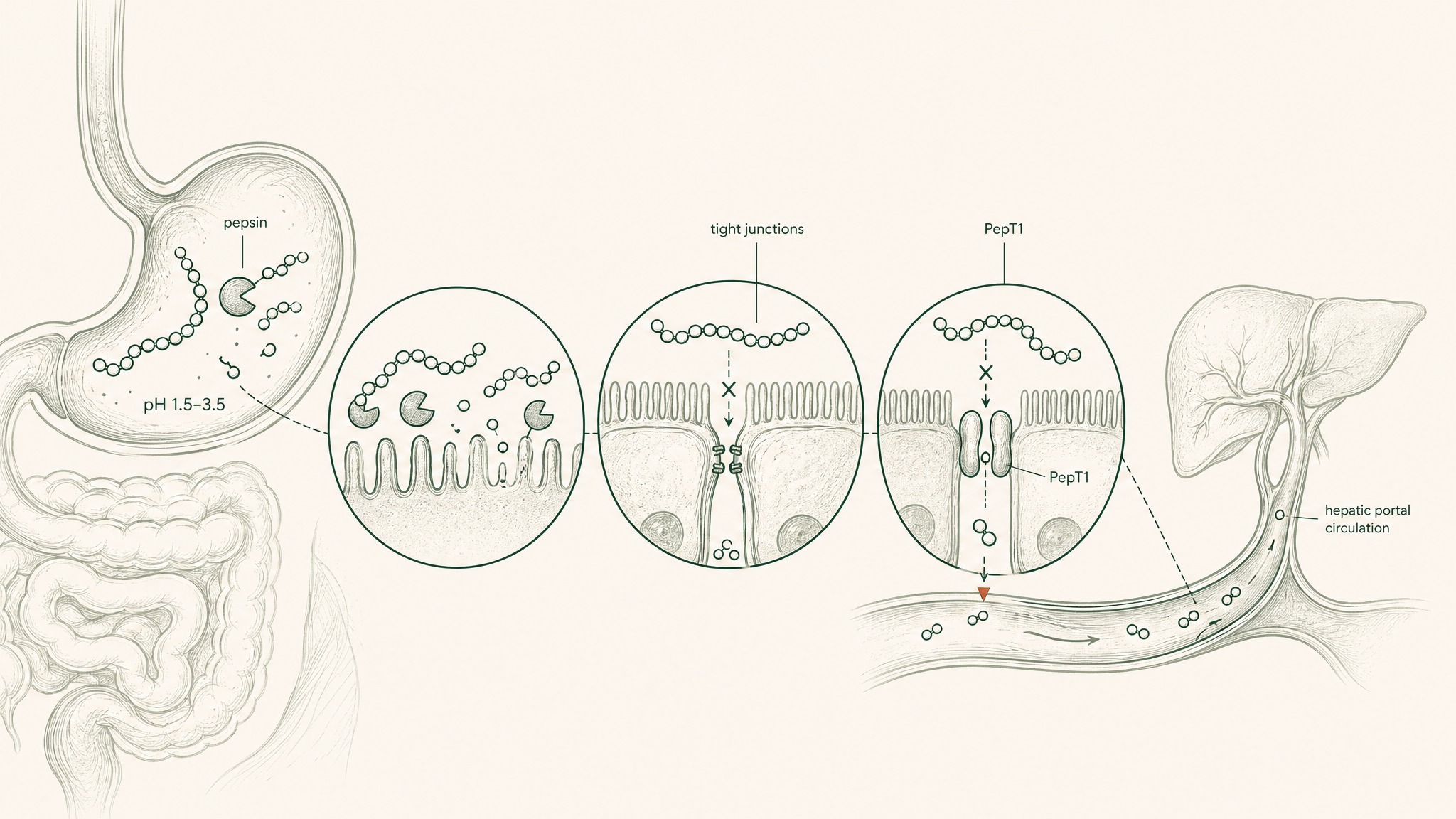
And then there's the final cut. Everything absorbed from the gut goes through the liver before it reaches your general circulation — a stage called first-pass hepatic metabolism (meaning your liver processes the compound and takes its cut before the rest of your body sees it).
The documented output of this sequence: unmodified oral peptides typically achieve bioavailability of less than 1 to 2 percent (Renukuntla et al. 2013, Int J Pharm, PMC3680128). This isn't specific to one compound. It's what the sequential barriers produce consistently across peptide classes. Enzymatic degradation, poor membrane permeability, and first-pass hepatic metabolism don't stack additively — they compound, each operating on what the previous stage left behind (Verma et al. 2021, Drug Dev Res, PMID 33988872).
There is one gut mechanism that does absorb intact peptides: a transporter protein called PepT1 (SLC15A1) that shuttles di-peptides and tri-peptides — fragments of exactly two or three amino acids — across the intestinal wall using a hydrogen-ion co-transport mechanism (Freeman 2015, World J Gastrointest Pharmacol Ther, PMC4419090). PepT1 exists to absorb the output of food digestion. The limitation is substrate window: most research peptides are far longer than three amino acids. BPC-157 is 15 amino acids. The transporter that could theoretically help is built for the wrong chain length. It's a small window in a very tall wall.
What Pharmaceutical Engineering Has Pushed the Oral Ceiling To
If the baseline sits at sub-2%, the next question is what happens when serious resources are directed at closing the gap. The research has a concrete answer, and it's instructive about the scale of what's achievable.
The most commercially significant attempt at oral peptide delivery is semaglutide in tablet form — the compound in Rybelsus, used to manage type 2 diabetes. It's co-formulated with SNAC (sodium N-(8-[2-hydroxybenzoyl]amino) caprylate), a proprietary absorption enhancer that creates a transient localised rise in gastric pH, allowing semaglutide to absorb directly across the stomach wall before it encounters the small intestine's enzymatic environment. The dosing requirements are strict: 30 minutes before any food, no more than 120 millilitres of water.
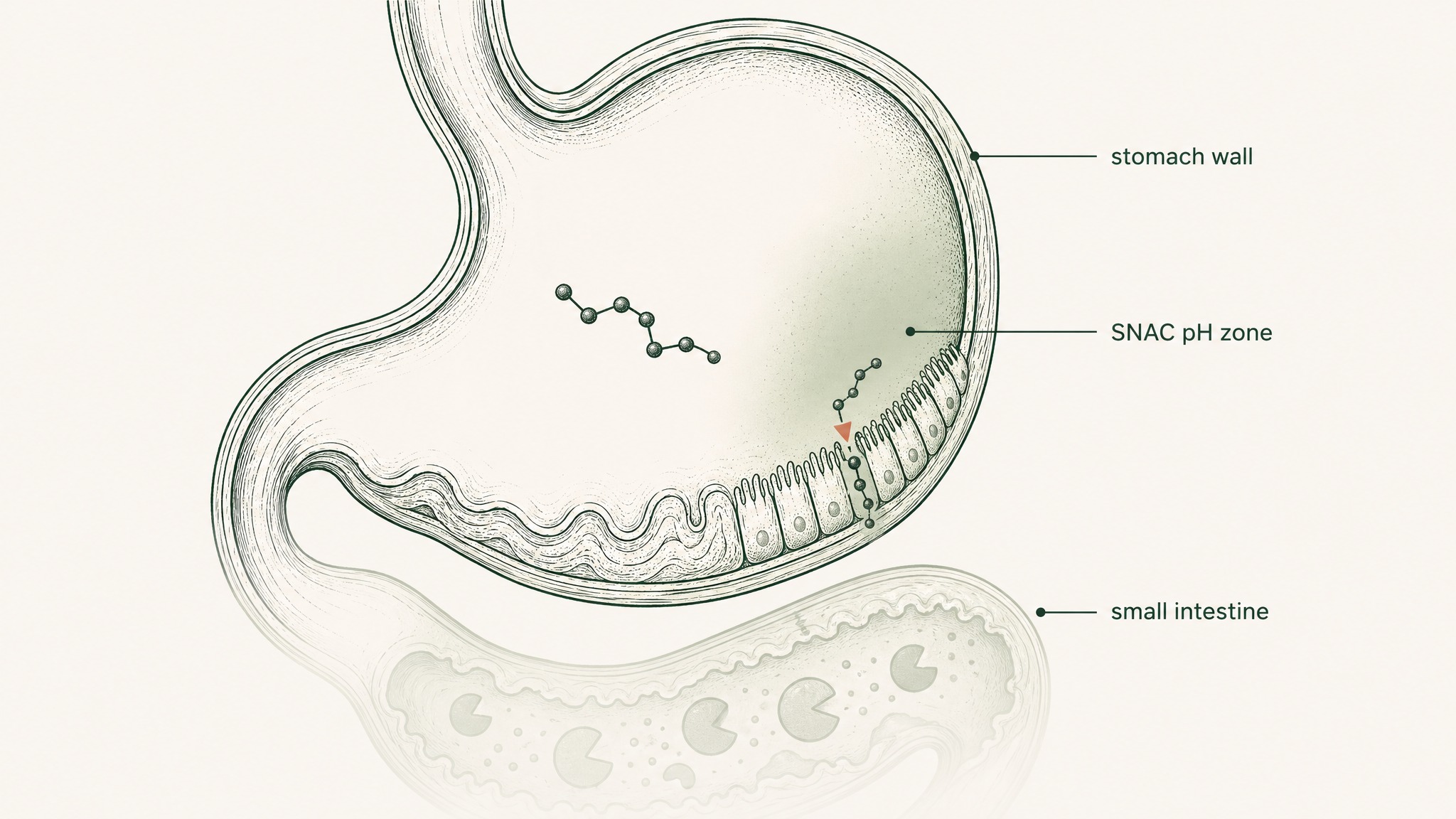
Under those conditions, absolute bioavailability: 0.8 percent (Overgaard et al. 2021, Clin Pharmacokinet, PMID 33969456). Dose-to-dose variability on a single administration runs at around 137 percent. At steady state — after enough daily doses that the long half-life of semaglutide smooths out the variation — that narrows to approximately 33 percent. Semaglutide works at 0.8% because it's pharmacologically potent enough that even that fraction produces a meaningful effect. That characteristic doesn't transfer to most research peptides.
Research-stage liposome technology has pushed individual compounds further. A 2022 review cataloguing liposome-based oral peptide delivery documented approximately 3.4 percent bioavailability for human growth hormone using tetraether-lipid liposomes with bioenhancers, approximately 19 percent for exendin-4 using glycocholic acid-conjugated chitosan-coated liposomes, and approximately 12 percent for insulin using biotin-modified liposomes (Cui et al. 2022, Pharmaceuticals, PMC9501131). These figures represent proprietary formulation approaches engineered for individual compounds. They don't transfer between peptides.
So the documented spectrum for oral delivery runs from under 2 percent for unformulated compounds up to 19 percent for the most advanced research-stage liposome formulations for a specific peptide. That's the real ceiling — not 50 percent, not 80 percent. Nineteen, under optimal research conditions, for one compound. Commercial oral peptide products without SNAC-class absorption engineering don't have access to the upper end of that range.
Why Subcutaneous Injection Remains the Research Reference Standard
Subcutaneous injection — placing a compound directly into the vascularised tissue just beneath your skin — bypasses the gut entirely. No gastric acid. No pepsin. No brush-border peptidases. No epithelial barrier. No tight junctions. The compound deposits into tissue that is already in direct contact with your circulatory system.
That's why injectable formulations remain the dominant delivery formats for therapeutic and research peptides — specifically subcutaneous, intravenous, and intramuscular routes (Zhang et al. 2020, Int J Pharm, PMID 32622810). The preference isn't convention. It's what injection delivers that no oral route currently matches: a predictable relationship between the dose administered and the systemic exposure produced. The dose-to-exposure ratio is reliable because the biology standing between administration and absorption has been reduced to one step, not six.
Intravenous injection is technically superior in speed and completeness — it delivers directly into the bloodstream with immediate systemic distribution. But it requires IV access and clinical-grade administration. Subcutaneous is the research standard because it's practically achievable without a clinical facility and still produces reliable, well-characterised systemic exposure in a consistent pharmacokinetic profile.
Users report that the practical requirements of injectable protocols accumulate across a sustained research period. Sterile preparation, consistent injection technique, site rotation, tolerability of the injection itself across weeks or months — none of these affect the pharmacokinetics, but they do affect what actually gets done. The delivery-science question the field keeps returning to is whether alternative routes can achieve systemic exposure that's meaningful in a research context, without the injection burden. That's exactly the gap sublingual delivery is designed to address.
Sublingual Delivery: Where the Third Route Actually Sits in the Data
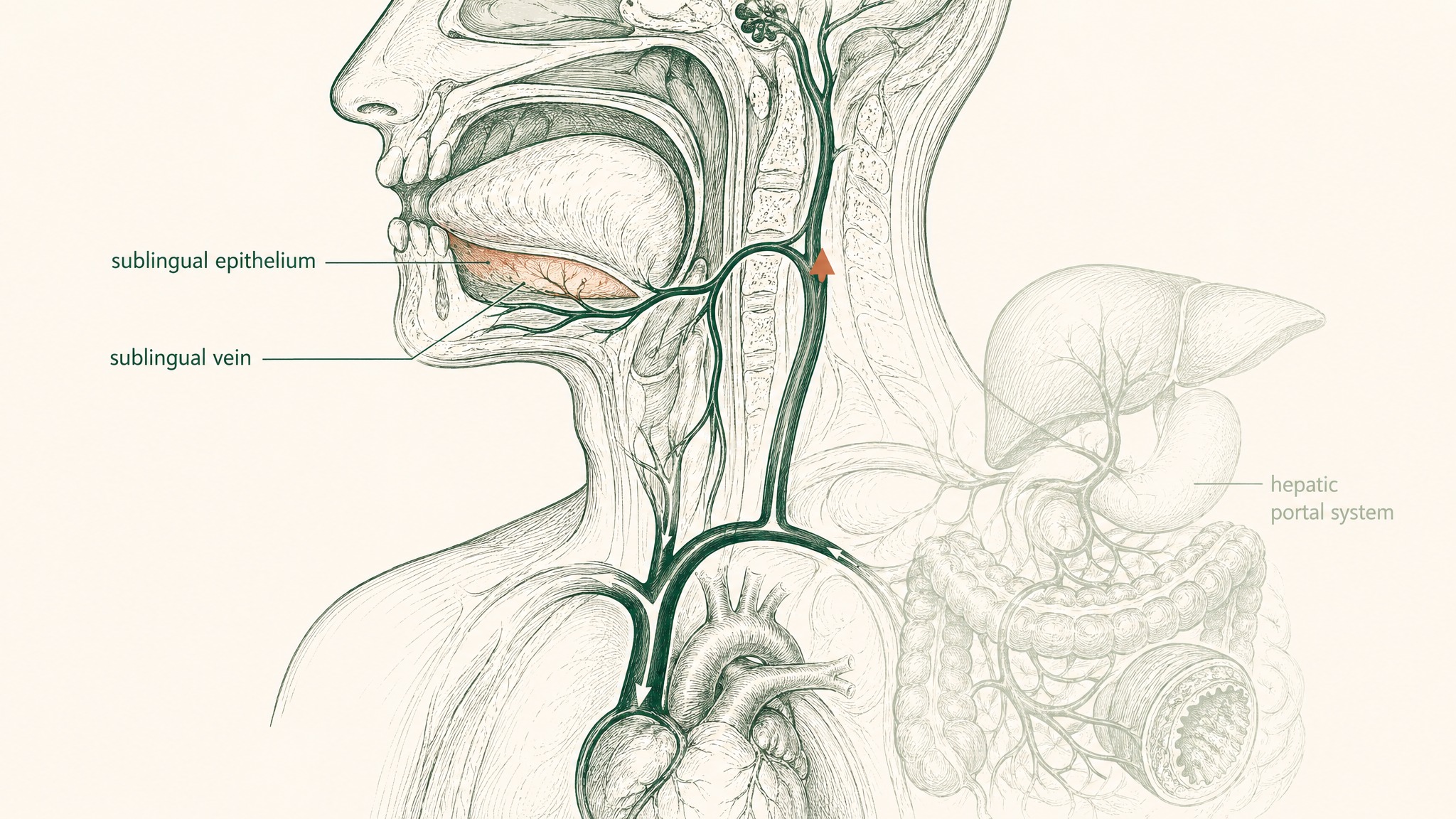
The tissue under your tongue is anatomically different from anything in your gastrointestinal tract. Non-keratinised epithelium — thinner, more permeable, and without the enzymatic artillery of the intestinal wall. Compounds absorbed here drain into the sublingual and lingual veins, then into the internal jugular vein and superior vena cava, entering systemic circulation before the liver processes them. Think of it as a bypass lane around the security checkpoint system: the GI tract is never in the pathway, and first-pass hepatic metabolism doesn't apply (Zhang, Zhang & Streisand 2002, Clin Pharmacokinet, PMID 12126458; Lam et al. 2014, Adv Drug Deliv Rev, PMID 23999459).
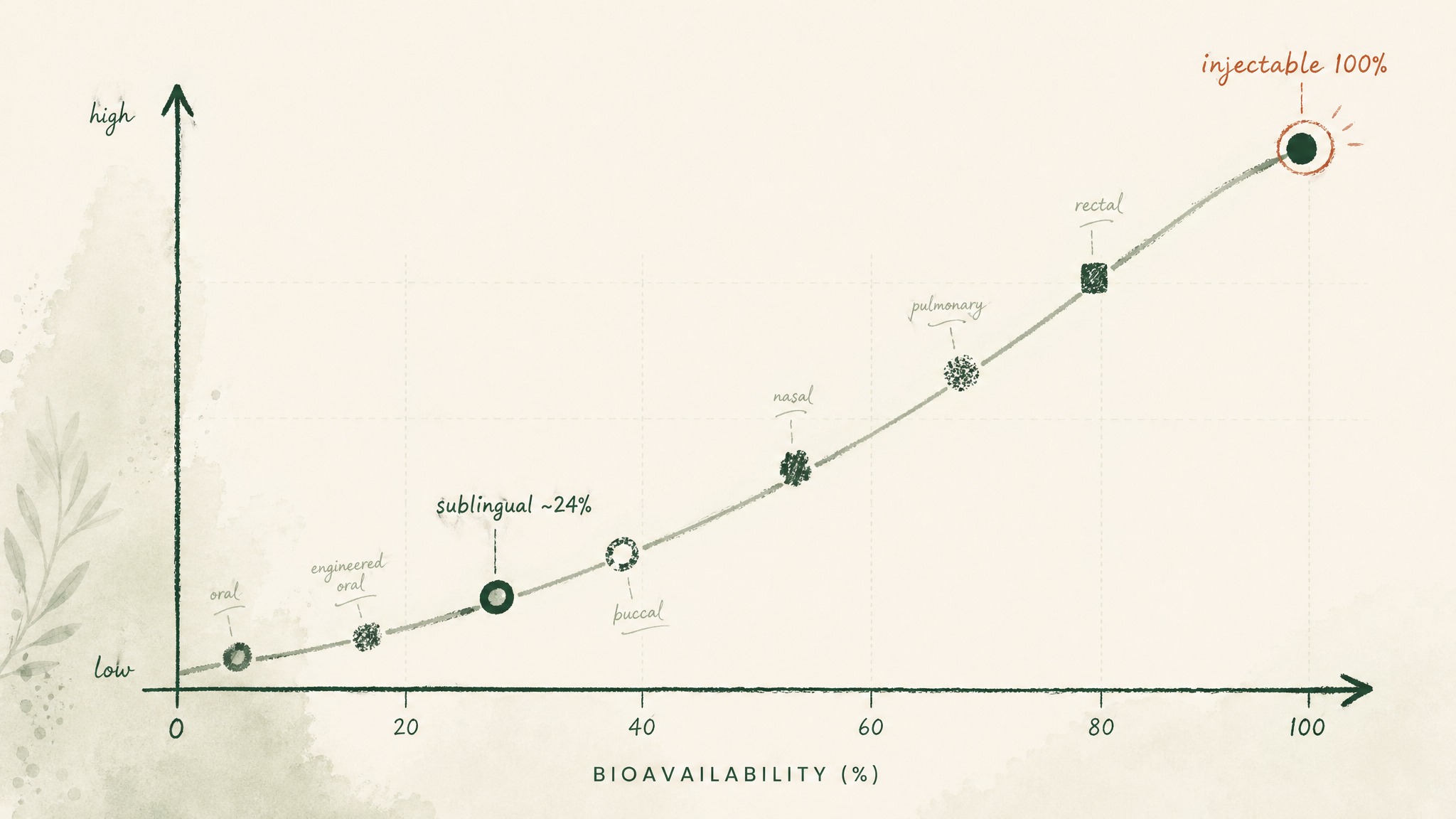
The data on properly formulated sublingual peptide delivery gives concrete numbers for where this route sits. Sublingual delivery of insulin loaded into alginic acid nanoparticles achieved absolute pharmacological availability of 20.2 percent and absolute bioavailability of 24.1 percent relative to a subcutaneous injection reference in streptozotocin-induced diabetic rat models (Patil & Devarajan 2016, Drug Deliv, PMID 24901208). Compound-specific, formulation-specific, animal model data — it doesn't translate directly across species or peptide types. What it establishes is the scale of the difference: an order of magnitude above oral, within range of the injectable reference, for a formulation purpose-built for the route.
More recent work has pushed the molecular weight question further. A 2024 Journal of Controlled Release study demonstrated that novel lipid-conjugated protamine peptides enabled systemic sublingual delivery of proteins ranging from 22 to 150 kilodaltons — spanning from insulin through full-size immunoglobulin G — in mouse models, with pharmacological effects comparable to subcutaneous preparations and no measured toxicity in the animals tested (Wu et al. 2024, J Control Release, PMID 38423473). Research suggests the molecular weight ceiling for sublingual delivery isn't fixed — it extends with formulation sophistication.
The sublingual mucosa does have its own permeability constraints. Compounds with low molecular weight, high potency, and long biological half-life are the natural fit — characteristics that allow efficient absorption across the mucosal surface without heavy formulation assistance (Harris & Robinson 1992, J Pharm Sci, PMID 1619560). Larger or more hydrophilic molecules need permeation enhancers, mucoadhesive polymers, or lipid-based carriers to cross efficiently. An unformulated peptide placed under the tongue doesn't automatically absorb. A purpose-built delivery vehicle is the variable that makes the route work. The sublingual mucosa removes five barriers from the oral route; a well-engineered formulation addresses the one that remains.
What These Numbers Mean When You're Designing a Protocol
The spectrum of non-parenteral peptide delivery routes — oral, sublingual, buccal, nasal, pulmonary, rectal — each sit at different positions on both bioavailability and practicality axes (Zaman et al. 2016, Curr Pharm Des, PMID 27510479). Injectable routes anchor one end. Swallowed oral anchors the other. The numbers across those routes determine what the body actually receives, independent of what the label says went in.
The practical selection question for any research protocol comes down to three variables: what systemic exposure does the research endpoint require, which route can deliver that exposure reliably, and what compliance burden is sustainable across the protocol duration. Route of administration determines systemic peptide exposure as directly as the label dose does. A compound administered orally at 10 mg with 1.5 percent bioavailability produces a fundamentally different systemic exposure than the same compound at the same label dose administered subcutaneously. Designing a protocol without accounting for route-specific bioavailability is designing against the data.
Members experience the compliance variable most concretely in extended protocols. A delivery format that achieves meaningful systemic exposure without requiring injection changes what gets followed through consistently across six or twelve weeks. Consistent dosing is itself a pharmacokinetic variable — irregular adherence introduces exposure variability that compounds what the delivery route contributes biochemically. The format that sustains the protocol is as important as the one that delivers the best single-dose numbers.
The bioavailability data across these routes isn't ambiguous. Less than 2 percent for unformulated oral delivery. Up to 19 percent for the most engineered oral formulations in research settings. Around 24 percent for purpose-formulated sublingual delivery relative to subcutaneous in animal models. The injectable reference as the benchmark that alternative routes are measured against. These are the verified numbers — the ones to hold any product claim against.
Key Takeaways
- Unmodified oral peptides reach the bloodstream at documented bioavailability below 1 to 2 percent. The gut's sequential barriers — gastric acid, proteases, brush-border peptidases, the epithelial wall, tight junctions, and hepatic first-pass metabolism — compound rather than add (Renukuntla et al. 2013, PMC3680128; Verma et al. 2021, PMID 33988872).
- The PepT1 intestinal transporter absorbs di-peptides and tri-peptides only. Most research peptides exceed this chain-length threshold and don't benefit meaningfully from this pathway (Freeman 2015, PMC4419090).
- Subcutaneous injection remains the research reference standard for systemic peptide delivery because it removes the GI barriers entirely and produces a predictable dose-to-exposure relationship (Zhang et al. 2020, PMID 32622810).
- The highest documented oral peptide bioavailability via pharmaceutical engineering is 0.8 percent — oral semaglutide with SNAC under strict conditions (Overgaard et al. 2021, PMID 33969456). Research-stage liposome formulations have reached up to 19 percent for specific compounds in preclinical data (Cui et al. 2022, PMC9501131).
- Sublingual delivery bypasses the GI tract and hepatic first-pass metabolism via oral mucosal tissue (Zhang, Zhang & Streisand 2002, PMID 12126458). Formulated sublingual insulin nanoparticles achieved 24.1 percent absolute bioavailability relative to subcutaneous in rat models (Patil & Devarajan 2016, PMID 24901208). Recent work extends sublingual delivery to proteins up to 150 kDa with comparable effects to subcutaneous in animal models (Wu et al. 2024, PMID 38423473).
- Route selection determines systemic peptide exposure as directly as label dose. Building a protocol without accounting for route-specific bioavailability is designing against the data.
The RESTORE Protocol is built around a sublingual delivery system engineered specifically to address the absorption gap described here. That's where VERO's formulation approach starts — if you want to explore the delivery science behind it.
This content is published for research purposes only and does not constitute medical advice. It is not intended to diagnose, treat, cure, or prevent any disease. Anyone considering a peptide research protocol should consult a qualified clinician before beginning.
RESTORE™
BPC-157Engineered around 500mcg BPC-157. Accelerates deep tissue and joint recovery
Clinical Context
Important Notice: VERO protocols are nutritional and systemic optimisation formats. They are not intended to diagnose, treat, cure, or prevent any medical condition. These statements have not been evaluated by the Food and Drug Administration.