Peptide pill brands borrow injection research and call it clinical proof. Here's how to spot the tactic — and what legitimate products actually disclose.
Here's the honest answer before we get into the mechanics: most peptide pill products aren't scams in the strict legal sense. They don't necessarily contain fake ingredients. What's almost universal in the category is something more subtle: a marketing tactic that borrows research from injected peptide studies and implies those results apply to a swallowed capsule. This content is published for research purposes only.
The gap between injected delivery and a swallowed capsule is the whole ballgame. Understanding that gap is how you go from "I feel like I got ripped off" to knowing exactly what happened and why.
The Borrowed Research Tactic: How Companies Mislead You Without Technically Lying
The most common tactic in the oral peptide supplement category works like this. A company sells BPC-157 capsules. On their product page, they link to genuine peer-reviewed research. The citations are real papers, published in real journals, by real researchers.
What the page doesn't say: those studies used subcutaneous injection (a needle injected just under the skin, bypassing the gut entirely) or intravenous administration (delivered directly into the bloodstream). The research they're citing is evidence that injected BPC-157 produces certain outcomes in controlled settings. It's not evidence that a swallowed capsule does anything comparable.
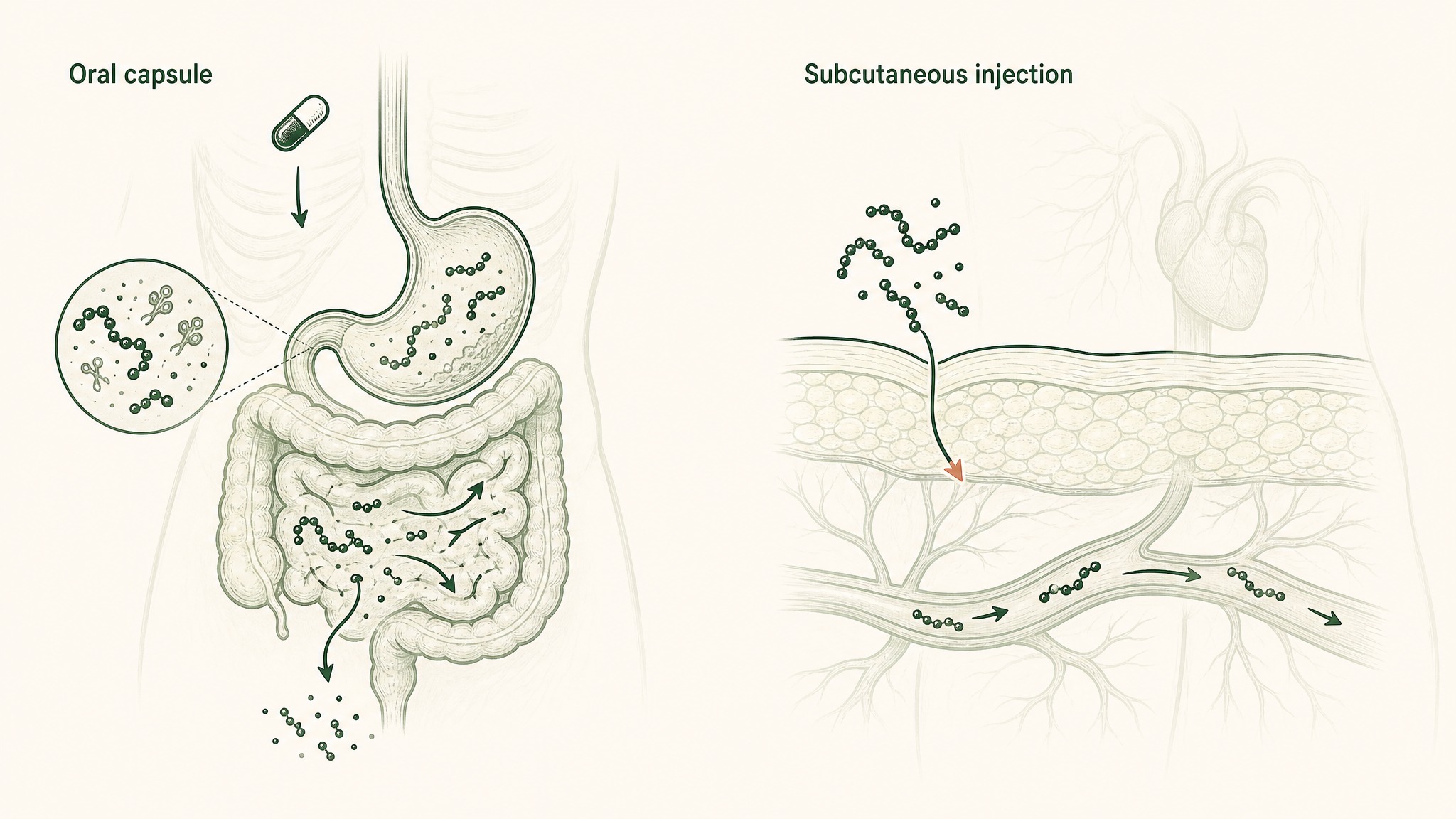
Why a swallowed peptide capsule reaches your bloodstream differently than an injected one.
You've been handed a real citation for a different product.
This pattern shows up across the category:
- "Clinically researched formula" (the research used injection protocols, not oral capsules)
- "Backed by science" (the science used a delivery route this product doesn't use)
- "Shown to support [outcome]" (shown via subcutaneous injection, IV, or in vitro cell studies)
- Study citations with no disclosure of the delivery route used in the methodology
- Before-and-after testimonials with no mention of whether the person switched from another delivery format
This isn't unique to cut-rate brands. It's the default operating procedure for most oral peptide supplement companies, from budget products to premium-positioned ones. The citations are technically real. The implied connection to oral capsule efficacy is not.
Why the Delivery Route Is the Whole Story
The biology behind this matters, even if companies don't make it easy to find. When you swallow a peptide capsule, your digestive system runs it through a multi-stage dismantling process: stomach acid and digestive enzymes begin breaking peptide bonds, brush-border enzymes (enzymes coating your small intestine wall) continue the breakdown, the intestinal wall blocks most peptide-sized molecules from crossing, and the liver processes anything that makes it through before it enters your circulation (Verma et al. 2021, Drug Dev Res, PMID 33988872).
The result of those compounding barriers is a bioavailability figure (how much of what you swallowed actually reaches your bloodstream) that sits below 1 to 2 percent for unmodified oral peptides (Renukuntla et al. 2013, Int J Pharm, PMC3680128). Out of 100 milligrams in a capsule, at least 98 milligrams are destroyed before they reach your blood.
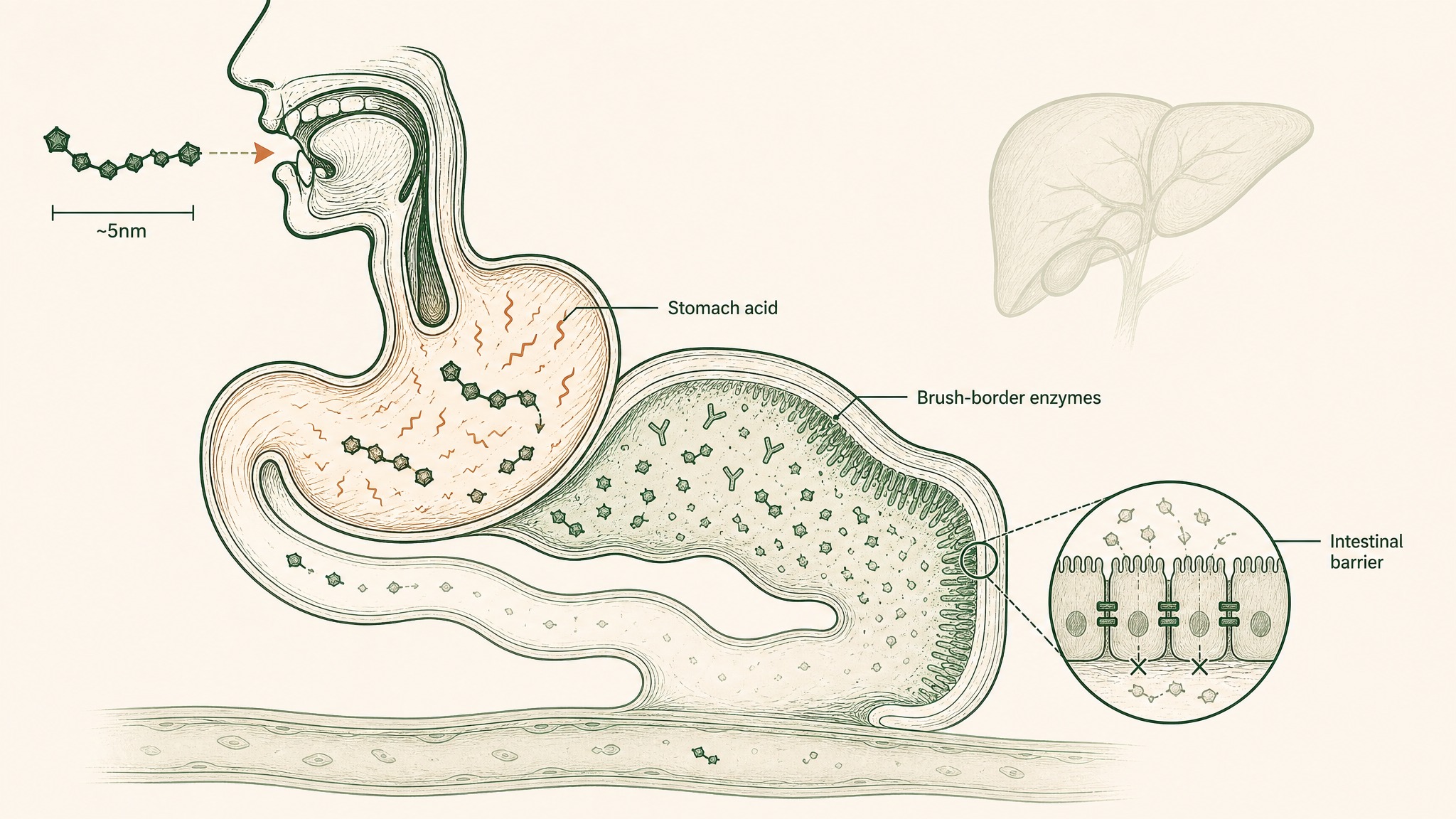
Why 98 percent of a swallowed peptide gets destroyed before reaching your blood.
Subcutaneous injection entirely bypasses this destruction sequence. That's why the research that used injection got results.
A citation from an injection study isn't evidence for what a swallowed capsule does. It's evidence for what the injected compound does — genuinely interesting science that doesn't transfer to the oral format. The tactic works because most people never check the methods section of the cited papers to read the delivery route used. Once you know to look, the gap is impossible to miss.
What "Enteric Coated," "Liposomal," and "Enhanced Absorption" Actually Mean
Once you can spot the borrowed research tactic, you'll start noticing the second layer: formulation marketing claims that sound like they've solved the delivery problem.
The most common ones in the oral peptide category:
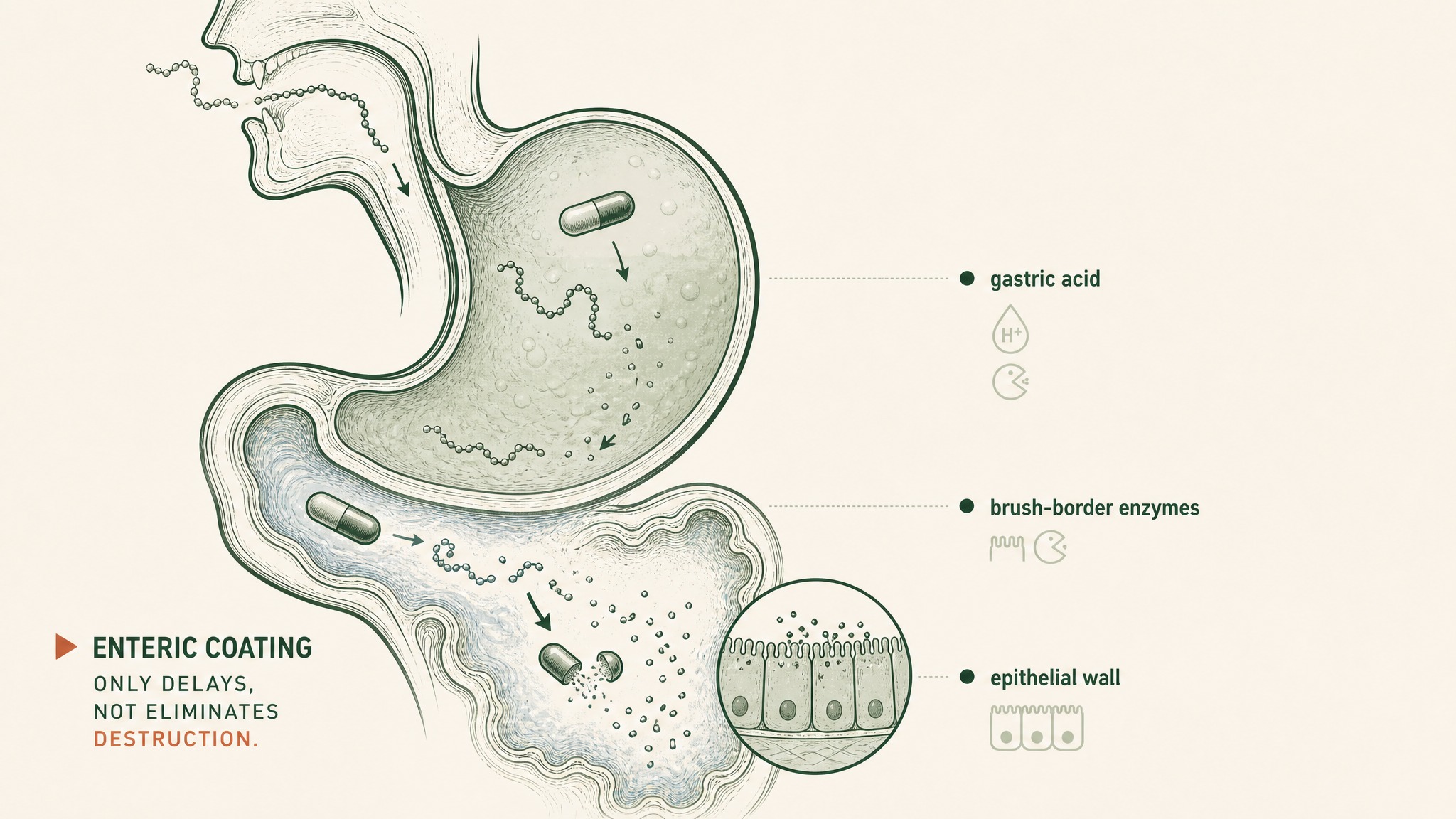
Why enteric coating delays peptide breakdown in the stomach but doesn't — it in the intestine.
- Enteric coating: a shell that delays capsule dissolution until the compound reaches your small intestine rather than your stomach. It bypasses some gastric acid exposure. The brush-border enzymes, the epithelial barrier, and hepatic first-pass metabolism are all still present. Enteric coating delays one part of the destruction sequence; it doesn't eliminate the others.
- Liposomal delivery: encasing peptides in lipid spheres that may improve passage through the gut wall. Real formulation research exists in this area, but results are compound-specific and formulation-specific. Whether a given supplement's "liposomal" formulation uses research-grade lipid chemistry or just the word "liposomal" in marketing copy is nearly impossible to verify without independent testing.
- "Enhanced absorption matrix" or "proprietary delivery system": phrases with no standardised meaning in supplement regulation. Without specifics about which polymers, which permeation enhancers, and what compound-specific testing was done, these are marketing language with no verifiable content.
For context on what serious pharmaceutical engineering actually achieves: Novo Nordisk developed a proprietary absorption enhancer for semaglutide called SNAC, which creates a localised pH rise in the stomach allowing the compound to absorb across the stomach wall under strict dosing conditions (no food, specific water volume, 30-minute window). The result of years of pharmaceutical research targeting one specific compound: absolute oral bioavailability of 0.8 percent (Overgaard et al. 2021, Clin Pharmacokinet, PMID 33969456).
That is the pharmaceutical ceiling. For one purpose-built drug, with proprietary technology a supplement company couldn't license.
An off-the-shelf capsule with "enhanced absorption matrix" on the label hasn't matched what Novo Nordisk built. The label doesn't claim that. But the marketing implies something close.
How to Read a Peptide Product Page Like a Skeptic
Most of the information you need to evaluate a peptide pill is already on the product page. You just need to know where to look and what questions to ask.
The five-question evaluation:
- What route of administration was used in the cited studies? Check the methods section of any paper they link. If the studies used subcutaneous injection, IV, or animal model injection protocols, that research doesn't support the oral capsule being sold.
- Does the brand explain specifically how their formulation achieves absorption? Not "advanced delivery" as marketing shorthand. An actual mechanism: which polymers, which permeation enhancers, tested against which specific peptide. Vague language here is a signal, not a feature.
- Is there a batch-specific certificate of analysis from an independent third-party lab? Not a generic certificate somewhere on the website. A document specific to the batch you're ordering, with compound identity confirmation and purity measurements. If you can't find it, you don't know what's in the capsule.
- Are outcome claims qualified by the level of evidence? Accurate framing for early-stage peptide data looks like "preliminary evidence suggests" or "in animal model research." "Clinically proven to..." without disclosing the conditions of that evidence is a flag worth investigating.
- Does the brand address the oral bioavailability problem at all? A company that has genuinely engaged with the delivery science explains why their approach can overcome the gut's barriers. Complete silence on how the compound reaches your bloodstream is itself meaningful data.
If you can't find clear answers to these questions on the product page, that's the answer.
When Ineffective Becomes Actually Fraudulent
There's a real difference between a product that doesn't work because the oral delivery format can't overcome gut biology, and a product that misrepresents what's in the capsule or makes claims that cross legal lines.
The first is extremely common. The second is a different category of problem.
Under US supplement regulation (the DSHEA framework, which doesn't require companies to prove efficacy before bringing a product to market), companies can sell peptide capsules without demonstrating they deliver anything meaningful to your bloodstream. What they can't do:
- Claim a specific outcome without meeting the FTC's substantiation standard (competent and reliable scientific evidence)
- List an ingredient on the label that isn't in the product or isn't at the stated dose
- Use testimonials implying clinical outcomes the company can't substantiate
- Market a compound in ways that would classify it as a drug (drug-indication language)
Users report a wide range within this space. Some brands are genuinely trying to produce quality research compounds and simply haven't engaged with the delivery science. Others use technically compliant marketing language engineered to imply more than the evidence supports. A smaller group makes claims that are outright deceptive by the FTC's standard.
None of these look dramatically different from the outside of the packaging. The distinction shows up in the details: the delivery route in cited studies, the specificity of absorption explanations, the presence or absence of batch-specific documentation. Those are the variables worth reading for.
What Legitimate Peptide Research Products Actually Disclose
Research suggests the difference between an honest product and a misleading one isn't usually the underlying compound. It's what the brand is willing to say clearly about the limits of what they're selling.
Legitimate peptide research products have a consistent pattern:
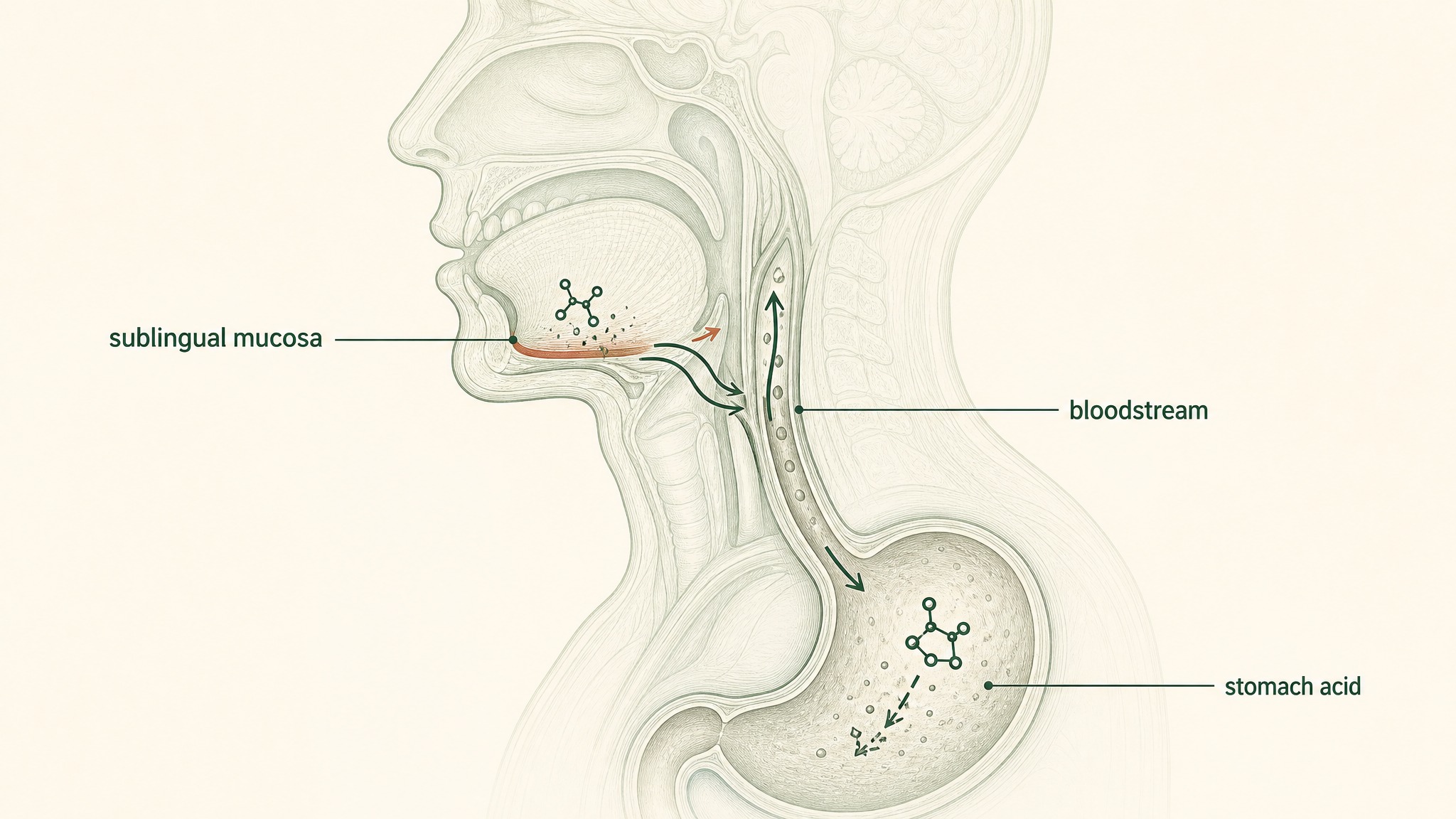
Sublingual peptides absorb through mouth tissue directly into blood; swallowed peptides break down in stomach acid.
They address the delivery problem directly. If a product uses sublingual delivery, the brand explains the sublingual anatomy, why absorption through the oral mucosa bypasses the gut barriers, and what their specific formulation does to address the mucosal membrane. If they sell an oral capsule, they explain what their formulation actually achieves in concrete terms you can cross-check.
They use research-appropriate framing. Most peptide research sits at the preclinical or early human data stage: animal models, in vitro cell studies, small observational cohorts. Honest brands describe their products accordingly. "Not a substitute for clinical care," "preliminary data indicates," "research compound": these framings aren't cover for liability. They're accurate descriptions of where most peptide compounds sit in the scientific evidence hierarchy.
They provide verifiable documentation. Batch-specific certificates of analysis from named, accredited labs, not generic documentation. A specific document for the batch you're buying, accessible before you order.
Members experience a genuinely different conversation with brands operating this way, because the transparency itself is evidence the company understands the science well enough to know what it can and can't claim.
The borrowed research tactic persists because most people don't check delivery routes in cited studies. Once you know to look, you'll find the pattern across most oral peptide supplement marketing. That's not a reason to distrust peptide research as a category. It's a reason to apply a consistent filter when deciding which products are worth your attention.
If you want a deeper look at the biology behind why the oral delivery problem is so difficult to solve, the full breakdown is here.
Key Takeaways
- The dominant tactic in oral peptide pill marketing is borrowing research from injection studies to imply results for a swallowed capsule. The citations are real. They're for a different delivery method.
- Oral bioavailability for unmodified peptides sits below 1 to 2 percent (Renukuntla et al. 2013, PMC3680128). The pharmaceutical ceiling for engineered oral peptide delivery is 0.8 percent, for one specific drug, with proprietary absorption technology built specifically for that compound (Overgaard et al. 2021, PMID 33969456).
- "Enteric coated," "liposomal," and "enhanced absorption matrix" are formulation claims that require specific, independently verified evidence to mean anything. Without it, they're marketing language.
- Your five-question checklist: check the delivery route in cited studies; ask for the specific absorption mechanism; require batch-specific certificates of analysis; look for research-appropriate claim framing; note whether the brand addresses the bioavailability gap.
- There is a meaningful difference between a product that's ineffective due to delivery format limitations and one misrepresenting its ingredients, dosing, or clinical outcomes.
- Legitimate peptide research brands explain their delivery technology specifically, use research-consistent framing, and provide verifiable batch documentation.
Want to see what a delivery system built around sublingual absorption actually looks like? Explore the RESTORE Protocol →
This content is published for research purposes only and does not constitute medical advice. It is not intended to diagnose, treat, cure, or prevent any disease. Anyone considering a peptide research protocol should consult a qualified clinician before beginning.
RESTORE™
BPC-157Engineered around 500mcg BPC-157. Accelerates deep tissue and joint recovery
Clinical Context
Important Notice: VERO protocols are nutritional and systemic optimisation formats. They are not intended to diagnose, treat, cure, or prevent any medical condition. These statements have not been evaluated by the Food and Drug Administration.

