GHK-Cu targets skin collagen, tissue repair, and antioxidant defence simultaneously. Here's how to build a structured protocol around all three — what to stack, how to cycle, and what to track.
A GHK-Cu protocol is a structured research approach that targets three biological domains simultaneously: skin collagen production, tissue repair pathways, and antioxidant defence at the cellular level. That scope is unusual for a single compound. Most molecules are narrow specialists. GHK-Cu isn't. This content is published for research purposes only.
GHK-Cu stands for glycine-histidine-lysine — a tripeptide (three amino acids linked in a chain) attached to a copper ion. Your body makes it naturally. Loren Pickart and M.M. Thaler first isolated it from human serum in 1973 (PMID 4349963). The biological rationale for supplemental GHK-Cu protocols starts with a measurable decline: research suggests plasma GHK levels run at around 200 nanograms per millilitre in your twenties and fall to around 80 ng/mL by the time you're 60 — a roughly 60% drop across four decades (Pickart & Margolina, Int J Mol Sci 2018, PMC6073405). You're studying the effects of restoring a signalling molecule your body used to produce more of.
What follows covers the three research domains, how to structure cycles and stacking, and what endpoints to track.
What GHK-Cu Is and Why Plasma Levels Decline with Age
Think of GHK-Cu as a signalling molecule your body made and gradually stopped prioritising. For most of your early life, it circulates in your blood at concentrations high enough to keep collagen synthesis, tissue maintenance, and antioxidant gene expression running at a solid baseline. Once you hit your thirties and forties, that concentration starts falling — quietly, without obvious symptoms, but measurably.
The 60% decline between your twenties and sixties isn't just a number on a chart. Research suggests GHK modulates expression by at least 50% in 31.2% of human genes, covering pathways involved in tissue remodelling, antioxidant defence, anti-inflammatory signalling, and neurological maintenance at the same time (Pickart & Margolina, Int J Mol Sci 2018, PMC6073405). A 2014 genomic analysis confirmed GHK changed expression by at least 50% in 32.1% of 13,424 human genes across 22,277 probe sets (Pickart, Vasquez-Soltero & Margolina, BioMed Res Int 2014, PMC4180391).
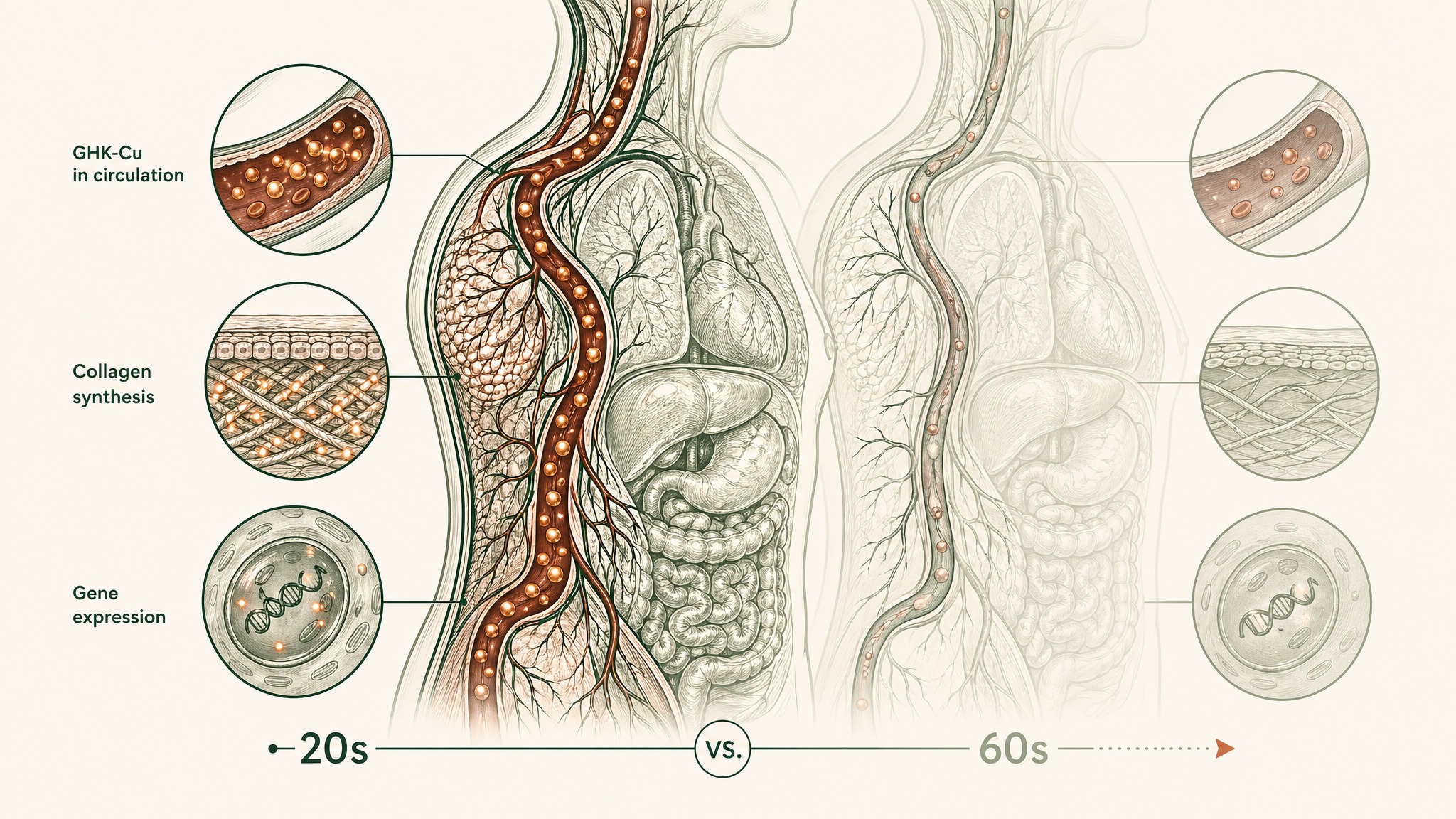
A third of your entire genome. One small signalling molecule your body already recognises as its own.
The practical implication for protocol design: you're not choosing between skin, repair, and longevity outcomes. You're deciding which endpoints you're going to measure — because all three are active in the same protocol.
The Three Research Domains a GHK-Cu Protocol Covers
GHK-Cu and Skin Collagen: What the Research Measured
The skin research on GHK-Cu is the most clinically accessible of the three domains and the one with the clearest human study data.
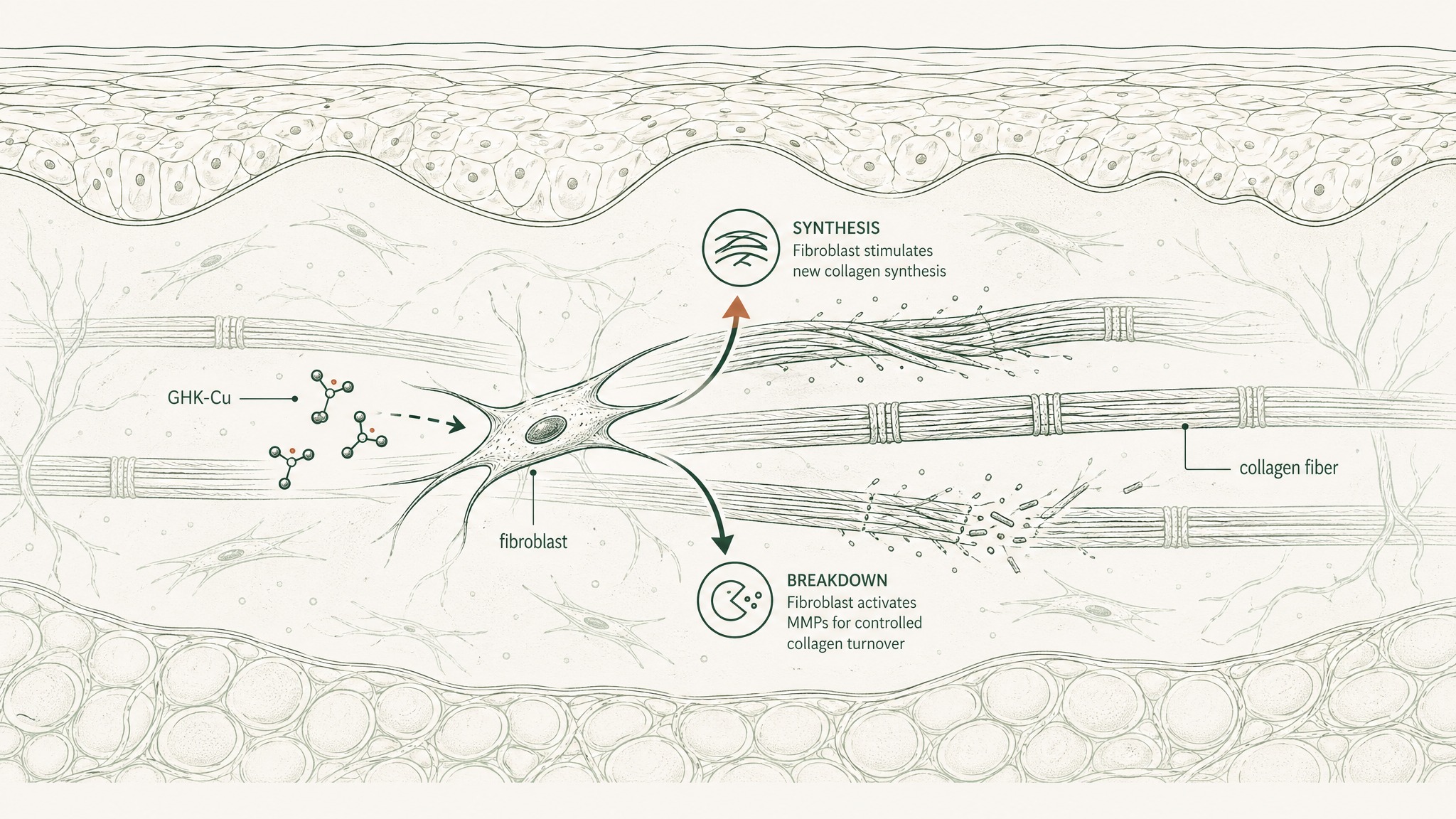
In a placebo-controlled study, researchers applied GHK-containing creams to thighs for one month and measured collagen production changes via skin biopsy and immunohistological analysis. Increases in collagen were found in 70% of the women in the GHK-Cu group — compared to 50% with vitamin C cream and 40% with retinoic acid, a vitamin A derivative widely used in anti-aging skincare (Pickart, Vasquez-Soltero & Margolina, BioMed Res Int 2015, PMC4508379). Vitamin C and retinoic acid are two of the most well-researched skin compounds available. GHK-Cu outperformed both in the same study conditions.
What makes the result more interesting than the number alone: at concentrations as low as 1 to 10 nanomolar — an amount so small it barely registers on a standard measurement scale — GHK-Cu didn't just stimulate collagen synthesis. It also regulated the enzymes responsible for controlled collagen turnover. It was coordinating both the production and the breakdown cycle simultaneously, which is what produces organised dermal structure rather than excess or disorganised tissue.
Members experience observable skin changes most consistently in the 4–8 week window of a protocol. Texture tends to shift first; density and elasticity take longer to accumulate.
GHK-Cu and Tissue Repair: What Research Protocols Found
Tissue repair research on GHK-Cu consistently uses engineered carrier systems rather than free peptide — because unprotected peptide doesn't survive intact to reach the target tissue. This detail matters when you're reading the studies: the delivery system isn't background information. It's the variable that made the biology accessible.
Wang et al. (2017) encapsulated GHK-Cu in liposomes — spherical lipid carriers that function like a protective envelope around the peptide — to study wound repair in a mouse scald model. Results: a 33.1% increased rate of human umbilical vein endothelial cell proliferation (the cells that build new blood vessels were multiplying significantly faster), wound closure shortening to approximately 14 days post-injury, and documented upregulation of both VEGF and FGF-2 — the growth factors that drive new blood vessel formation and tissue regeneration (Wang et al., Wound Repair Regen 2017, PMID 28370978). The same compound without the liposomal carrier showed weaker results in the same study conditions.
The VEGF (vascular endothelial growth factor) finding is worth pausing on. VEGF is the signalling protein that tells your body to grow new blood vessels into damaged tissue. It's the same pathway that TB-500 targets — via a different molecular mechanism. That's why GHK-Cu and TB-500 frequently appear together in repair-focused research stacks.
A separate 2023 study used thermodynamically stable ionic liquid microemulsions to study GHK-Cu delivery to hair follicles. The engineered system achieved approximately threefold improvement in local peptide delivery compared to non-formulated controls and activated the Wnt/β-catenin signalling pathway — a key regulator of follicle cycling — in mouse models, alongside documented increases in VEGF and HGF expression (Liu et al., Bioactive Materials 2023, PMID 38026438). Two different research teams, two different tissue targets, both treating the carrier system as the essential variable.
GHK-Cu Antioxidant Effects: HDAC Inhibition and Free Radical Reduction
The antioxidant research on GHK-Cu is arguably the least well-known of the three domains, and also one of the most relevant to longevity-focused protocols.
A 2012 review of GHK-Cu's antioxidant and anti-inflammatory properties found a 75% reduction in lipid peroxidation — the process by which free radicals damage the fats in cell membranes — in gastric tissue samples. The researchers also documented suppression of TGF-beta and TNF-alpha, two pro-inflammatory signalling proteins that drive chronic systemic inflammation when they stay elevated for too long (Pickart, Vasquez-Soltero & Margolina, Oxid Med Cell Longev 2012, PMC3359723).
The same paper identified GHK as a strong HDAC inhibitor. Here's what that means in plain terms: HDACs (histone deacetylases) are enzymes that physically compress DNA, silencing gene expression in the process. When HDACs become overactive — which happens as cells age — healthy genes get turned off even when the cellular environment would otherwise support their activity. GHK-Cu's HDAC-inhibiting activity suggests it may be partially reversing that silencing.
Research suggests this is part of why GHK-Cu appears in longevity protocols alongside NAD+ precursors and sirtuin activators. NAD+ works through the sirtuin pathway. GHK-Cu works through HDAC inhibition and broad gene modulation. Different molecular routes toward a similar goal: maintaining gene expression patterns associated with younger biological age.
Structuring a GHK-Cu Protocol: Cycles, Delivery, and Stacking
Delivery Format Is a Biological Gate, Not a Product Detail
The most common error in GHK-Cu protocol design is treating delivery as a cosmetic product-selection detail rather than a biological prerequisite. Every tissue repair and systemic finding cited above — the VEGF upregulation, the HDAC inhibition, the gene modulation numbers — was produced using engineered delivery systems: subcutaneous injection, liposomal encapsulation, or ionic liquid microemulsions. Free peptide swallowed in a plain capsule doesn't connect to those research outcomes. Neither does an unformulated sublingual drop without mucoadhesive polymers to extend mucosal contact time and permeation enhancers to get the peptide across the membrane.
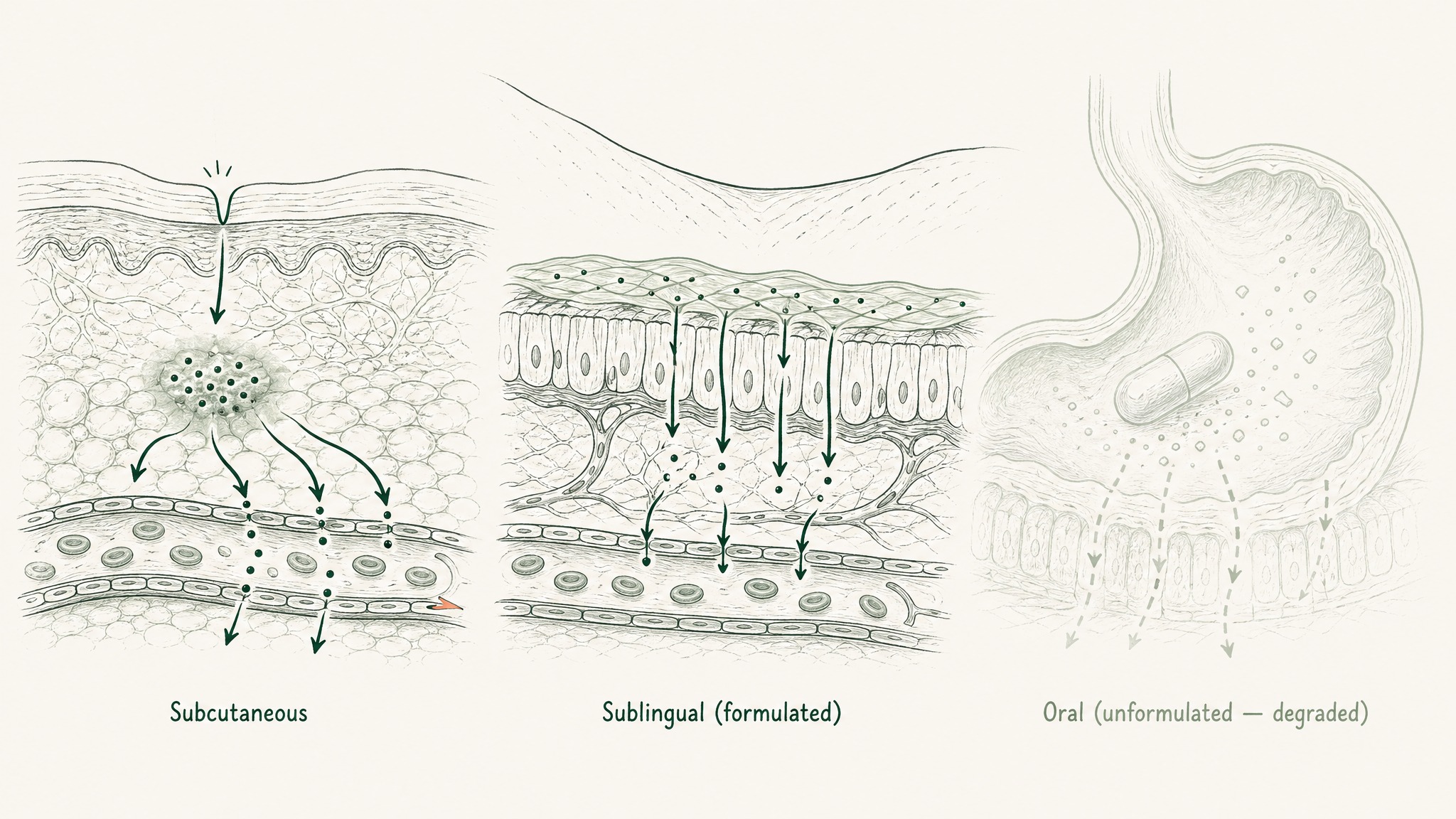
The full delivery route comparison — subcutaneous injection vs. formulated sublingual vs. topical vs. plain oral — is covered in detail in GHK-Cu Sublingual: Why Delivery Format Determines Protocol Outcomes. The short version: subcutaneous injection and properly formulated sublingual are the two routes with a credible path to systemic exposure. Everything else requires specific engineering to approach that bar, and a plain product without that engineering doesn't get you to your bloodstream.
Cycle Length and Observation Windows
The skin collagen research used a one-month measurement window and found observable changes in collagen production (PMC4508379). The tissue repair data in Wang et al. 2017 measured acute endpoints over days to weeks (PMID 28370978). Those time windows give you a rough framework for what's observable when:
- Acute tissue repair contexts: 14–21 days for initial response signals
- Skin and collagen endpoints: 4–8 weeks for measurable changes
- Gene regulatory and antioxidant effects: Longer cycles; not a short-window outcome
A common structure in GHK-Cu research protocols runs 8–12 weeks on, followed by a rest period before reassessment. That cycle length isn't mandated by a single study — it's consistent with how longevity peptide research tends to structure cyclical observation windows and creates enough runway to see both the short-term tissue repair signals and the longer-term skin and antioxidant endpoints.
Stacking GHK-Cu with Other Research Peptides
GHK-Cu's three-domain activity makes it a natural foundation for stacks rather than a standalone protocol. Here's how the most common combinations work at the biological level:
GHK-Cu + BPC-157 (repair stack): BPC-157 targets tendon, gut, and musculoskeletal repair primarily via upregulation of growth hormone receptor expression and nitric oxide synthase activation — an inflammatory-resolution pathway. GHK-Cu adds ECM remodelling (ECM — extracellular matrix — is the structural scaffolding between and around your cells) and antioxidant signalling. Together you're covering the inflammatory resolution side and the structural repair side in parallel rather than sequentially. These are complementary mechanisms, not overlapping ones.
GHK-Cu + TB-500 (angiogenic repair stack): TB-500 (thymosin beta-4) works via actin polymerisation, promoting cell migration to injury sites and new blood vessel formation. GHK-Cu's VEGF and FGF-2 upregulation findings (PMID 28370978) are directly additive here: both compounds support the angiogenic pathway — new blood vessel growth into repaired tissue — from different molecular starting points.
GHK-Cu + NAD+ precursors (longevity stack): For longevity-focused protocols, GHK-Cu and NAD+ precursors (NMN or NR) address gene expression from complementary angles. NAD+ activates sirtuins, a family of proteins involved in DNA repair and metabolic regulation. GHK-Cu inhibits HDACs and modulates broad gene expression patterns. Users report this combination as synergistic at the energy and cognitive clarity level over longer protocol windows, though the molecular mechanisms are distinct.
What to Track in a GHK-Cu Protocol
The research endpoints translate into practical markers you can document during a protocol:
Skin texture and density. The collagen research measured changes via biopsy — not available outside a clinical setting. But skin density, elasticity, and fine-line changes are observable without clinical tools and align with the one-month collagen endpoint from the 2015 study (PMC4508379). Weekly photos in consistent lighting give you a comparable baseline.
Recovery from physical stress. VEGF and FGF-2 upregulation (PMID 28370978) suggest GHK-Cu has relevance for post-exercise muscle and connective tissue recovery. How quickly your body bounces back from training stress is an observable endpoint in the 2–4 week window.
Inflammatory markers in bloodwork. TGF-beta and TNF-alpha are exactly what the 2012 antioxidant paper (PMC3359723) measured. Both are standard on longevity-focused panels. Drawing blood at the start and end of a cycle gives you a before/after comparison on the markers the research actually tracked.
Sleep quality and cognitive clarity. The HDAC inhibition and neurological maintenance findings from the 2012 paper provide the mechanistic basis for cognitive endpoints in GHK-Cu protocols. These are subjective — but consistent enough that members experience changes in sleep depth and mental clarity as among the earlier signals in the first few weeks of a well-delivered protocol.
Key Takeaways
- GHK-Cu is a naturally occurring tripeptide first isolated from human serum in 1973 (PMID 4349963). Plasma levels decline roughly 60% between your twenties and sixties — from ~200 ng/mL to ~80 ng/mL (PMC6073405).
- Research suggests GHK modulates expression by at least 50% in 31.2% of human genes, covering tissue remodelling, antioxidant defence, and neurological maintenance simultaneously (PMC6073405; PMC4180391). A single protocol covers all three domains — you're choosing what to measure, not what to restrict.
- Skin collagen research recorded increases in 70% of women over one month, outperforming vitamin C cream (50%) and retinoic acid (40%) in the same study conditions (PMC4508379).
- Tissue repair research using liposomal GHK-Cu showed wound closure shortening to approximately 14 days and a 33.1% increase in endothelial cell proliferation, alongside documented VEGF and FGF-2 upregulation (PMID 28370978). A 2023 ionic liquid microemulsion study showed approximately threefold improvement in local peptide delivery and Wnt/β-catenin pathway activation for hair follicle research (PMID 38026438).
- Antioxidant research found a 75% reduction in lipid peroxidation, suppression of TGF-beta and TNF-alpha, and HDAC-inhibiting activity with implications for gene expression maintenance and cognitive health (PMC3359723).
- Delivery format is a biological gate: the research findings were produced using engineered carrier systems. Subcutaneous injection and properly formulated sublingual are the two routes with credible paths to systemic exposure.
- Common stacks: GHK-Cu + BPC-157 for repair (complementary anti-inflammatory and structural mechanisms), GHK-Cu + TB-500 for angiogenic support (both support VEGF pathway from different angles), GHK-Cu + NAD+ precursors for longevity (HDAC inhibition + sirtuin activation).
- Observation windows: 14–21 days for acute repair signals, 4–8 weeks for skin and collagen, longer cycles for antioxidant and gene regulatory endpoints.
Ready to explore a structured GHK-Cu sublingual protocol? Start RADIANCE →
This content is published for research and educational purposes only. It is not intended to diagnose, treat, cure, or prevent any disease, and it does not constitute medical advice. Readers should consult a qualified healthcare professional before making any decisions about their own health. Statements on this page have not been evaluated by the FDA. VERO's peptide products are supplied for research purposes only.
RADIANCE™
Coming SoonEngineered around 5mg GHK-Cu. Increases dermal thickness and systemic collagen synthesis
Clinical Context
Important Notice: VERO protocols are nutritional and systemic optimisation formats. They are not intended to diagnose, treat, cure, or prevent any medical condition. These statements have not been evaluated by the Food and Drug Administration.

