MOTS-c is a 16-amino-acid signalling peptide encoded inside your mitochondrial DNA. Here's how it was discovered, what its mechanism actually does, and what the research shows about levels declining with age.
Your mitochondria are famous for one thing. They're the powerhouses of the cell — you've heard it since biology class. But they do something less talked about: they also send chemical signals. MOTS-c is one of those signals. And researchers are only just beginning to understand what it means that your body makes less of it as you age. This content is for research purposes only.
Where MOTS-C Comes From
Every cell in your body contains mitochondria — tiny structures that convert food and oxygen into ATP, the energy currency your cells run on. Think of them as the engine rooms inside each cell, doing the burning so everything else can function.
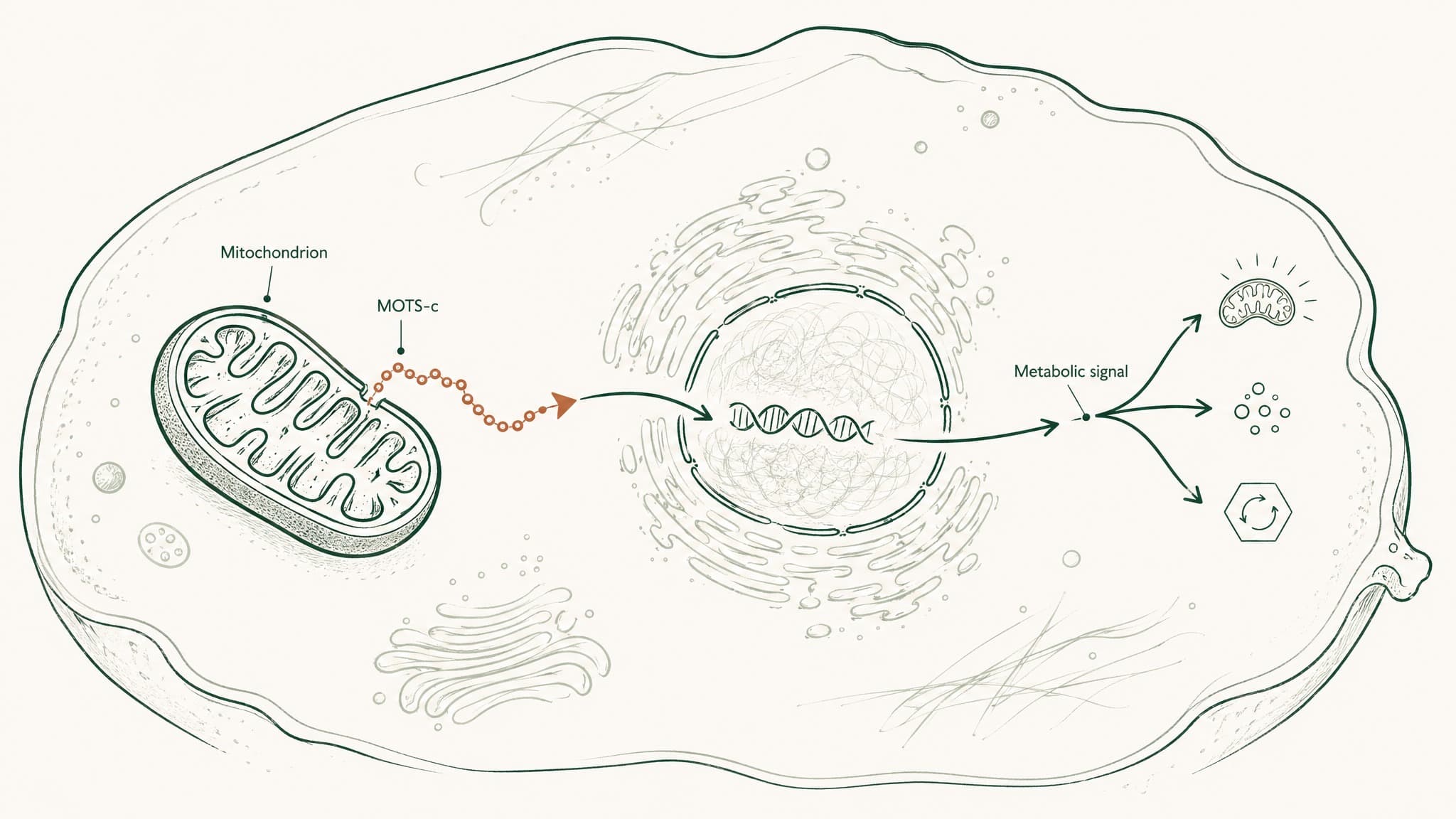
But your mitochondria don't just generate energy. They have their own DNA — a separate, circular strand of genetic code inherited entirely from your mother, distinct from the chromosomal DNA in your cell's nucleus. Most of what's in that DNA codes for machinery the mitochondria need to do their job. A small section also codes for something else: signalling molecules.
MOTS-c is one of them. It's a 16-amino-acid peptide — amino acids being the building blocks proteins are assembled from — encoded inside a stretch of mitochondrial DNA called MT-RNR1 (Lee et al. 2015, Cell Metab, PMID 25738459). The fact that it originates from mitochondrial DNA rather than from the chromosomes in your nucleus makes it genuinely unusual. It belongs to a recently identified family called mitochondrial-derived peptides, or MDPs — a class that barely existed in the scientific literature fifteen years ago.
How Scientists Discovered It
The discovery story is more recent than you'd expect.
In 2015, a research team at the University of Southern California led by Changhan Lee and Pinchas Cohen published a paper in Cell Metabolism identifying MOTS-c for the first time. They weren't specifically hunting for it. They were studying the 12S ribosomal RNA gene in mitochondrial DNA — a region widely assumed to be purely structural, like the scaffolding inside a building. No one expected it to produce bioactive signalling peptides.
They found a 16-amino-acid sequence hiding inside it that, when isolated and tested in mice, had measurable effects on metabolism. Fat mice on a high-fat diet didn't develop insulin resistance. Older mice with already-established age-related insulin resistance responded better to insulin after receiving the peptide. The effects were significant enough to name it: MOTS-c stands for Mitochondrial ORF of the Twelve S rRNA type-c (Lee et al. 2015, PMID 25738459).
Worth sitting with: this peptide your body has been producing your entire life was only described in the scientific literature eleven years ago.
The Mechanism: What MOTS-C Actually Does Inside Your Cells
The mechanism works in two stages, and each has a useful analogy to make it concrete.
Stage One: Disrupting Your Cells' Carbon Accounting
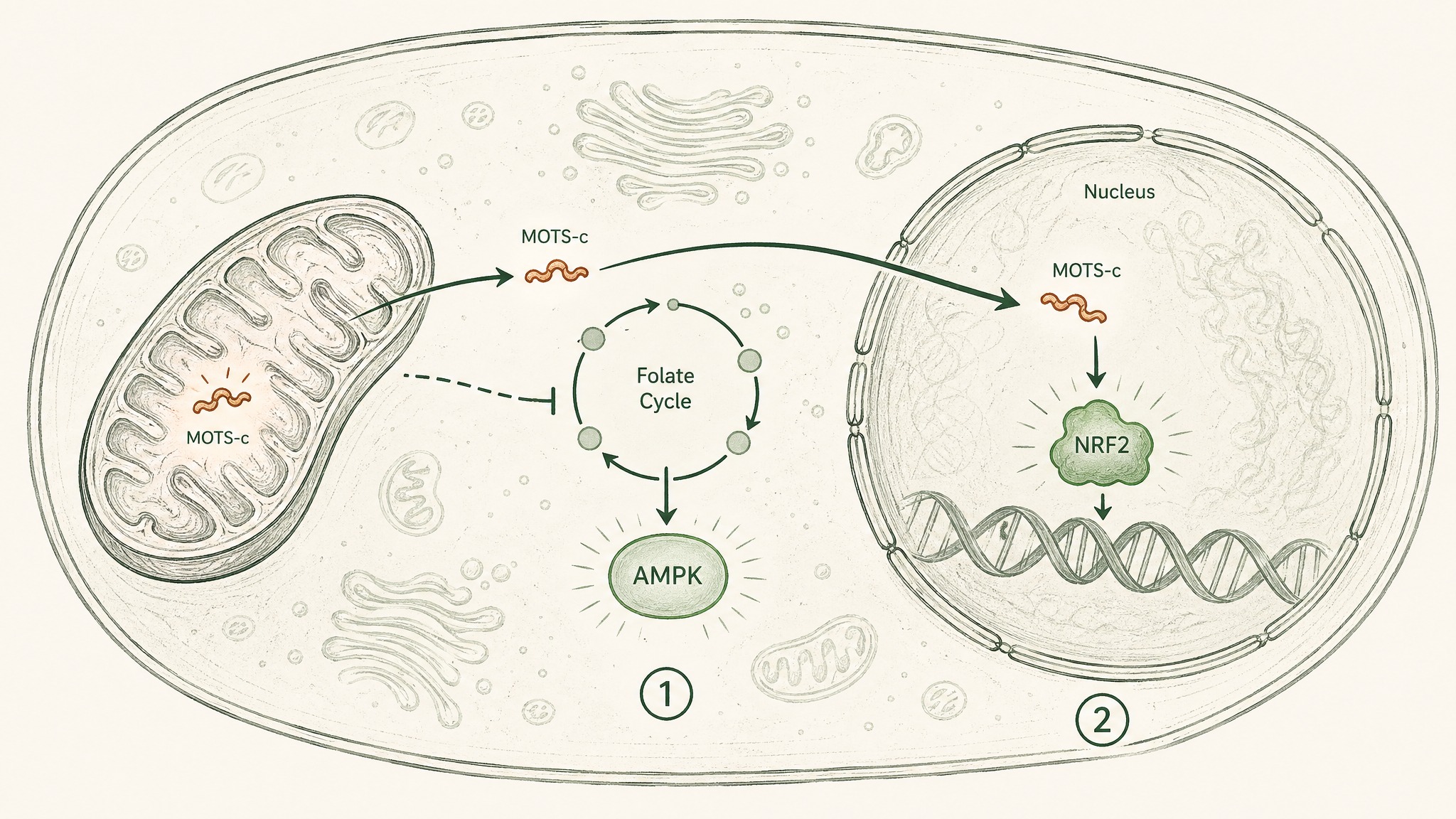
Think of the folate cycle as your cells' internal carbon-accounting system. It's a metabolic pathway that recycles tiny molecular fragments — called one-carbon units — to keep amino acid production, DNA synthesis, and energy balance ticking over. When it's running smoothly, your cell knows what resources it has and how to spend them.
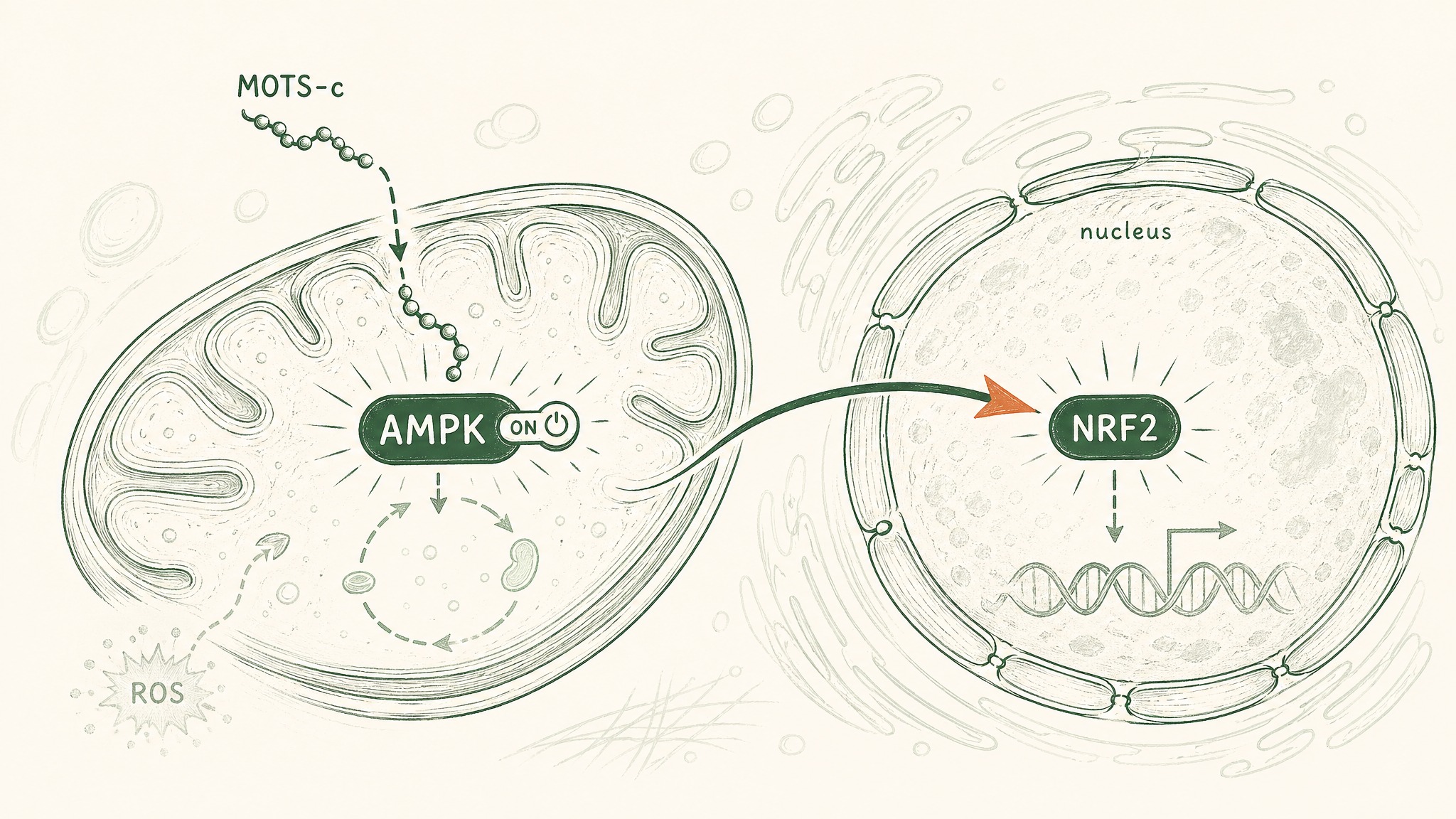
MOTS-c interferes with that accounting system on purpose. By blocking a key step in the folate cycle and a connected pathway called purine biosynthesis — where your cells build the components of DNA and RNA — it creates a controlled resource shortage. Your cell's energy sensor, an enzyme called AMPK, detects the shortfall and switches on.
AMPK is what researchers call a master metabolic regulator — the sensor your cells use to detect whether they're energy-rich or energy-poor and adjust their behaviour accordingly. When AMPK activates, your cells pull glucose out of the bloodstream more efficiently, burn fat more readily, and become more sensitive to insulin. MOTS-c doesn't flip this switch directly. It sends the upstream metabolic signal that makes AMPK respond (Lee et al. 2015, PMID 25738459).
Stage Two: Going Nuclear Under Stress
There's a second mechanism, and it's stranger. Under conditions of metabolic stress — when blood glucose drops, or when reactive molecules accumulate and start damaging cellular components — MOTS-c doesn't stay in the cytoplasm (the liquid interior of the cell). It moves into the nucleus, the compartment where your chromosomal DNA is stored.
Once inside, it influences which genes get switched on. It activates a molecule called NRF2 — your cells' master switch for antioxidant defence — triggering a cascade of protective genes that help your cells handle and repair oxidative damage. Think of NRF2 as the emergency response coordinator: when MOTS-c arrives in the nucleus and activates it, your cell's repair crew goes to work.
A 2018 paper in Cell Metabolism by Kim, Son, Benayoun, and Lee mapped this process in detail, showing that MOTS-c coordinates signals from both the mitochondrial genome and the nuclear genome — a form of cross-organelle communication that hadn't been described before (Kim et al. 2018, Cell Metab, PMID 29983246).
Crucially, MOTS-c also circulates in the bloodstream. It's not just acting inside the cell that made it. It behaves more like a hormone — produced in one tissue, released into circulation, and capable of signalling to tissues elsewhere in your body. A 2016 review by Lee, Kim, and Cohen described this as "an entirely novel mitochondrial signalling mechanism to regulate metabolism within and between cells" (Lee et al. 2016, Free Radic Biol Med, PMID 27216708).
Why Your MOTS-C Levels Change Over Time
Your MOTS-c levels aren't constant. They shift with your biology — and the pattern of that shift tracks closely with markers of metabolic ageing.
Research suggests that circulating MOTS-c declines as you get older. The 2022 review by Mohtashami et al. in International Journal of Molecular Sciences identifies this age-related decline as one of the core reasons therapeutic research into the peptide is worth pursuing — it's not just that MOTS-c does useful things, it's that your body makes less of it over time (Mohtashami et al. 2022, Int J Mol Sci, PMID 36233287).
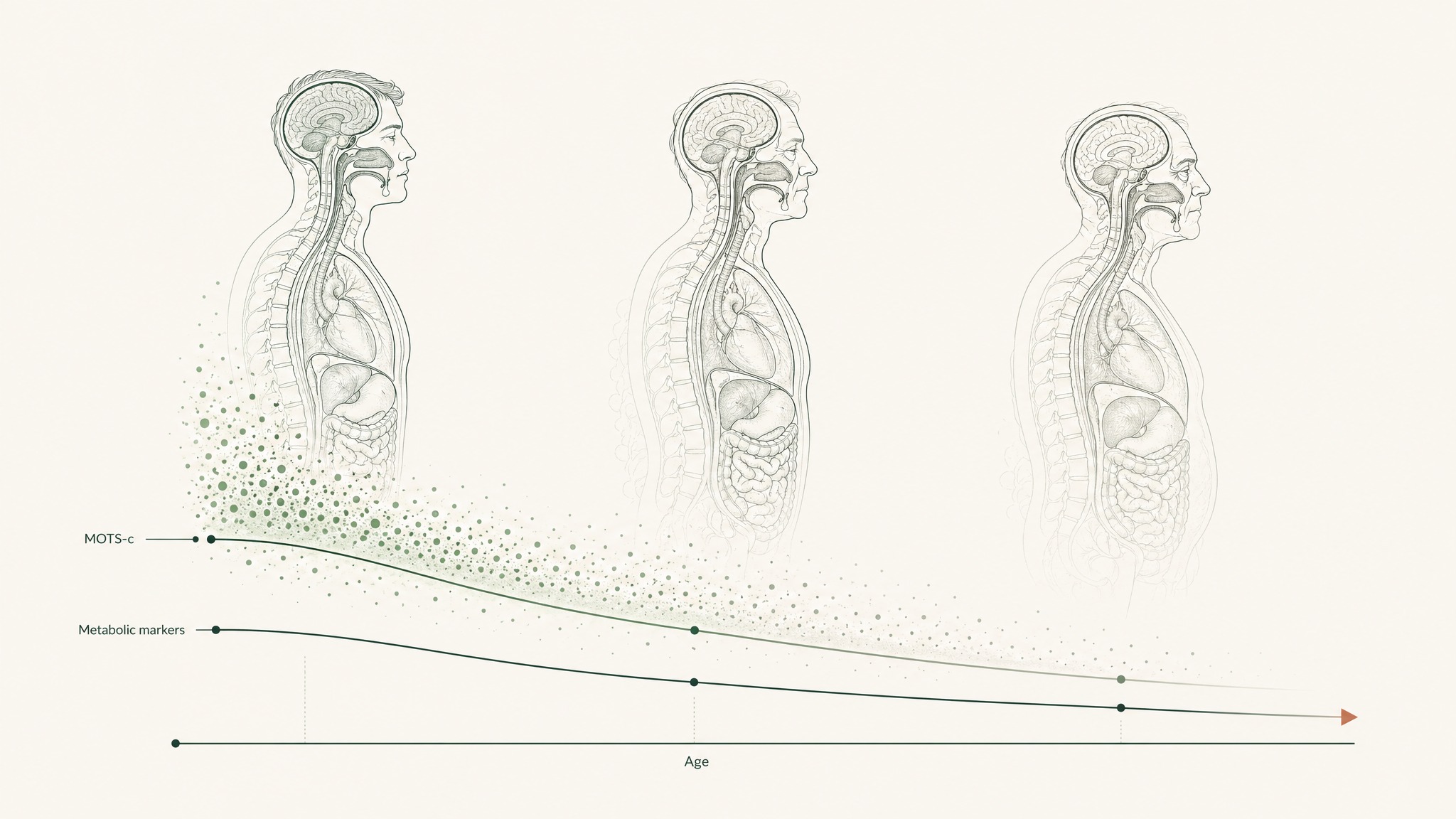
Human observational data makes this pattern visible. A cross-sectional study of 225 people, split across metabolically healthy controls, prediabetic, and type 2 diabetes groups, found that circulating MOTS-c was significantly lower in those with diabetes. Levels correlated negatively with HbA1c — a three-month rolling average of blood sugar control — and with age itself. The worse the metabolic picture, the lower the MOTS-c (Ramanjaneya et al. 2019, Front Endocrinol, PMID 31214116).
What this shows, in plain terms: your circulating MOTS-c level reflects the state of your metabolic health. It's tracking alongside the same biological processes that drive insulin resistance, reduced cellular energy efficiency, and the metabolic deterioration that characterises biological ageing. Whether MOTS-c is a cause, a symptom, or an active participant in that decline is exactly what current research is trying to untangle.
The Exercise Connection
The most reliable thing known to raise your endogenous MOTS-c is exercise — specifically aerobic exercise.
Think of MOTS-c as part of the chemical conversation your muscles send to the rest of your body after a hard run or a long cycle. When you do sustained aerobic activity, your muscle fibres produce MOTS-c and release it into circulation. It's one of several molecular messengers that explain why regular cardio produces benefits in organs and tissues far beyond the muscles actually doing the work.
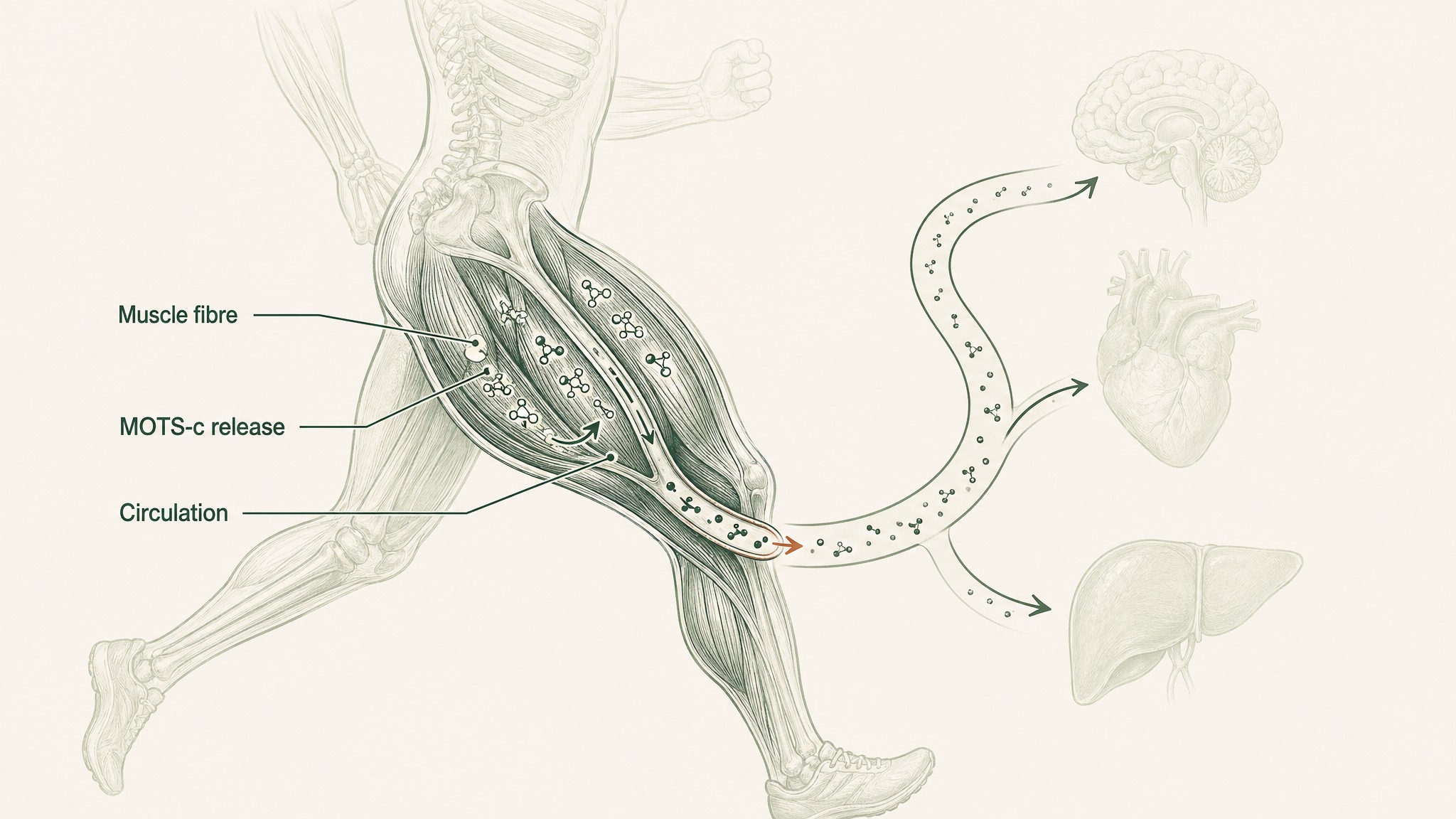
A 2021 study in Nature Communications by Reynolds et al. demonstrated that exercise stimulates MOTS-c expression in human skeletal muscle, and that MOTS-c administration in aged mice — given three times per week — improved physical performance and extended healthspan even in late-life animals (Reynolds et al. 2021, Nat Commun, PMID 33473109). Research suggests this positions MOTS-c as one of the molecular mechanisms linking sustained aerobic activity to its broader anti-ageing effects.
A separate 2021 study by von Walden et al. measured circulating mitochondrial-derived peptides in healthy adults before and after acute endurance exercise, finding that levels rose following aerobic activity but not resistance training (Von Walden et al. 2021, J Appl Physiol, PMID 34351816). Users report curiosity about whether MOTS-c can eventually be tracked as a direct biomarker of aerobic fitness — something the research field is actively developing tools to assess.
Where MOTS-C Fits Among Other Mitochondrial Peptides
MOTS-c doesn't exist in isolation. It belongs to an emerging family of molecules called mitochondrial-derived peptides — short signalling sequences encoded in mitochondrial DNA that act as messengers between the mitochondria, the broader cell, and the organism as a whole.
The first MDP to be identified was humanin, discovered in the early 2000s and found to have neuroprotective and metabolic effects. MOTS-c was the second, described in 2015. Several more have since been characterised, including the SHLP family — SHLP1 through SHLP6, where SHLP stands for Small Humanin-Like Peptide, so named because they're structurally similar to humanin.
What makes this whole class interesting is an evolutionary question it raises. Mitochondria were originally free-living bacteria that merged with primitive eukaryotic cells roughly 1.5 to 2 billion years ago. They kept their own genome ever since — but over evolutionary time, most of the genes they originally carried migrated into the nuclear genome. The fact that some signalling sequences stayed in the mitochondrial genome and continued to evolve suggests they serve coordination roles that are only beginning to be mapped.
The 2023 Zheng et al. review in Frontiers in Endocrinology identifies MOTS-c as the most therapeutically promising MDP currently under investigation, with active research into metabolic disease, cardiovascular function, bone health, and longevity mechanisms (Zheng et al. 2023, Front Endocrinol, PMID 36761202).
Where the Research Stands in 2026
Here's an honest summary of where MOTS-c sits right now, because the evidence isn't all equal.
The strongest tier is cell and animal data. The mechanism — AMPK activation via the folate cycle, nuclear translocation and NRF2 activation under stress — has been confirmed across independent labs in multiple countries. It's internally consistent and reproducible. That's real science with coherent biological logic behind it.
The next tier is human observational data. The 225-person Ramanjaneya study shows MOTS-c declining in line with metabolic disease. The exercise studies show your own endogenous MOTS-c rising with aerobic training. These are genuine associations, not fabricated patterns — but correlation isn't causation. They tell you MOTS-c tracks with these states; they don't prove that artificially raising MOTS-c reverses them.
What doesn't exist yet, as of early 2026, is a published randomised controlled trial of exogenous MOTS-c administration in humans. That's the honest gap. Members experience the field as moving quickly — the Mohtashami and Zheng reviews both identify human trials as the next necessary step — but that confirmation hasn't landed yet.
For a deeper look at the five specific benefit domains where the research is most substantive — metabolic health, exercise response, cellular stress defence, bone health, and cardiovascular function — see the companion article MOTS-C Benefits: What the Research Actually Shows, which covers those areas with full citations and an evidence-tier breakdown.
Key Takeaways
- MOTS-c is a 16-amino-acid peptide encoded inside the 12S ribosomal RNA gene of your mitochondrial DNA — only the second mitochondrial-derived peptide ever identified, first described in 2015 (Lee et al. 2015, PMID 25738459).
- Its primary mechanism is disrupting the folate cycle and purine biosynthesis, which activates AMPK — your cells' master metabolic energy sensor and a key upstream regulator of insulin sensitivity and fat metabolism.
- Under metabolic stress, MOTS-c moves from the cytoplasm into the cell nucleus, activating NRF2-driven antioxidant defence genes and coordinating signals between the mitochondrial and nuclear genomes (Kim et al. 2018, PMID 29983246).
- MOTS-c circulates in the bloodstream like a hormone — it acts on tissues beyond the cell that produced it, which is why its effects are systemic rather than localised (Lee et al. 2016, PMID 27216708).
- Plasma MOTS-c declines with age and correlates with deteriorating metabolic health; in a 225-person study, levels were significantly lower in type 2 diabetes and tracked inversely with HbA1c (Ramanjaneya et al. 2019, PMID 31214116).
- Aerobic exercise is the most reliable known stimulus for endogenous MOTS-c production; it's expressed in skeletal muscle during exercise and rises in circulation after acute endurance activity (Reynolds et al. 2021, PMID 33473109; Von Walden et al. 2021, PMID 34351816).
- No published human RCT of exogenous MOTS-c exists as of early 2026. The evidence is mechanistically strong and observationally consistent — human interventional confirmation is still ahead.
- The 2023 Zheng review identifies MOTS-c as the most therapeutically promising mitochondrial-derived peptide under active investigation (Zheng et al. 2023, PMID 36761202).
Interested in exploring protocols that support your metabolic health and longevity research? Explore VERO Protocols →
This content is published for research and educational purposes only. It is not intended to diagnose, treat, cure, or prevent any disease, and it does not constitute medical advice. Readers should consult a qualified healthcare professional before making any decisions about their own health. Statements on this page have not been evaluated by the FDA. VERO's peptide products are supplied for research purposes only.
RESTORE™
BPC-157Engineered around 500mcg BPC-157. Accelerates deep tissue and joint recovery
Clinical Context
Important Notice: VERO protocols are nutritional and systemic optimisation formats. They are not intended to diagnose, treat, cure, or prevent any medical condition. These statements have not been evaluated by the Food and Drug Administration.