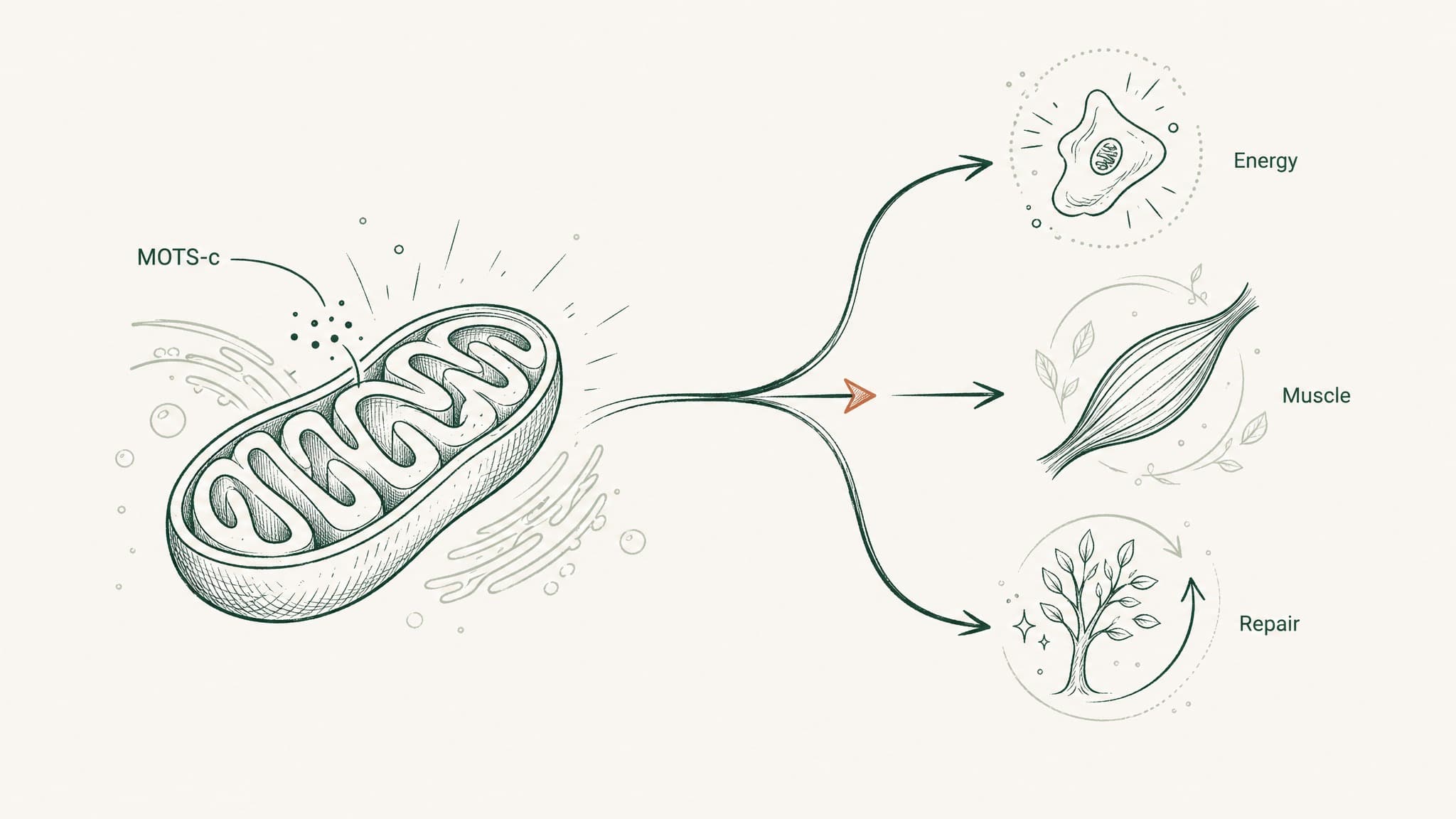
Research into MOTS-c — a peptide encoded inside your mitochondrial DNA — points to five specific domains where the evidence is most substantive: metabolic health, exercise performance, cellular stress defence, bone density, and cardiovascular function.
MOTS-c sits in an unusual position in peptide science. It was only described in the scientific literature in 2015 — which means researchers have had just over a decade to understand what it actually does. In that time, five benefit domains have accumulated the most consistent evidence: metabolic health, exercise performance, cellular stress protection, bone density, and cardiovascular function.
The evidence is not equal across those five areas. Some have strong mechanistic data backed by human observational studies. Others have compelling results in animal models but no human trial yet. A couple are genuinely early — the rationale is coherent but the confirmation is still forthcoming. This article maps each domain honestly, so you know what's well-supported and what's still being established. This content is published for research purposes only and is not intended as medical advice.
If you haven't yet read the companion piece on what MOTS-c is and how it works, that's the right starting point — it covers the mechanism, the discovery story, and why your body makes less of this peptide as you age. This article picks up where that one ends.
Metabolic Health: The Benefit With the Most Evidence
Think of your cells' relationship with insulin like a lock-and-key system. Insulin is the key; your cells' insulin receptors are the locks. When the system is working well, the key turns easily and glucose from your bloodstream enters the cell to be used as fuel. When insulin resistance develops — as it does for many people over time, driven by diet, stress, reduced activity, and ageing — the locks start to jam. More and more insulin is needed to get the same result. Eventually glucose builds up in the blood rather than getting burned, and your metabolic health begins to deteriorate downstream.
MOTS-c's effect on this system is the most thoroughly documented of its five benefit areas. The original 2015 discovery paper, published in Cell Metabolism by Lee et al. at the University of Southern California, showed that MOTS-c improves insulin sensitivity through a specific pathway. By disrupting the folate cycle — your cells' internal carbon-accounting system — it triggers AMPK (your cells' master energy-shortage sensor) into action. When AMPK activates, your cells pull glucose in more efficiently, burn fat more readily, and become more responsive to insulin. In mice, this translated to prevention of both diet-induced obesity and age-related insulin resistance (Lee et al. 2015, Cell Metab, PMID 25738459).
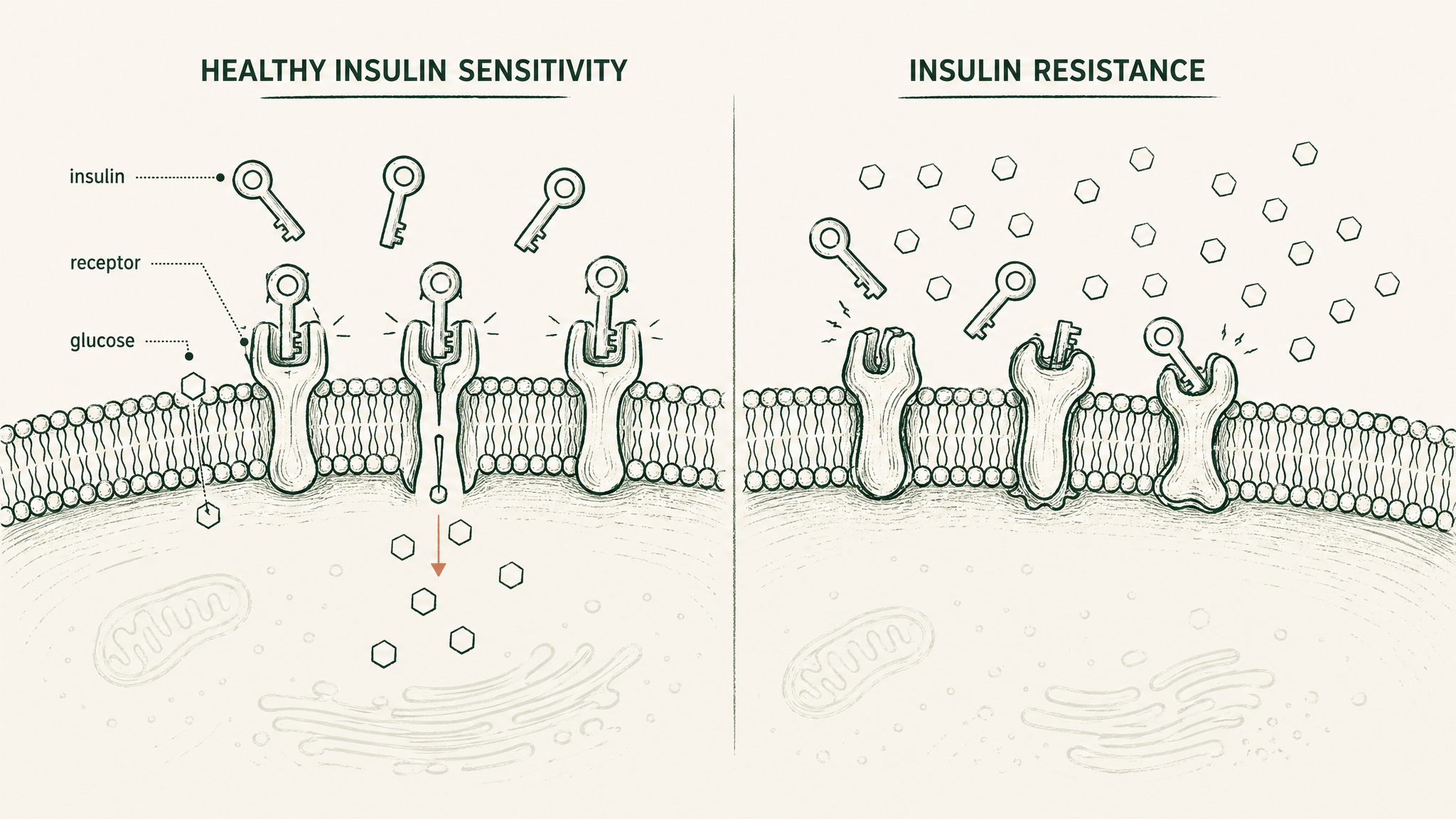
That's animal data. What makes metabolic health the strongest benefit domain is that the human observational picture points in the same direction. A cross-sectional study of 225 people, published in Frontiers in Endocrinology by Ramanjaneya et al. in 2019, found that circulating MOTS-c was significantly lower in people with type 2 diabetes compared to metabolically healthy controls. Levels tracked inversely with HbA1c — the rolling three-month average of blood sugar control — and with age. The worse the metabolic picture, the lower the circulating MOTS-c (Ramanjaneya et al. 2019, Front Endocrinol, PMID 31214116).
That's observational data, which means it shows correlation, not causation. It tells you MOTS-c moves in step with metabolic health — not that supplementing it will restore metabolic function. But the mechanistic foundation from animal work and the consistent human pattern together make this the most coherent body of MOTS-c evidence in the published record. No other benefit domain has both a clearly mapped mechanism and a multi-hundred-person human study showing the association.
Exercise Performance and Recovery
Here's a useful way to think about MOTS-c and exercise: it's part of the molecular signal your muscles send to the rest of your body during a hard training session. When you do sustained aerobic work — running, cycling, rowing — your muscle fibres produce MOTS-c and release it into the bloodstream. It becomes part of the chemical conversation that explains how cardio produces benefits in your brain, your liver, and your metabolic tissues, not just in the muscles doing the actual contracting.
A 2021 study in Nature Communications by Reynolds et al. established this clearly. Their work demonstrated that exercise induces MOTS-c expression in human skeletal muscle, and then asked what happens when you administer MOTS-c directly to aged mice — three doses a week. The results were significant: improved physical capacity, better skeletal muscle metabolism, and extended healthspan, even when the treatment started in late life (Reynolds et al. 2021, Nat Commun, PMID 33473109). The mice getting MOTS-c in their final months were outperforming untreated age-matched mice on physical performance measures.
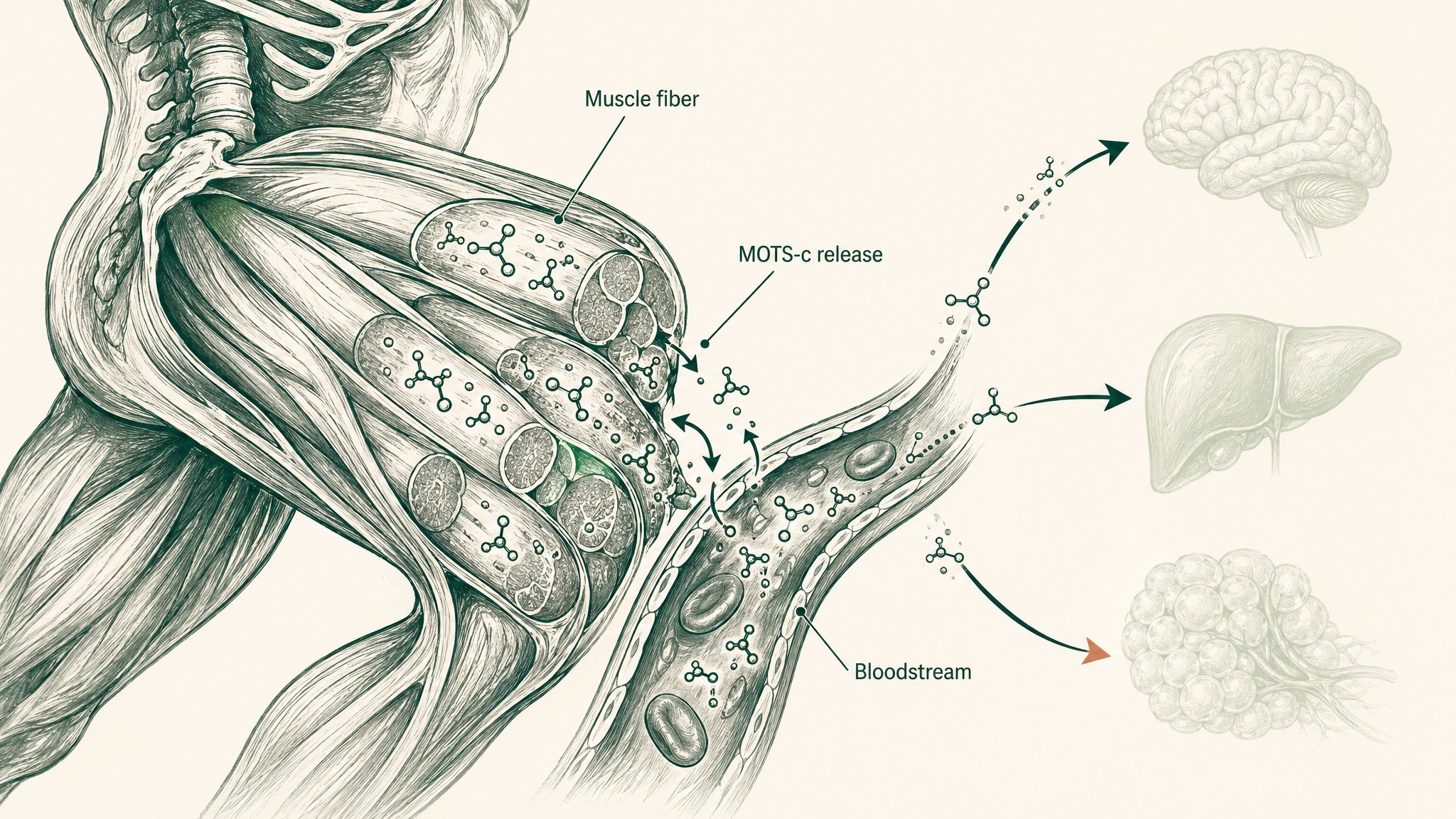
A separate 2021 study by Von Walden et al., published in the Journal of Applied Physiology, brought this into human blood samples. Thirty healthy adults were assigned to endurance exercise (cycling), resistance exercise (leg work), or a control condition. MOTS-c levels showed a trend toward increasing in the endurance group but not in the resistance group — consistent with the Reynolds finding that MOTS-c is specifically linked to aerobic exercise rather than strength work (Von Walden et al. 2021, J Appl Physiol, PMID 34351816). Users report interest in whether MOTS-c will eventually be trackable as a direct biomarker of aerobic fitness — and that's exactly the kind of practical application the research community is now working toward.
The gap that remains: no published human interventional trial has yet tested whether exogenous MOTS-c improves exercise performance. The animal evidence is strong. The human data shows the association exists. The causal confirmation in humans is the next required step.
Cellular Stress Protection
Think of your cells as running a constant balancing act between damage and repair. On one side: reactive molecules called free radicals, produced as a normal byproduct of energy production and environmental stress. On the other: your cells' antioxidant defence systems, which neutralise those molecules and repair what they damage. When ageing tips that balance toward damage, cells deteriorate faster. MOTS-c has a role in shifting the balance back.
Under conditions of metabolic stress — when blood glucose drops sharply, or reactive molecules accumulate — MOTS-c doesn't stay in the fluid inside the cell. It moves into the nucleus, where your chromosomal DNA is stored. Once there, it activates NRF2 — the master switch for your cells' antioxidant gene programme — triggering a cascade of protective responses. It's not acting as an antioxidant itself; it's activating the machinery that produces your cells' own antioxidant defences. That's a meaningfully different mechanism.
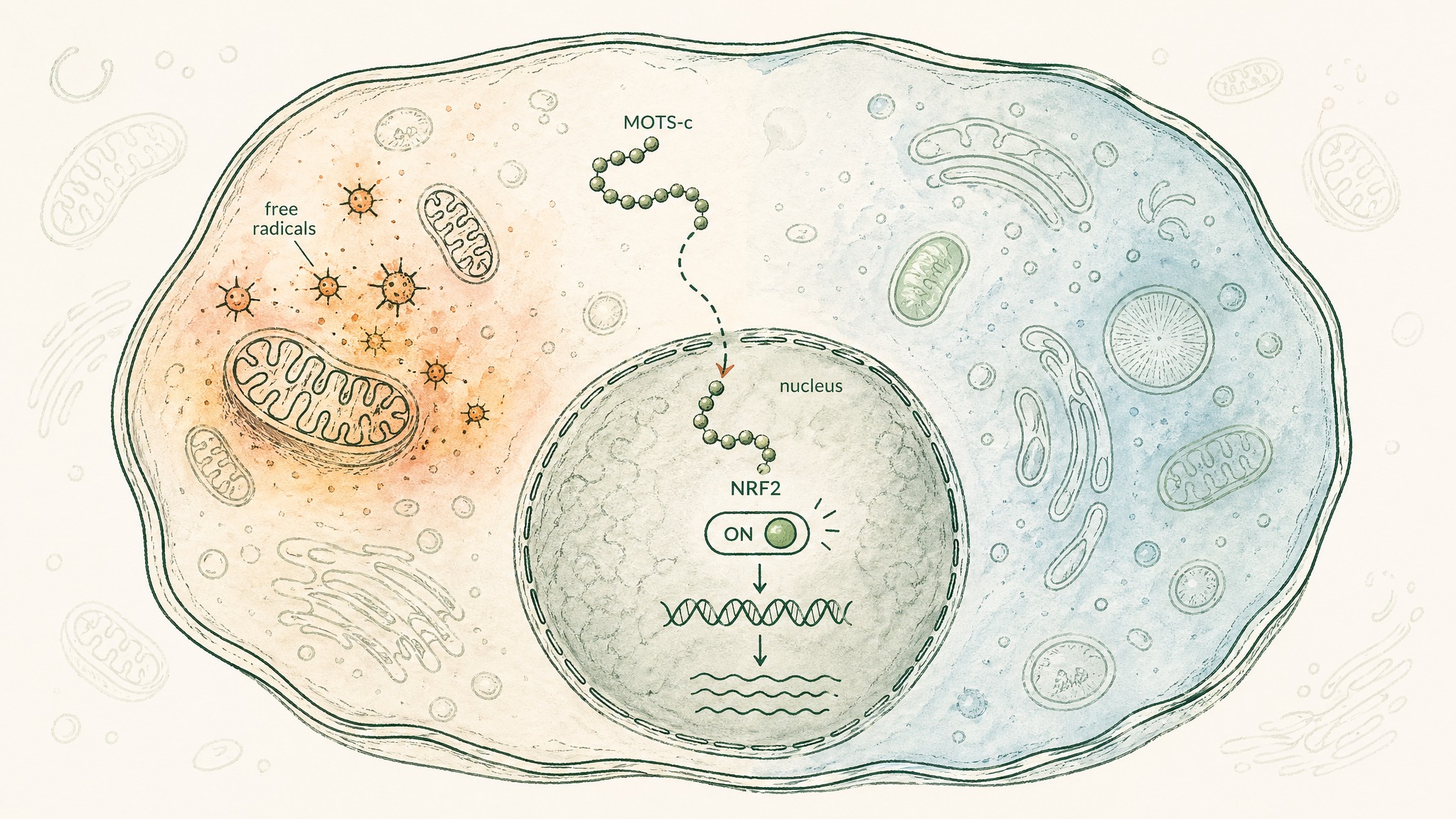
This was mapped in a 2018 paper in Cell Metabolism by Kim, Son, Benayoun, and Lee, which identified MOTS-c's nuclear translocation for the first time and showed it coordinates gene expression from two separate genomes simultaneously — mitochondrial and nuclear — which had not been described before in this context (Kim et al. 2018, Cell Metab, PMID 29983246). The AMPK pathway is also involved: MOTS-c activates antioxidant response elements through an AMPK-dependent mechanism, meaning its metabolic effects and its stress-protective effects share overlapping machinery.
The 2022 review by Mohtashami et al. in the International Journal of Molecular Sciences positions this cellular protective mechanism as a core reason MOTS-c research has expanded into longevity science — not just metabolic disease management. If cells can maintain balance under stress for longer, the downstream effects on tissue ageing may be significant (Mohtashami et al. 2022, Int J Mol Sci, PMID 36233287). The mechanism here is among the best-characterised of MOTS-c's documented effects. The translation to measurable longevity outcomes in humans is still an open question.
Bone Health: An Emerging Research Area
Bone loss is one of the less intuitive MOTS-c research areas, but it follows a coherent logic once you understand how bone actually works.
Think of bone as a living tissue in constant renovation. Two types of specialised cells are doing the work: osteoblasts build new bone; osteoclasts break it down. That renovation cycle is how your skeleton repairs micro-damage and responds to the demands placed on it. In youth, the cycle runs approximately in balance. Ageing disrupts that balance: breakdown gradually outpaces construction, and bone mineral density declines. At a certain threshold, that's osteoporosis.
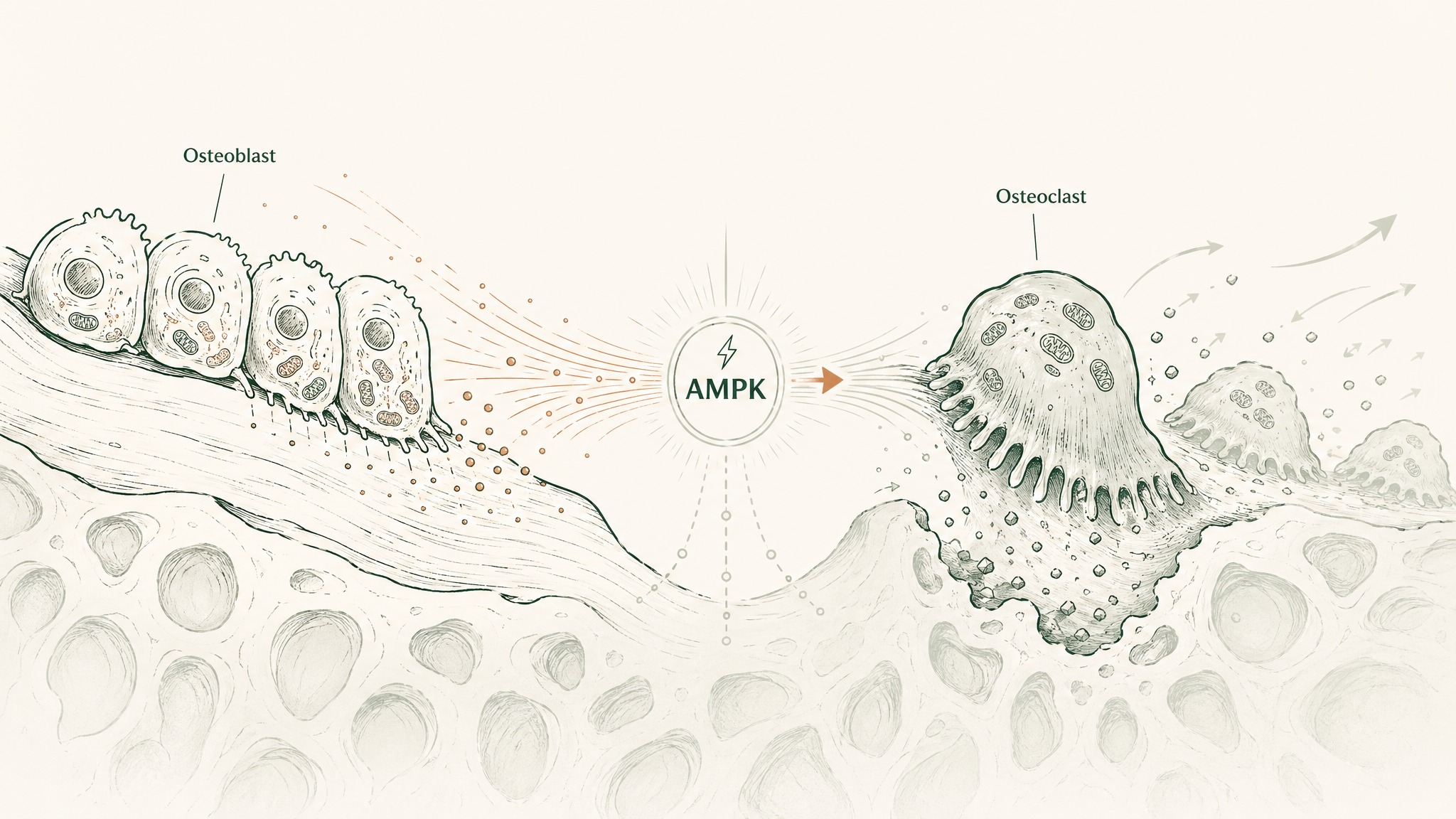
MOTS-c's relevance here comes from its role as a regulator of cellular energy balance. Osteoblasts and osteoclasts are metabolically active cells — they depend on efficient energy metabolism to do their work. The AMPK pathway that MOTS-c activates has been shown to influence the osteoblast-osteoclast balance in cell and animal studies, shifting it toward bone-building activity. The Mohtashami 2022 review lists osteoporosis explicitly among the age-related conditions within MOTS-c's therapeutic scope (Mohtashami et al. 2022, Int J Mol Sci, PMID 36233287). The 2023 review by Zheng et al. in Frontiers in Endocrinology similarly identifies bone health as one of the emerging research areas warranting investigation (Zheng et al. 2023, Front Endocrinol, PMID 36761202).
This is genuinely early-stage. There are no published human trials specifically on MOTS-c and bone density. What exists is mechanistic logic and the inclusion of osteoporosis in the major recent reviews. Research suggests this will become an active primary research area over the next few years, particularly given the intersection of bone health with the ageing-related decline in MOTS-c levels.
Cardiovascular Function
The cardiovascular research area overlaps significantly with metabolic health — which makes sense, because insulin resistance and cardiovascular disease share many of the same upstream causes.
Here's the connection: when cells handle glucose and fat inefficiently, excess lipids can accumulate in the walls of blood vessels. When oxidative stress is high, blood vessel linings take more damage over time. When inflammation is chronic, arterial stiffness increases. MOTS-c's documented effects — improving cellular energy metabolism, activating antioxidant defences via NRF2, reducing the metabolic conditions that promote fat accumulation — all have downstream relevance for cardiovascular health, even if the cardiovascular effects haven't yet been studied in isolation.
The 2023 Zheng review identifies cardiovascular disease as one of the primary therapeutic targets for ongoing MOTS-c research, alongside metabolic disease and ageing (Zheng et al. 2023, Front Endocrinol, PMID 36761202). The Mohtashami 2022 review covers cardiovascular disease as part of its scope on age-related conditions where declining MOTS-c levels are thought to be mechanistically significant (Mohtashami et al. 2022, Int J Mol Sci, PMID 36233287).
Like bone health, this is an area where the rationale is scientifically sound and the research direction is clear, but primary human studies targeting cardiovascular outcomes specifically don't exist yet. Members experience the research landscape here as moving faster than the published literature fully reflects — and that's plausible given how recently the broader research community took MOTS-c seriously as a therapeutic target. The cardiovascular work will likely follow the metabolic research by several years.
Reading the Evidence Honestly
Five domains. Different evidence tiers. Here's a direct summary.
Metabolic health has the most substantive evidence: the original mechanistic discovery in mice, corroborated by multiple subsequent animal studies, and a 225-person human cross-sectional showing MOTS-c tracks with glycaemic health markers. No human RCT yet — but the mechanistic picture and the human observation point in the same direction.
Exercise performance has strong animal data — late-life MOTS-c administration extending healthspan and physical capacity in aged mice — alongside early human evidence that aerobic exercise raises circulating MOTS-c. The interventional human study hasn't happened yet.
Cellular stress protection is the most thoroughly characterised mechanistically. The nuclear translocation and NRF2 activation pathway has been confirmed across independent research groups. Translation to measurable longevity outcomes in humans is the remaining gap.
Bone health and cardiovascular function are legitimately early-stage. The mechanistic argument is coherent — both systems depend on the cellular energy metabolism that MOTS-c regulates. Both appear in the major recent reviews as active research targets. Primary studies on these systems in humans are still ahead.
No published human randomised controlled trial of exogenous MOTS-c administration exists as of mid-2026. That's the honest state of the field. What does exist is a mechanistic foundation that's unusually solid for a molecule only described a decade ago, and a growing human observational picture pointing in consistent directions. The interventional confirmation is what the research needs — and it's what the major reviews from 2022 and 2023 identify as the necessary next step.
Key Takeaways
- Metabolic health has the strongest evidence base: MOTS-c activates AMPK to improve insulin sensitivity and glucose uptake in animals, and a 225-person human cross-sectional study found circulating MOTS-c significantly lower in people with type 2 diabetes, tracking inversely with HbA1c (Lee et al. 2015, PMID 25738459; Ramanjaneya et al. 2019, PMID 31214116).
- Exercise is the most reliable known stimulus for endogenous MOTS-c production; aerobic training raises it in human muscle and circulation, while late-life MOTS-c administration in aged mice improved physical performance and extended healthspan (Reynolds et al. 2021, PMID 33473109; Von Walden et al. 2021, PMID 34351816).
- Cellular stress protection is the most mechanistically detailed benefit domain: MOTS-c moves into the cell nucleus under metabolic stress and activates NRF2-driven antioxidant defences, coordinating signals from mitochondrial and nuclear genomes simultaneously (Kim et al. 2018, PMID 29983246).
- Bone health and cardiovascular function are emerging areas — both included in the major 2022–2023 reviews as active research targets, but without dedicated primary human studies yet (Mohtashami et al. 2022, PMID 36233287; Zheng et al. 2023, PMID 36761202).
- Circulating MOTS-c declines with age, which is the foundational reason supplementation research is underway — the body makes less of the signal, and researchers are asking what happens when you replace it.
- No published human RCT of exogenous MOTS-c exists as of mid-2026. The mechanistic foundation is strong; the interventional human confirmation is what the field is working toward.
Interested in exploring longevity protocols informed by the latest peptide research? Explore VERO Protocols →
This content is published for research and educational purposes only. It is not intended to diagnose, treat, cure, or prevent any disease, and it does not constitute medical advice. Readers should consult a qualified healthcare professional before making any decisions about their own health. Statements on this page have not been evaluated by the FDA. VERO's peptide products are supplied for research purposes only.
RESTORE™
BPC-157Engineered around 500mcg BPC-157. Accelerates deep tissue and joint recovery
Clinical Context
Important Notice: VERO protocols are nutritional and systemic optimisation formats. They are not intended to diagnose, treat, cure, or prevent any medical condition. These statements have not been evaluated by the Food and Drug Administration.