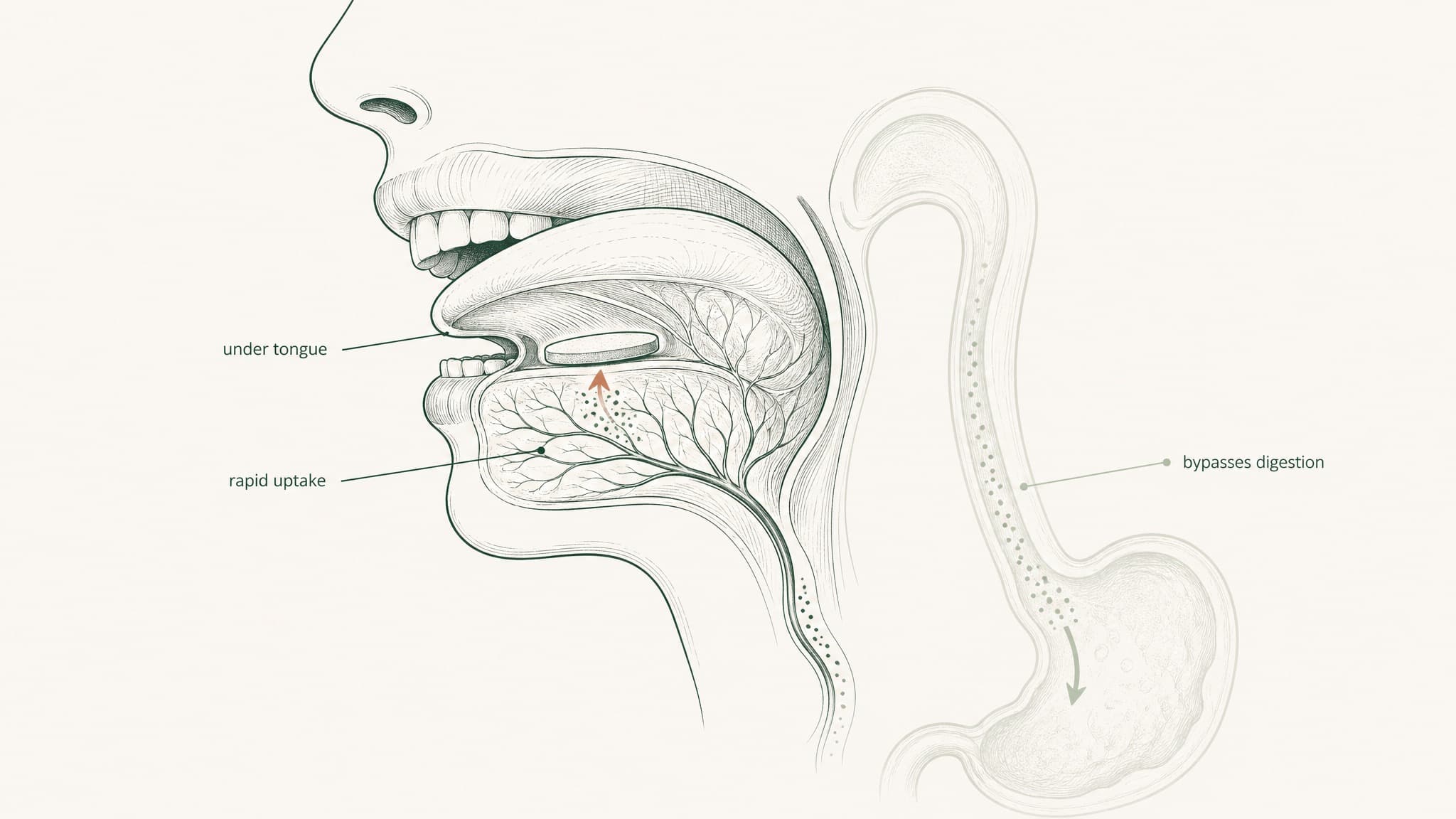
VERISORB is VERO's proprietary sublingual delivery system — engineered to get peptides from under your tongue into your bloodstream without injection. Here's what it is, how it works, and why formulation engineering is the difference between a peptide that absorbs and one that doesn't.
Here's the shortest version of what VERISORB is: a delivery system designed to solve a problem your digestive system creates.
Peptides are extraordinarily fragile in your gut. Swallow one in a capsule and the demolition sequence starts immediately — stomach acid, enzymes, intestinal walls, and finally your liver each take a cut before anything reaches your bloodstream. By the time that sequence finishes, less than 1 to 2 percent of the original dose survives intact (Renukuntla et al. 2013, Int J Pharm, PMC3680128). That's not a pessimistic estimate. That's what the research consistently documents.
VERISORB is VERO's proprietary sublingual delivery platform — built to route peptides through a completely different pathway. Under your tongue, into your blood, before your gut ever gets involved. But here's what most "sublingual" product descriptions skip entirely: the route alone doesn't do the work. The formulation engineering does. This content is published for research purposes only.
The Problem VERISORB Was Built to Solve
Think of your gut as a tiered security system with five checkpoints, each one built to dismantle protein structures before they can pass through. Peptides are chains of amino acids. Your digestive system was designed to break down chains of amino acids. The two don't coexist well.
Your stomach runs at pH 1.5 to 3.5 — roughly battery acid territory — and produces an enzyme called pepsin whose entire function is cutting the chemical bonds that hold peptide chains together. Anything that survives the stomach moves into the small intestine, where a second wave of enzymes called brush-border peptidases lines the intestinal wall and continues the work. Beneath those enzymes is the epithelial barrier: a continuous cell layer acting as both a physical wall and a biochemical checkpoint, complete with intracellular enzymes that process anything attempting to cross. The gaps between those cells are sealed by structures called tight junctions — selective channels that block most larger molecules from slipping through.
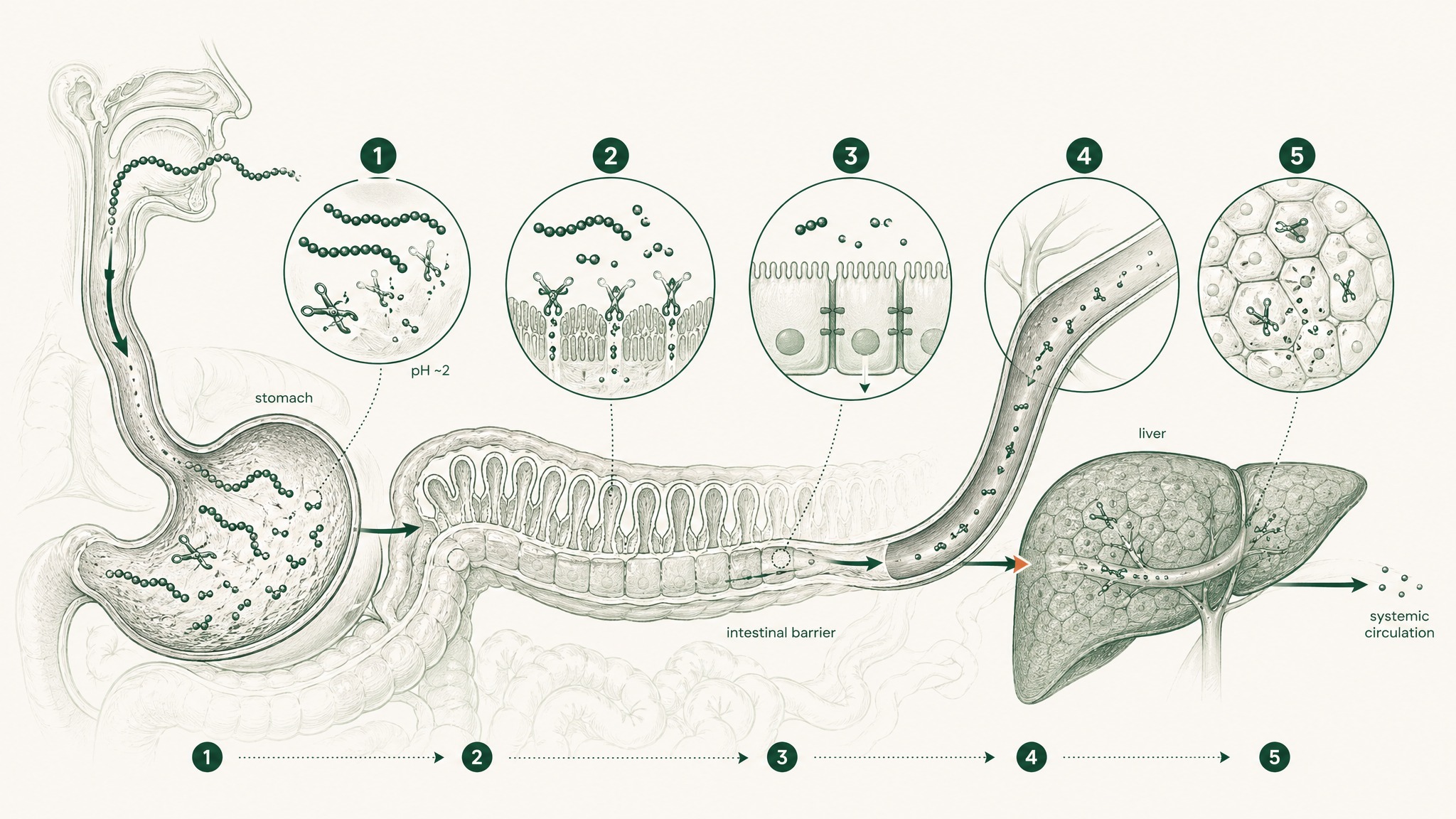
And then there's the final cut. Everything absorbed from the gut enters the portal circulation — a direct route from the intestines to the liver — before the rest of your body sees any of it. Your liver identifies compounds arriving from the gut and processes them. For peptides, this typically means further enzymatic breakdown. This is first-pass hepatic metabolism (your liver's first go at whatever you absorbed before the rest of your body gets it), and it represents the last in a sequence that each compounds on whatever the previous stage left behind (Verma et al. 2021, Drug Dev Res, PMID 33988872).
The 1 to 2 percent bioavailability figure isn't the product of any single checkpoint. It's what happens when five stages of degradation operate sequentially on the same compound. That's the problem VERISORB was engineered around.
Why Sublingual Is a Fundamentally Different Route
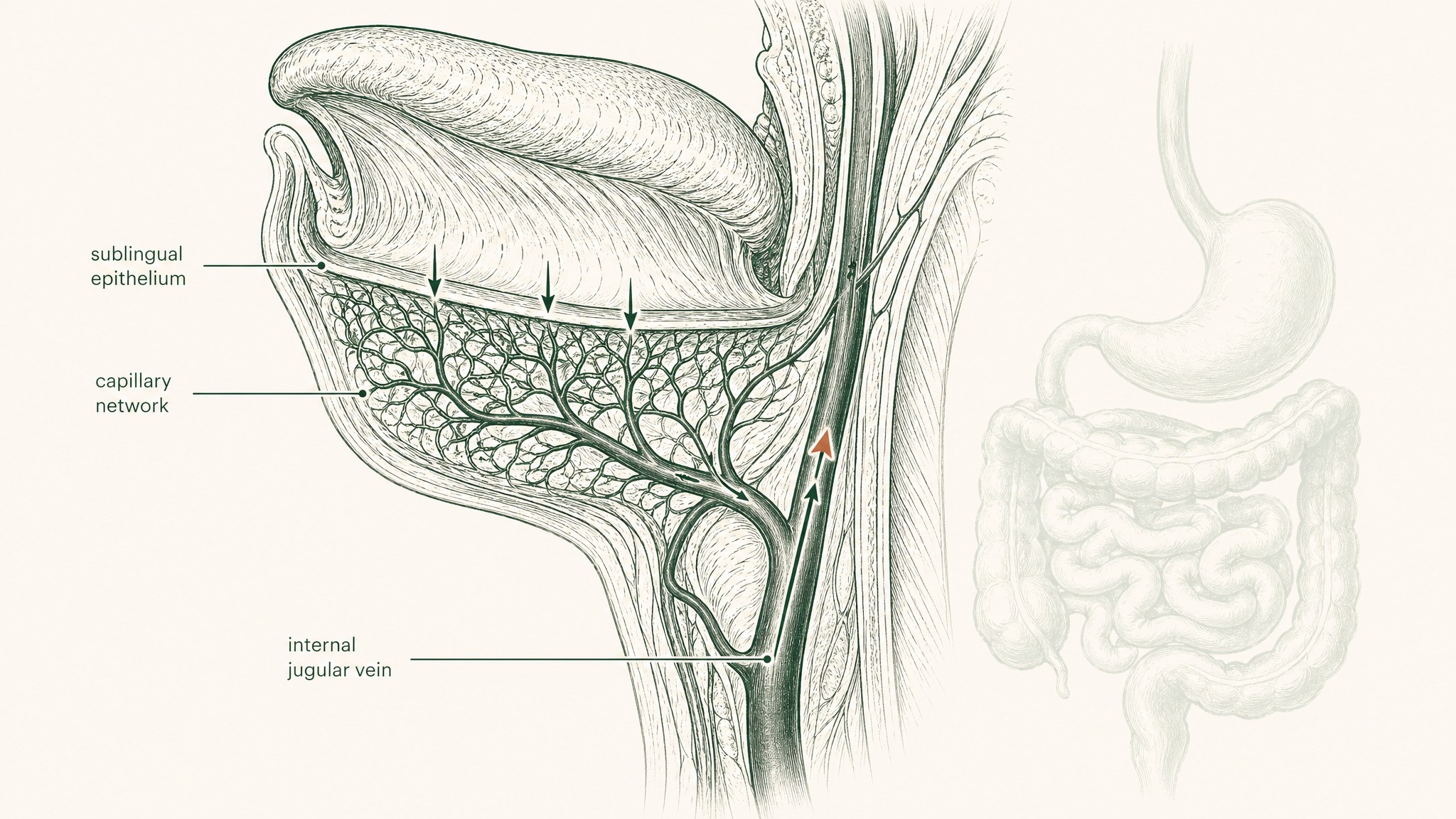
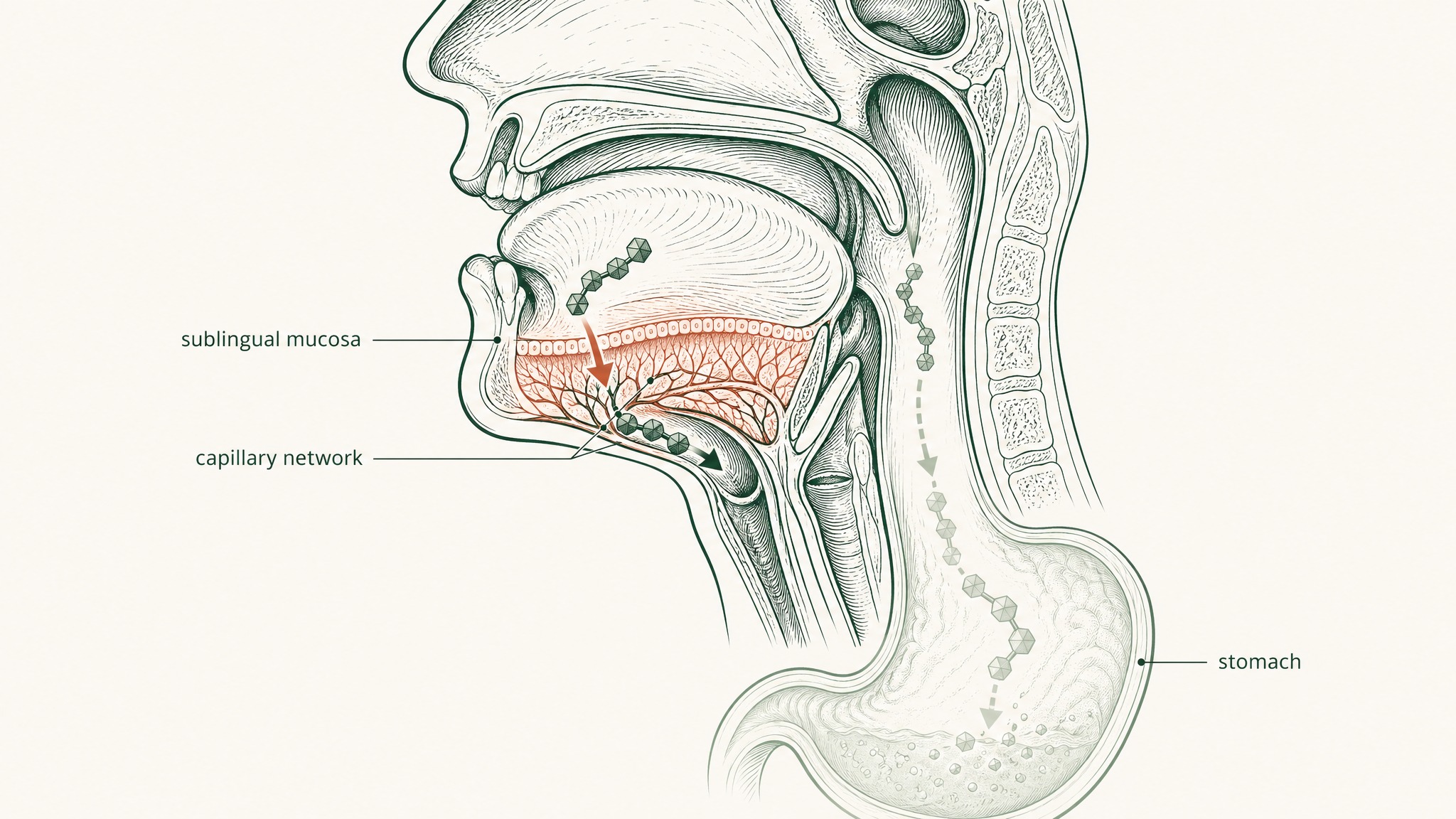
The tissue under your tongue is anatomically unlike anything in your gastrointestinal tract. It's non-keratinised epithelium — thinner, more permeable, and without the enzymatic arsenal that lines the intestinal wall. Beneath it sits a dense network of capillaries and small veins. When a compound crosses that membrane, it enters venous blood that drains to the lingual and sublingual veins, then to the internal jugular, and from there into systemic circulation (Zhang, Zhang & Streisand 2002, Clin Pharmacokinet, PMID 12126458).
Think of it as a bypass lane that routes around all five checkpoints at once. Your gut is never in the pathway. And because the compound enters systemic circulation before it reaches the liver, first-pass hepatic metabolism doesn't apply — what absorbs sublingually goes directly into your blood, not through your liver first (Lam et al. 2014, Adv Drug Deliv Rev, PMID 23999459).
Injectable delivery achieves something similar. Subcutaneous injection deposits a compound directly into vascularised tissue just beneath your skin, bypassing the gut entirely — which is why injectable peptide formats remain the reference standard for systemic exposure in research settings (Zhang et al. 2020, Int J Pharm, PMID 32622810). Sublingual delivery offers a route with comparable pharmacokinetic logic — sidestepping the same barriers — without the needle.
The catch is that crossing the sublingual mucosa isn't automatic. An unformulated peptide placed under the tongue doesn't absorb reliably. And this is where most sublingual products fall short.
What a Basic Sublingual Drop Gets Wrong
There's a detail most sublingual peptide products quietly skip: placing something under your tongue doesn't mean it absorbs there.
You swallow involuntarily every 30 to 60 seconds. Saliva continuously flushes the sublingual space. An unformulated solution placed under your tongue — peptide dissolved in liquid, no engineering, just placed there — is mostly swallowed before meaningful mucosal absorption can occur. The absorption window is measured in seconds before saliva clears the compound downward into the gut where the destruction sequence starts. Effectively, you've taken an oral supplement via a longer route.
Research suggests compounds with low molecular weight, high potency, and long biological half-life are the best natural candidates for sublingual absorption — because those characteristics allow efficient transit across the mucosal surface before salivary clearance removes them (Harris & Robinson 1992, J Pharm Sci, PMID 1619560). Most research peptides don't have that combination of properties. They need formulation engineering to make the sublingual route work.
That's the gap VERISORB fills.
The Three Engineering Problems VERISORB Addresses
VERISORB isn't a sublingual drop. It's a structured delivery system built around three specific formulation problems that an unengineered compound can't overcome on its own.
Problem 1: Mucosal Contact Time
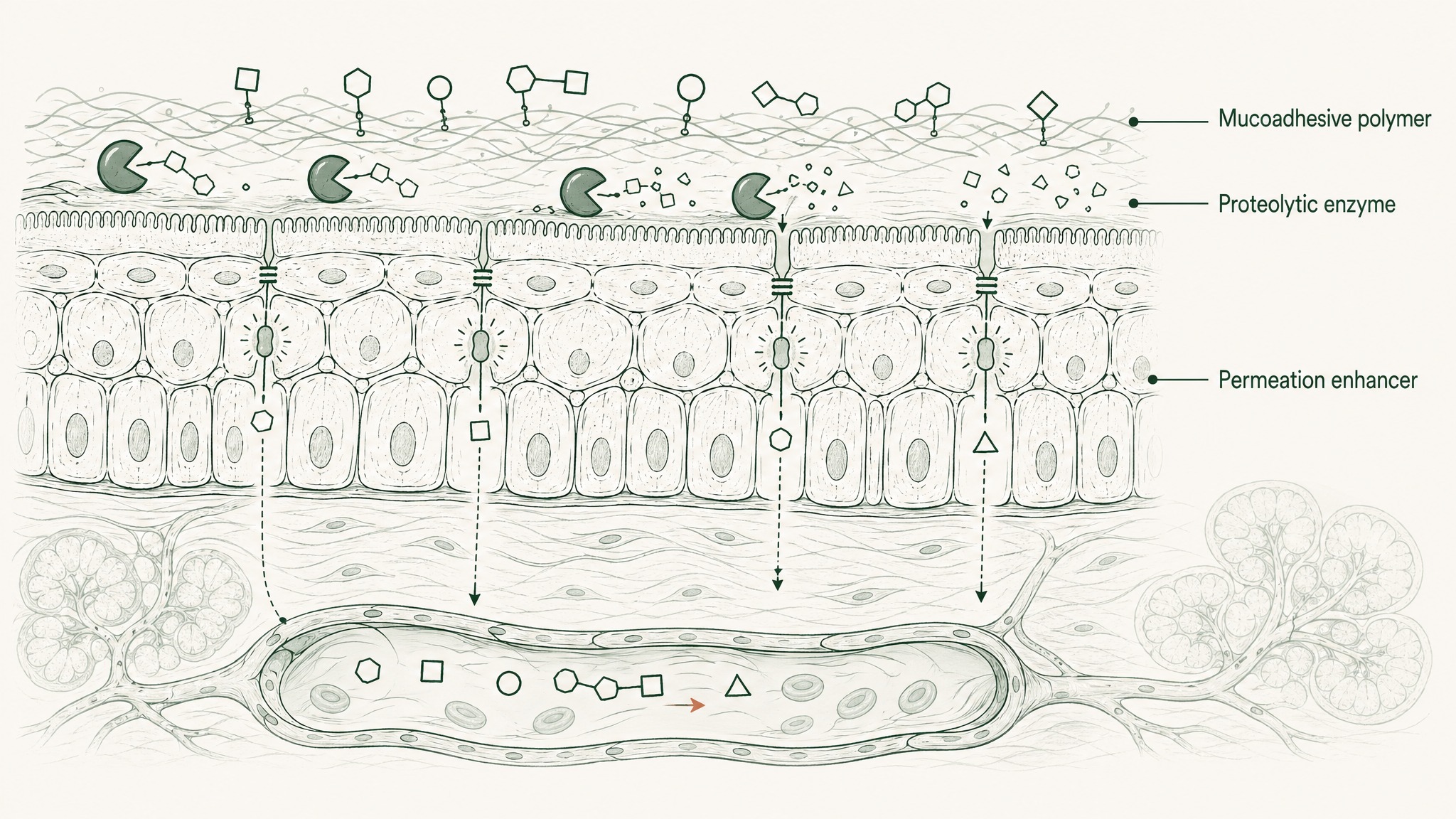
Absorption can't happen if the compound isn't in contact with the mucosal surface long enough for it to occur. An unformulated solution placed sublingually has seconds before saliva flushes it away.
Mucoadhesive polymers — compounds that adhere to mucosal surfaces and resist saliva-mediated clearance — are what change this. Think of them as a controlled-release anchor: instead of the compound washing away in 30 seconds, the polymer matrix holds it against the mucosal tissue for several minutes. Research confirms that mucoadhesive polymer-based films provide sustained delivery of compounds through the sublingual mucosal epithelium by remaining attached to the mucosal surface for sufficient time to enable absorption rather than washout (Chen et al. 2015, J Pharm Pharmacol, PMID 25303221). That extended contact window is what transforms the sublingual route from a theoretical pharmacokinetic advantage into a practical one.
Problem 2: Crossing the Membrane
Adhesion extends contact time. It doesn't solve the crossing problem — those are two different things.
The sublingual mucosal membrane is lipid-rich. Most peptides are hydrophilic (water-attracted), which means they're not inclined to dissolve through a lipid-based barrier. Think of it like trying to get a water droplet through an oil layer. Contact alone doesn't produce crossing. Sitting against the mucosal surface and actually moving through it into the submucosal vasculature are two very different events.
Permeation enhancers are excipients — inactive formulation components — that transiently modify the membrane to facilitate compound transit. They're the keycard that adhesion gets you to the door of. Current formulation research identifies permeation enhancers as one of the two primary strategies for overcoming the lipid-rich mucosal epithelial barrier to peptide delivery, alongside mucoadhesive polymers (Karki et al. 2025, Drug Deliv Transl Res, PMID 41354997). Without permeation enhancement, a hydrophilic peptide can be adhered to the membrane surface and still not transit into the bloodstream beneath it.
Problem 3: Salivary Enzyme Attack
Your saliva contains proteolytic enzymes — the same family the gut uses to digest food protein. They begin working on peptides on contact. This means a peptide sitting against the sublingual mucosa can simultaneously be attempting to absorb and being degraded from the outside. Absorption and destruction compete for the entire contact window.
A formulation that doesn't protect the compound during this window allows degradation to reduce what's available for absorption throughout the contact period. Salivary enzymatic instability is identified in the current peptide delivery literature as one of the three core barriers to oromucosal peptide delivery — alongside salivary washout and the mucosal barrier itself (Karki et al. 2025, PMID 41354997).
VERISORB applies matrix-based protection to reduce competitive salivary degradation during the absorption window. The compound arrives at the mucosal surface in a formulation designed to keep it intact long enough to absorb, not just long enough to be cleared.
What the Research Shows for Formulated Sublingual Delivery
The case for engineered sublingual delivery isn't theoretical. The research on properly formulated sublingual preparations makes the potential bioavailability numbers concrete.
Sublingual delivery of insulin loaded into alginic acid nanoparticles achieved absolute bioavailability of 24.1 percent relative to a subcutaneous injection reference in diabetic rat models (Patil & Devarajan 2016, Drug Deliv, PMID 24901208). Those are compound-specific, formulation-specific numbers from an animal model — they don't translate directly across species or peptide types. What they establish is the order-of-magnitude difference a sublingual route can produce versus swallowed oral delivery when the formulation is engineered for the route rather than just placed in a capsule.
More recent work has extended this thinking to larger molecules. Research suggests sublingual delivery of proteins ranging from 22 to 150 kilodaltons — a range spanning human growth hormone up to full-size immunoglobulin antibodies — can produce pharmacological effects comparable to subcutaneous preparations in mouse models, with no measured toxicity observed in the animals tested (Wu et al. 2024, J Control Release, PMID 38423473). The molecular weight ceiling for sublingual delivery isn't a fixed biological limit. It's a formulation engineering question.
Members experience the difference between an unformulated sublingual product and an engineered one most clearly in protocol consistency — what gets absorbed reliably versus what mostly gets swallowed. The pharmacokinetics of these two scenarios are very different. The formulation is the variable that separates them.
How VERISORB Works Across VERO Protocols
VERISORB is the delivery platform that runs through every VERO protocol. The specific peptide changes between RESTORE, SHIFT, RADIANCE, and CLARITY. The underlying engineering approach — mucoadhesive polymer matrix for extended contact time, permeation-enhancing excipients to facilitate membrane crossing, and compound protection during the contact window — is consistent across all four.
RESTORE uses VERISORB to target the systemic BPC-157 and related peptide research. RADIANCE delivers GHK-Cu via the same system, addressing the specific challenge of getting a hydrophilic linear tripeptide across the sublingual mucosa without injection. In each case, the protocol rationale connects directly to the research reviewed above: the documented biology of these peptides was characterised using delivery systems engineered to produce systemic exposure. VERISORB is designed to match that requirement.
Users report that the practical non-injection format changes what's sustainable across a sustained research protocol. Consistent dosing across weeks and months matters for what the pharmacokinetics actually produce over time. Irregular adherence introduces exposure variability that compounds on top of what the delivery route contributes. A format you'll actually use consistently is a pharmacokinetic variable, not just a convenience preference.
Every VERO product is supplied for research purposes only, has not been evaluated by the FDA for any indication, and is not intended to diagnose, treat, cure, or prevent any condition. Protocol decisions should be made in consultation with a qualified healthcare professional.
Key Takeaways
Unmodified oral peptides reach the bloodstream at documented bioavailability below 1 to 2 percent. The gut's sequential barriers — gastric acid, pepsin, brush-border peptidases, the epithelial wall, tight junctions, and hepatic first-pass metabolism — compound rather than add. Each stage operates on what the previous stage left (Renukuntla et al. 2013, PMC3680128; Verma et al. 2021, PMID 33988872).
Sublingual delivery bypasses the entire GI tract and hepatic first-pass metabolism via oral mucosal venous drainage into systemic circulation. Compounds absorbed under the tongue reach the blood before the liver processes them (Zhang et al. 2002, PMID 12126458; Lam et al. 2014, PMID 23999459).
Unformulated sublingual drops mostly get swallowed. Involuntary swallowing every 30 to 60 seconds clears the sublingual space before meaningful mucosal transit occurs. The route advantage is real; an unengineered formulation can't access it.
VERISORB addresses three specific engineering barriers: mucosal contact time (mucoadhesive polymers that resist salivary clearance and extend the absorption window), membrane crossing (permeation enhancers that facilitate hydrophilic peptide transit through the lipid-rich mucosal barrier), and compound protection during the contact window (matrix formulation that reduces competitive salivary enzymatic degradation). These three barriers are identified in the current formulation literature as the primary obstacles to oromucosal peptide delivery (Karki et al. 2025, PMID 41354997; Chen et al. 2015, PMID 25303221).
Formulated sublingual delivery of insulin nanoparticles achieved 24.1 percent absolute bioavailability relative to subcutaneous injection in animal models (Patil & Devarajan 2016, PMID 24901208). Research suggests the molecular weight ceiling for sublingual delivery extends to proteins of 150 kDa when formulation engineering is optimised (Wu et al. 2024, PMID 38423473).
The sublingual route removes the barriers. The formulation engineering accesses the route. Both are required.
VERISORB is the delivery platform underlying every VERO protocol. Explore the protocols →
This content is published for research and educational purposes only. It is not intended to diagnose, treat, cure, or prevent any disease, and it does not constitute medical advice. Readers should consult a qualified healthcare professional before making any decisions about their own health. Statements on this page have not been evaluated by the FDA. VERO's peptide products are supplied for research purposes only.
RESTORE™
BPC-157Engineered around 500mcg BPC-157. Accelerates deep tissue and joint recovery
Clinical Context
Important Notice: VERO protocols are nutritional and systemic optimisation formats. They are not intended to diagnose, treat, cure, or prevent any medical condition. These statements have not been evaluated by the Food and Drug Administration.