Most oral peptide supplements never reach your bloodstream. Here's the biology behind why — and what the bioavailability numbers actually show.
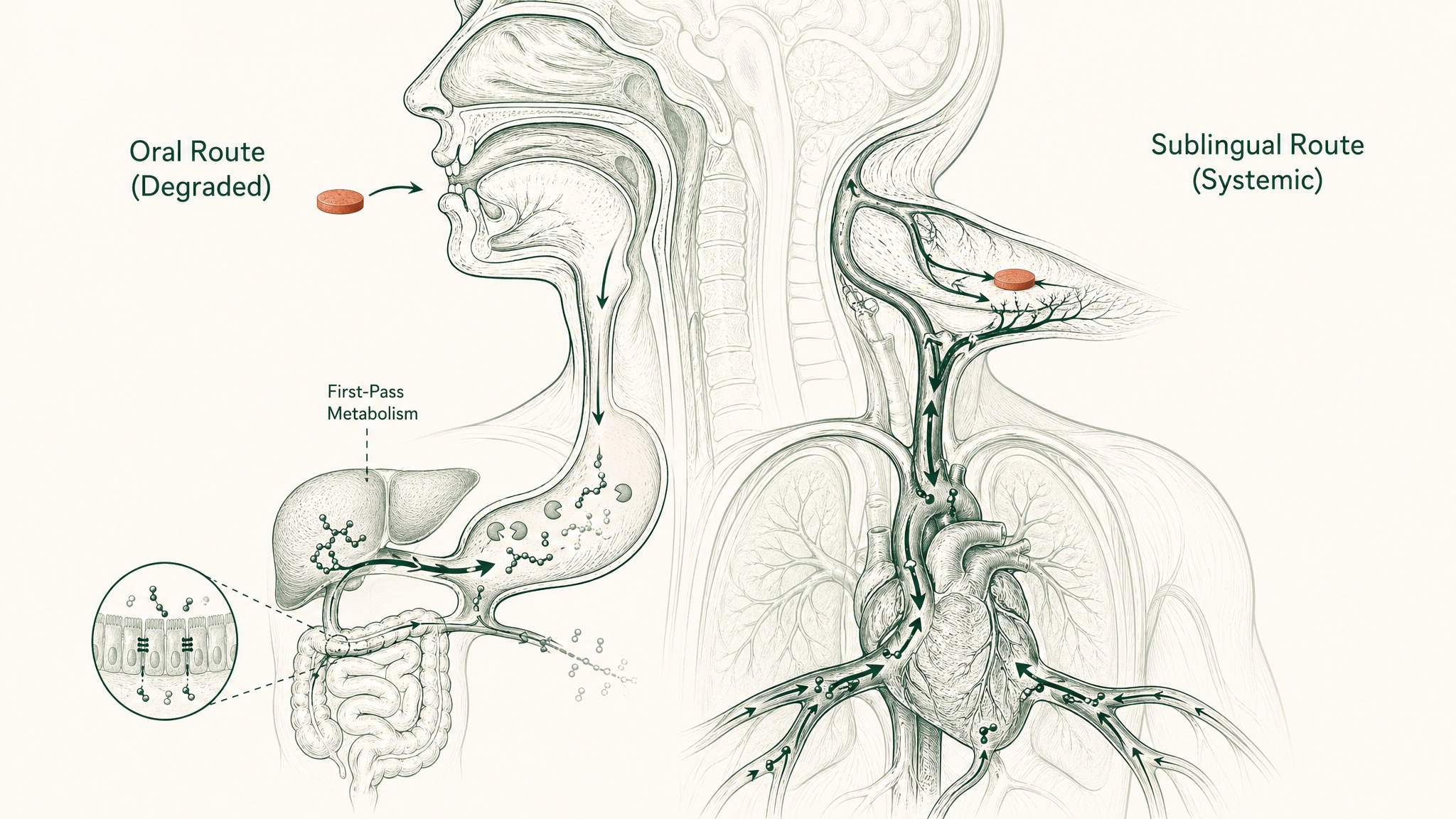
If you've taken oral peptide capsules and wondered why the results don't match the marketing, the published research has a clear answer. Unmodified oral peptides typically achieve bioavailability (how much of what you swallow actually makes it into your bloodstream) of less than 1 to 2 percent (Renukuntla et al. 2013, Int J Pharm, PMC3680128). Your gut is a precision dismantling system for proteins. Peptides are chains of amino acids. The two don't coexist well. This content is published for research purposes only.
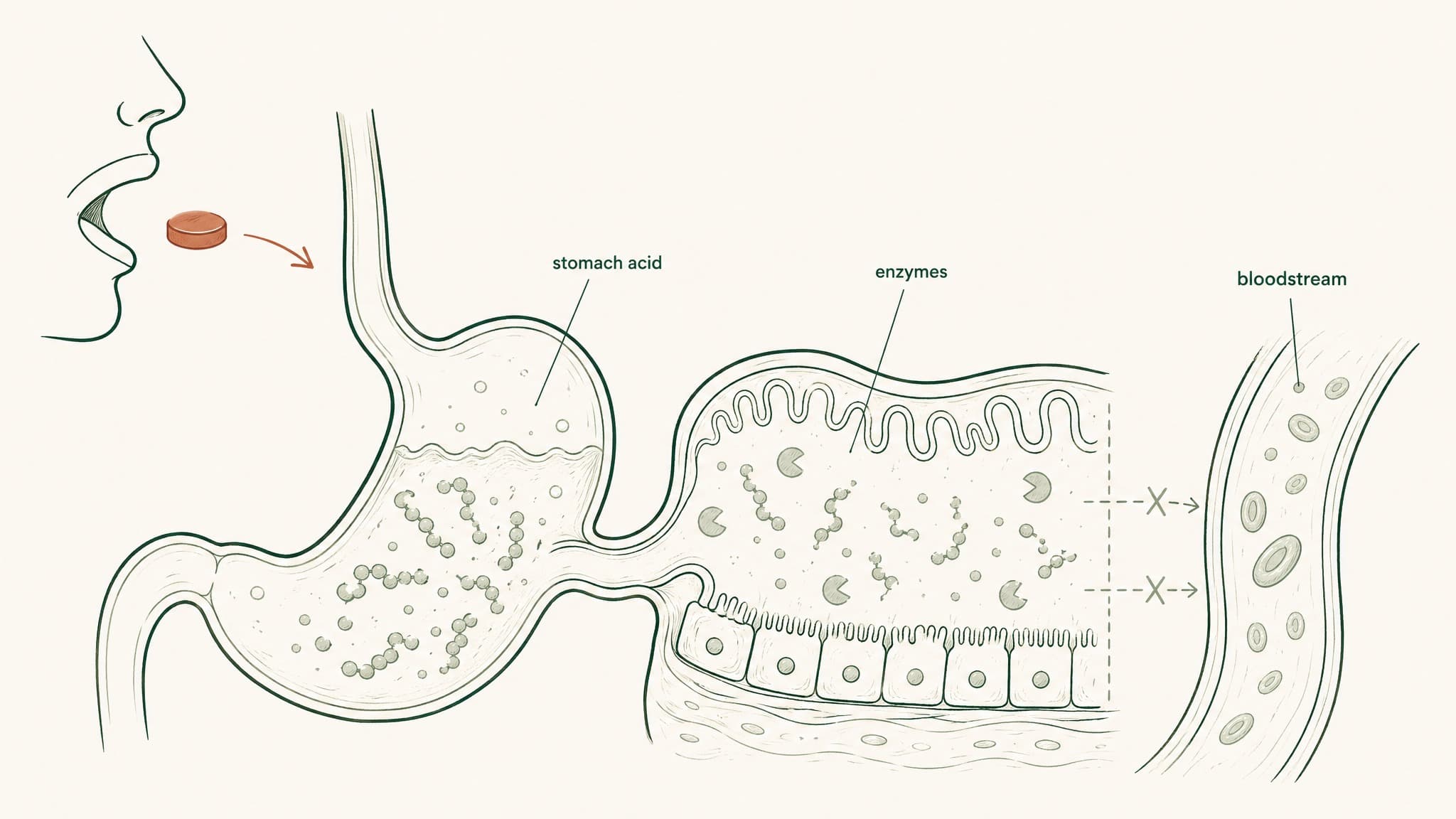
Your Stomach Is a Demolition Zone
Think of your stomach as a high-security processing facility built specifically to dismantle protein. Its primary job is to break down anything that looks like food protein before passing the pieces downstream. Peptides look exactly like food protein to your stomach.
The moment an oral peptide capsule opens in your stomach, it enters an environment running at pH 1.5 to 3.5. That's roughly in the same range as battery acid. Most peptide structures don't survive prolonged exposure to that environment without significant unfolding and disruption of their molecular architecture. But the acidic pH isn't even the main event. Your stomach produces pepsin, a protease, meaning an enzyme whose entire function is cutting peptide bonds. Pepsin is good at its job. It was doing this job long before peptide supplements existed.
What leaves the stomach and enters the small intestine is a partially degraded mixture. Some fraction of a peptide might survive intact or partially intact. That fraction then faces everything the small intestine has prepared for it.
Three Intestinal Barriers That Catch What the Stomach Misses
The small intestine is where most of the remaining damage gets done. It doesn't operate as a single barrier. It operates as three distinct layers of dismantling, each one catching what slipped through the last.
Brush-border peptidases. The lining of your small intestine is covered in tiny finger-like projections called villi, and the surface of those villi is dense with enzymes called brush-border peptidases. Their function is to continue the work the stomach started: breaking peptide fragments down to the smallest possible units, single amino acids or short two-to-three amino acid sequences, so they can be absorbed as nutrition. A research peptide arriving here gets processed by the same machinery as a piece of chicken breast you ate for lunch. The enzymes don't distinguish between "food protein" and "protocol compound."
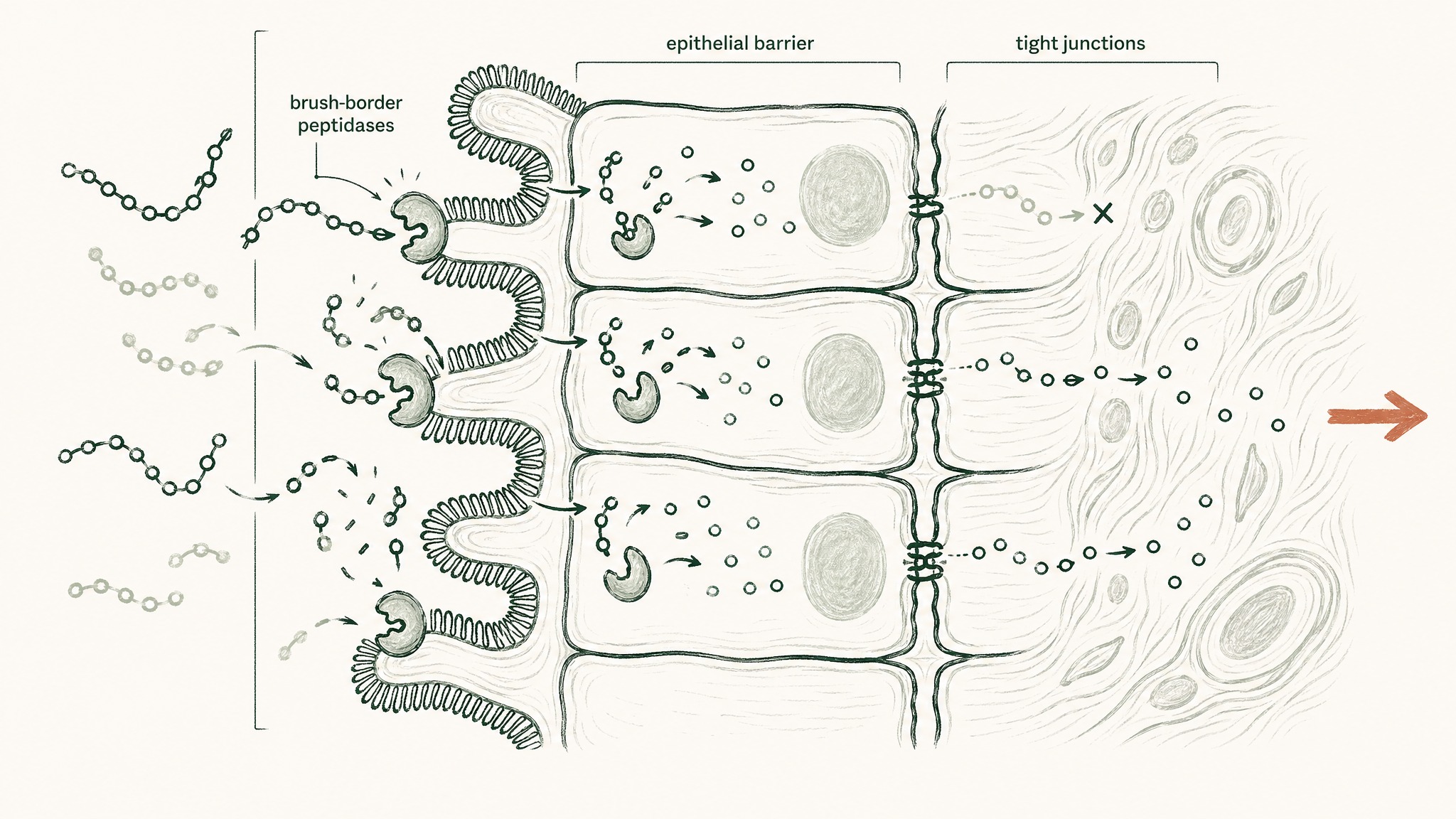
The epithelial barrier. Beneath the brush-border is the intestinal epithelium, a single continuous layer of tightly packed cells that regulates what enters your body. Researchers describe the gastrointestinal epithelium as acting simultaneously as a physical and biochemical barrier for the absorption of proteins (Renukuntla et al. 2013, PMC3680128). The physical component is the cell wall itself; the biochemical component is the fact that epithelial cells also contain intracellular enzymes that continue degradation of anything that begins to cross. Peptides face active resistance, not passive exclusion.
Tight junctions. The gaps between epithelial cells are sealed by protein structures called tight junctions. These aren't open pores. They're selective channels that allow small molecules through while blocking larger ones. The junctions exist to protect you: keeping undigested food antigens, bacteria, and foreign compounds out of your systemic circulation. Most research peptides are large enough that tight junctions are largely closed to them as an absorption pathway.
Working together, these three barriers mean that even a peptide that survives gastric acid and pepsin has a very small chance of crossing the intestinal wall in a usable form.
The Liver's Final Cut
Here's where the numbers become genuinely harsh. Say a small fraction of an oral peptide manages to slip across the intestinal epithelium. You might assume it goes straight to your bloodstream. It doesn't.
Everything absorbed from your gut enters the portal circulation, a direct route from the intestines to the liver, before the rest of your body sees any of it. The liver is your body's metabolic processing centre. It identifies compounds coming in from the gut and processes them. For peptides, this typically means further enzymatic breakdown by hepatic proteases. This stage is called first-pass hepatic metabolism, and it represents the last in a sequence of barriers that each take a significant cut from whatever survived the stage before.
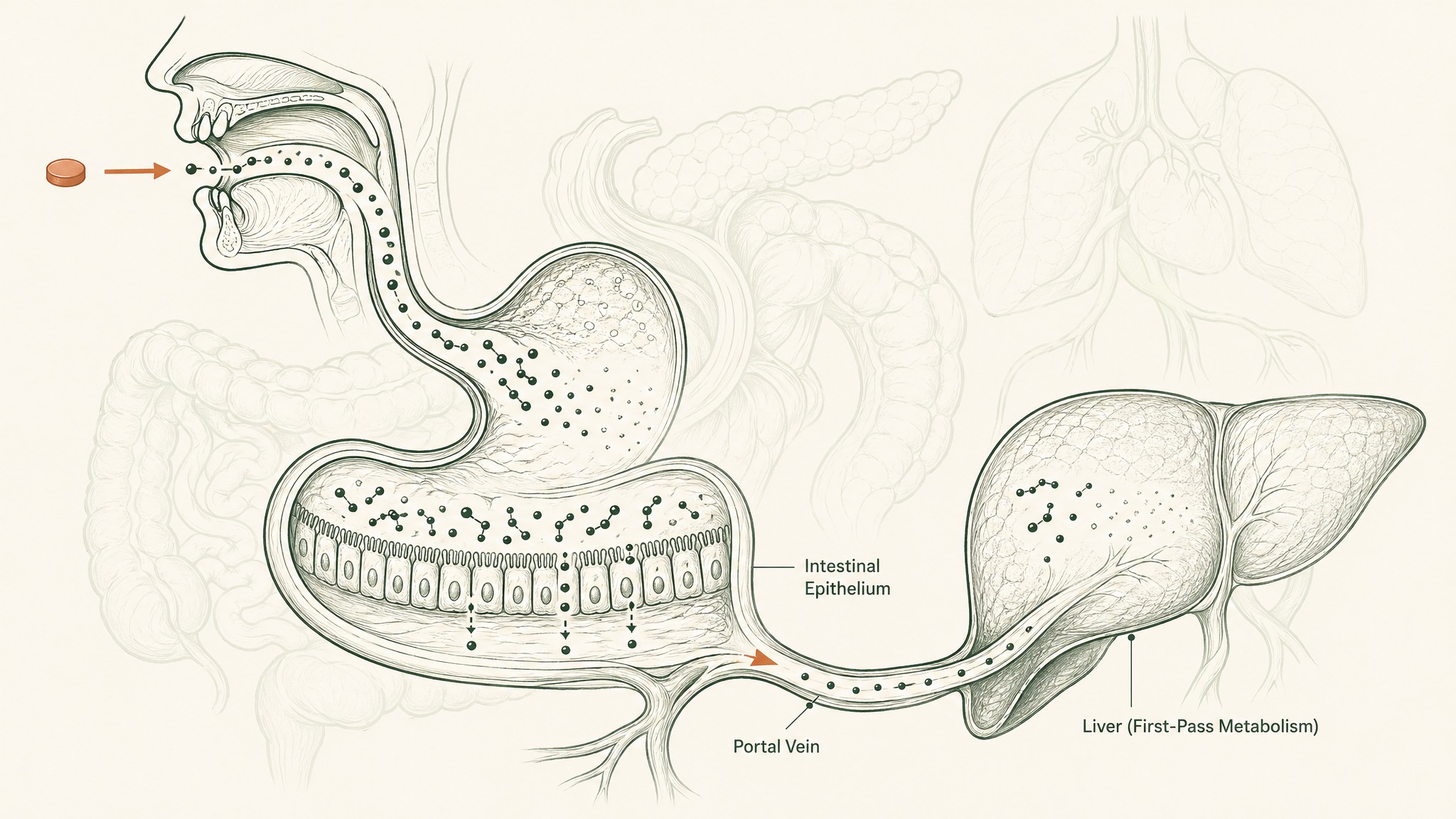
The compounding effect is the point. Oral peptide delivery faces enzymatic degradation, poor cellular membrane permeability, and first-pass hepatic metabolism as sequential obstacles (Verma et al. 2021, Drug Dev Res, PMID 33988872). Each one works on whatever the previous stage left over, not just reducing a percentage of the original. Multiplied together, you arrive at a sub-2-percent bioavailability figure that isn't an exaggeration or a pessimistic estimate. It's the documented outcome.
The One Absorption Pathway That Exists — And Why Most Peptides Miss It
There is one mechanism in the gut that actively absorbs intact peptides: a transporter protein called PepT1, encoded by the SLC15A1 gene. PepT1 sits on the surface of intestinal cells and transports peptides inward via a hydrogen-ion co-transport mechanism.
The practical limitation is the substrate window. PepT1 transports di-peptides and tri-peptides: fragments containing exactly two or three amino acids (Freeman 2015, World J Gastrointest Pharmacol Ther, PMC4419090). Its biological purpose is absorbing the end products of food protein digestion. It's designed for the output of the very destruction process described above.
Most research peptides are considerably longer than three amino acids. BPC-157 is 15 amino acids. GHK-Cu is three amino acids, but it carries a copper ion that changes its uptake characteristics significantly. The transporter that could theoretically help absorb intact peptides simply isn't built for the longer peptide sequences that define most research protocols. It's a small window in a very tall wall.
The Numbers: What Pharmaceutical Engineering Has Actually Achieved
The under-2-percent baseline is consistent across unmodified oral peptides. But the more instructive question is: what's the best that serious pharmaceutical engineering has ever achieved for oral peptide bioavailability?
The answer is oral semaglutide, marketed as Rybelsus for type 2 diabetes. It's co-formulated with a proprietary absorption enhancer called SNAC (sodium N-(8-[2-hydroxybenzoyl]amino) caprylate), which works by creating a transient rise in local gastric pH that allows semaglutide to absorb across the stomach wall before it reaches the enzymatic environment of the small intestine. The dosing protocol is strict: 30 minutes before food, no more than 120 ml of water.
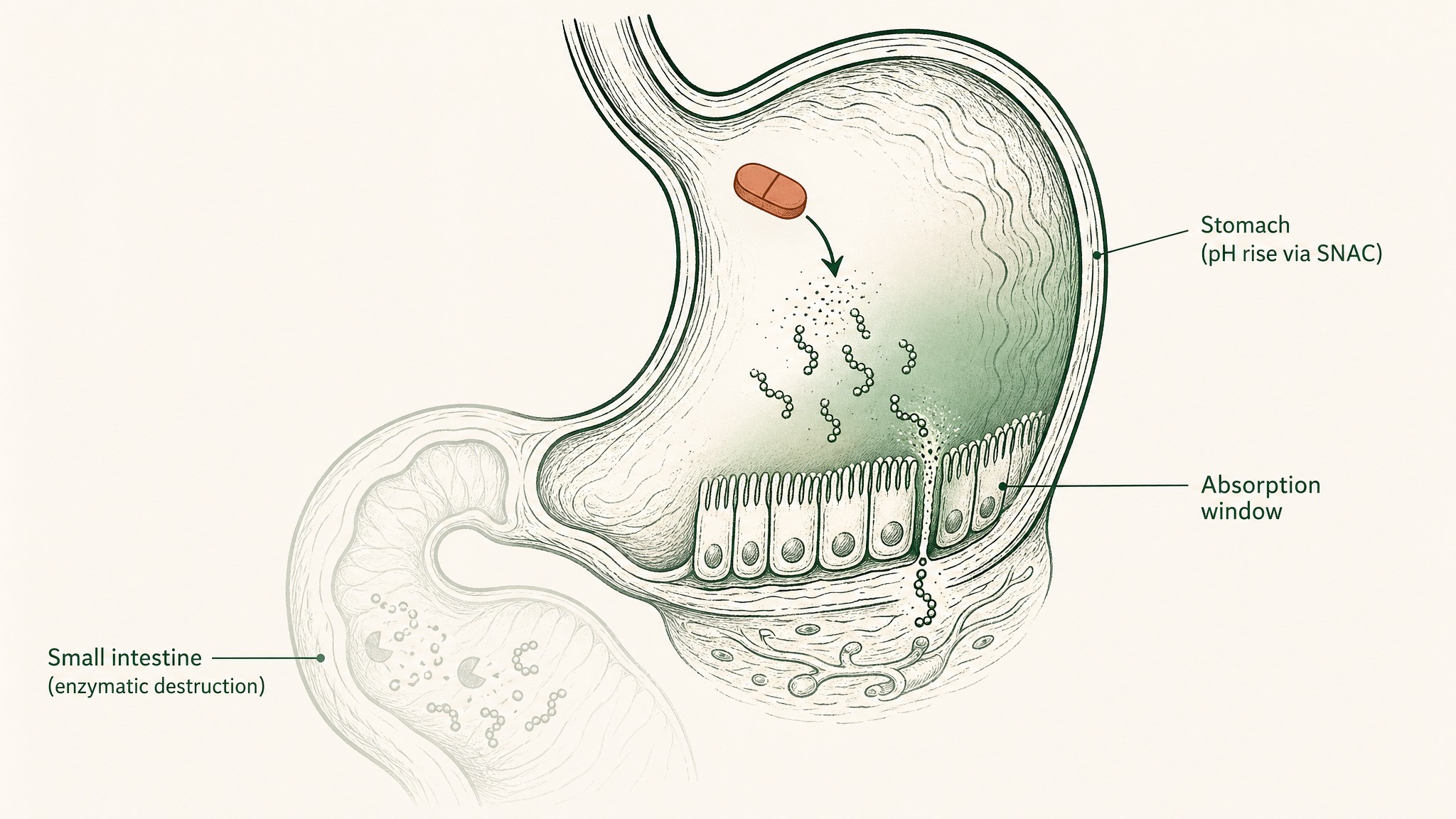
Under those conditions, absolute bioavailability: 0.8 percent (Overgaard et al. 2021, Clin Pharmacokinet, PMID 33969456).
Research suggests this 0.8 percent figure is the ceiling for commercially engineered oral peptide delivery: the product of substantial pharmaceutical investment targeting one specific absorption mechanism for one specific compound. Oral peptide capsules without that proprietary SNAC formulation have no access to the gastric absorption pathway it creates. They face the full destruction sequence from pH 1.5 onward.
Rybelsus works not because 0.8 percent bioavailability is impressive. It isn't. It works because semaglutide is potent enough at low concentrations that even 0.8 percent of the administered dose produces a meaningful effect. That pharmacological potency doesn't transfer to most research peptides, which typically require more reliable systemic exposure to produce the outcomes researchers are studying.
What This Means When You're Looking at the Label
Members experience a disconnect between oral peptide supplement labelling and actual outcomes partly because the absorption problem isn't visible on the packaging. The milligrams listed on the label are accurate for what's in the capsule. What happens to those milligrams between your mouth and your bloodstream isn't disclosed.
Injectable peptide formulations remain the most commonly used format in research specifically because subcutaneous injection bypasses every stage in this cascade. Depositing a compound directly into vascularised subcutaneous tissue means the gastrointestinal tract is never involved, and the dose-to-exposure relationship is predictable (Zhang et al. 2020, Int J Pharm, PMID 32622810). Users report greater protocol consistency with delivery routes that circumvent the gut's destruction sequence, which is exactly what the pharmacokinetics would predict.
What Bypassing the Gut Actually Looks Like
Two routes of administration genuinely bypass the destruction sequence. The first is subcutaneous injection, which is the reference standard for systemic peptide delivery in research. The second is sublingual delivery: absorbing a compound across the mucosal tissue beneath your tongue.
The anatomy is the key. The underside of your tongue is lined with non-keratinised epithelium, thinner, more permeable, and less enzymatically aggressive than any surface in the gastrointestinal tract. Compounds absorbed here drain into the sublingual and lingual veins, entering the internal jugular vein and superior vena cava, and from there into systemic circulation (Zhang, Zhang & Streisand 2002, Clin Pharmacokinet, PMID 12126458). The stomach, the intestinal enzymes, the epithelial barrier, the tight junctions: none of them are in this pathway. And because the absorbed compound enters systemic circulation before it reaches the liver, first-pass hepatic metabolism doesn't apply either.
Research suggests sublingual delivery is particularly well-suited to compounds with low molecular weight, high potency, and long biological half-life (Harris & Robinson 1992, J Pharm Sci, PMID 1619560). The sublingual mucosa does have its own permeability constraints: an unformulated peptide placed under the tongue doesn't automatically absorb. A purposefully engineered delivery vehicle is still needed to address the mucosal barrier. But that's one barrier, not five stacked in sequence.
When sublingual delivery is properly formulated for a peptide, the data is instructive. Sublingual insulin nanoparticles achieved absolute bioavailability of 24.1 percent relative to a subcutaneous injection reference (Patil & Devarajan 2016, Drug Deliv, PMID 24901208). Those are compound-specific and formulation-specific numbers in an animal model. What they demonstrate is the order-of-magnitude difference the sublingual route can produce versus swallowed oral delivery, when the formulation is engineered for the route rather than just placed in a capsule.
Key Takeaways
- Oral peptide bioavailability is consistently documented below 1 to 2 percent for unmodified compounds. The gut's destruction sequence (gastric acid, pepsin, brush-border peptidases, the epithelial barrier, tight junctions, and hepatic first-pass metabolism) operates sequentially, each stage reducing whatever the previous one left (Renukuntla et al. 2013, PMC3680128; Verma et al. 2021, PMID 33988872).
- The intestinal PepT1 transporter only accepts di-peptides and tri-peptides. Most research peptides exceed this chain-length threshold and don't use this pathway meaningfully (Freeman 2015, PMC4419090).
- The highest commercially engineered oral peptide bioavailability on record is 0.8 percent, achieved by oral semaglutide with SNAC absorption enhancer under strict dosing conditions (Overgaard et al. 2021, PMID 33969456). Standard oral peptide capsules don't have access to this technology.
- Subcutaneous injection bypasses the gut entirely and delivers to vascularised tissue where dose-to-exposure relationships are predictable. It remains the reference standard in research (Zhang et al. 2020, PMID 32622810).
- Sublingual delivery bypasses the gastrointestinal tract and first-pass hepatic metabolism via the oral mucosa. Formulated sublingual peptide preparations have demonstrated bioavailability of 24.1 percent versus subcutaneous in research settings (Patil & Devarajan 2016, PMID 24901208).
- What's on the label and what reaches your bloodstream are two very different numbers when the delivery route goes through your gut.
If you want to explore a protocol built on a sublingual delivery system specifically engineered to address the absorption barriers described here, the RESTORE Protocol is where VERO's formulation approach starts.
This content is published for research purposes only and does not constitute medical advice. It is not intended to diagnose, treat, cure, or prevent any disease. Anyone considering a peptide research protocol should consult a qualified clinician before beginning.
RESTORE™
BPC-157Engineered around 500mcg BPC-157. Accelerates deep tissue and joint recovery
Clinical Context
Important Notice: VERO protocols are nutritional and systemic optimisation formats. They are not intended to diagnose, treat, cure, or prevent any medical condition. These statements have not been evaluated by the Food and Drug Administration.